- Skip to Nav
- Skip to Main
- Skip to Footer


How to Use Oral Presentations to Help English Language Learners Succeed
Please try again

Excerpted from “ The ELL Teacher’s Toolbox: Hundreds of Practical Ideas to Support Your Students ,” by Larry Ferlazzo and Katie Hull Sypnieski, with permission from the authors.
Having the confidence to speak in front of others is challenging for most people. For English Language Learners, this anxiety can be heightened because they are also speaking in a new language. We’ve found several benefits to incorporating opportunities for students to present to their peers in a positive and safe classroom environment. It helps them focus on pronunciation and clarity and also boosts their confidence. This type of practice is useful since students will surely have to make presentations in other classes, in college, and/or in their future jobs. However, what may be even more valuable is giving students the chance to take these risks in a collaborative, supportive environment.
Presentations also offer students the opportunity to become the teacher—something we welcome and they enjoy! They can further provide valuable listening practice for the rest of the class, especially when students are given a task to focus their listening.
Research confirms that in order for ELLs to acquire English they must engage in oral language practice and be given the opportunity to use language in meaningful ways for social and academic purposes (Williams & Roberts, 2011). Teaching students to design effective oral presentations has also been found to support thinking development as “the quality of presentation actually improves the quality of thought, and vice versa” (Živković, 2014, p. 474). Additionally, t he Common Core Speaking and Listening Standards specifically focus on oral presentations. These standards call for students to make effective and well-organized presentations and to use technology to enhance understanding of them.
GUIDELINES AND APPLICATION
Oral presentations can take many different forms in the ELL classroom—ranging from students briefly presenting their learning in small groups to creating a multi-slide presentation for the whole class. In this section, we give some general guidelines for oral presentations with ELLs. We then share ideas for helping students develop their presentation skills and describe specific ways we scaffold both short and long oral presentations.
We keep the following guidelines in mind when incorporating oral presentations into ELL instruction:

Length —We have students develop and deliver short presentations (usually 2-4 minutes) on a regular basis so they can practice their presentation skills with smaller, less overwhelming tasks. These presentations are often to another student or a small group. Once or twice a semester, students do a longer presentation (usually 5-8 minutes), many times with a partner or in a small group.
Novelty —Mixing up how students present (in small groups, in pairs, individually) and what they use to present (a poster, a paper placed under the document camera, props, a slide presentation, etc.) can increase engagement for students and the teacher!
Whole Class Processing -- We want to avoid students “tuning out” during oral presentations. Not only can it be frustrating for the speakers, but students also miss out on valuable listening practice. During oral presentations, and in any activity, we want to maximize the probability that all students are thinking and learning all the time. Jim Peterson and Ted Appel, administrators with whom we’ve worked closely, call this “whole class processing” (Ferlazzo, 2011, August 16) and it is also known as active participation. All students can be encouraged to actively participate in oral presentations by being given a listening task-- taking notes on a graphic organizer, providing written feedback to the speaker, using a checklist to evaluate presenters, etc.
Language Support —It is critical to provide ELLs, especially at the lower levels of English proficiency, with language support for oral presentations. In other words, thinking about what vocabulary, language features and organizational structures they may need, and then providing students with scaffolding, like speaking frames and graphic organizers. Oral presentations can also provide an opportunity for students to practice their summarizing skills. When students are presenting information on a topic they have researched, we remind them to summarize using their own words and to give credit when using someone else’s words.
Technology Support —It can’t be assumed that students have experience using technology tools in presentations. We find it most helpful using simple tools that are easy for students to learn (like Powerpoint without all the “bells and whistles” or Google Slides). We also emphasize to students that digital media should be used to help the audience understand what they are saying and not just to make a presentation flashy or pretty. We also share with our students what is known as “The Picture Superiority Effect”-- a body of research showing that people are better able to learn and recall information presented as pictures as opposed to just being presented with words (Kagan, 2013).
Groups -- Giving ELLs the opportunity to work and present in small groups is helpful in several ways. Presenting as a group (as opposed to by yourself) can help students feel less anxious. It also offers language-building opportunities as students communicate to develop and practice their presentations. Creating new knowledge as a group promotes collaboration and language acquisition--an ideal equation for a successful ELL classroom!
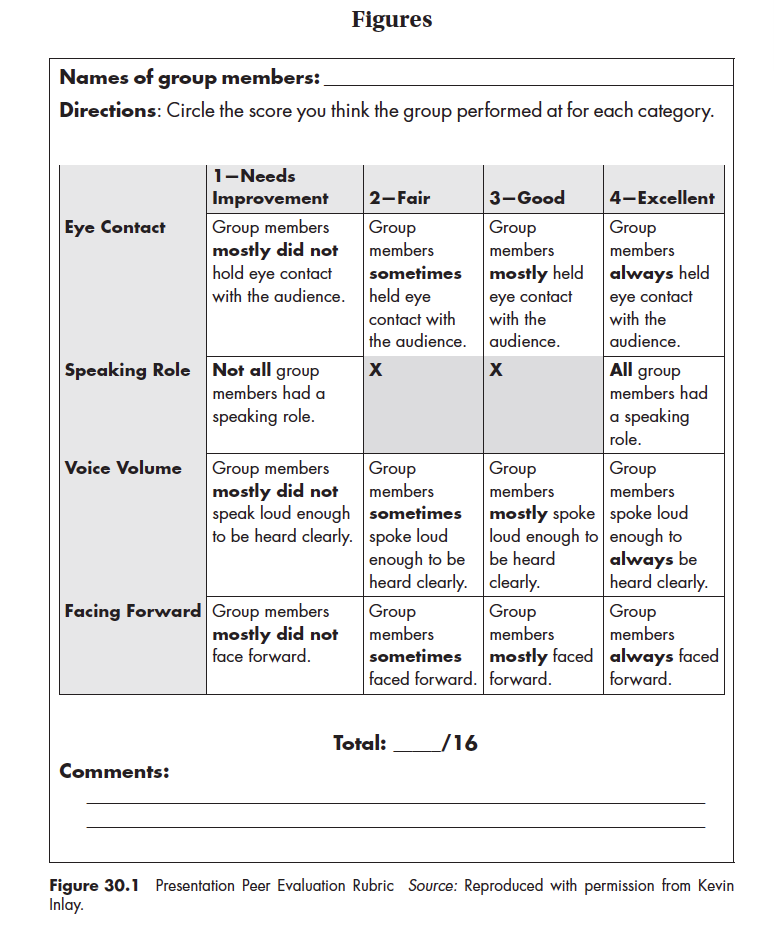
Teacher feedback/student evaluation --The focus of oral presentations with ELL students should be on the practice and skills they are gaining, not on the grade or “score” they are earning. Teachers can give out a simple rubric before students create their presentations. Then students can keep these expectations in mind as they develop and practice their presentations. The teacher, or classmates, can then use the rubric to offer feedback to the speaker. We also often ask students to reflect on their own presentation and complete the rubric as a form of self-assessment. Figure 30.1 – “Presentation Peer Evaluation Rubric” , developed by talented student teacher Kevin Inlay (who is now a teacher in his own classroom), is a simple rubric we used to improve group presentations in our ELL World History class.
Teaching Presentation Skills
We use the following two lesson ideas to explicitly teach how to develop effective presentation skills:
LESSON ONE: Speaking and Listening Do’s and Don’ts
We help our students understand and practice general presentation skills through an activity we call Speaking and Listening “Do’s and Don’ts.” We usually spread this lesson out among two class periods.
We first ask students to create a simple T-chart by folding a piece of paper in half and labeling one side “Do” and the other side “Don’t.” We then post Figure 30.2 “Speaking Do’s and Don’ts” on the document camera and display the first statement (the rest we cover with a blank sheet of paper).
We read the first statement, “Make eye contact with the audience,” and ask students if this is something they want to do when they are giving a presentation or if it is something they don’t want to do. Students write the statement where they think it belongs--under the “Do” column or “Don’t” Column. Students then share their answer with a partner and discuss why they put it in that column. After calling on a few pairs to share with the class, we move down the list repeating the same process of categorizing each statement as a “Do” or a “Don’t.” Students write it on their chart and discuss why it should be placed there.
After categorizing the statements for speaking, we give students Figure 30.3 “Listening Do’s and Don’ts .” We tell students to work in pairs to categorize the statements as something they do or something they don’t want to do when listening to a student presentation. This time, we ask students to make a quick poster with the headings “Do’s” and “Don’ts” for Listening. Under each heading students must list the corresponding statements--the teacher can circulate to check for accuracy. Students are asked to talk about why each statement belongs in each category and should be prepared to share their reasoning with the class. Students must also choose one “do” statement and one “don’t” statement to illustrate on their poster. Students can present their posters in small groups or with the whole class. This serves as a great opportunity to apply the speaking and listening “do’s” they just reviewed and heightens their awareness of the “don’ts!”

A fun twist, that also serves as a good review on a subsequent day, is to ask groups of students to pick two or three “do’s” and “don’ts” from both Speaking and Listening to act out in front of the class.
LESSON TWO Slide Presentations Concept Attainment
We periodically ask students to make slide presentations using PowerPoint or Google Slides to give them practice with developing visual aids (see the Home Culture activity later in this section). We show students how to make better slides, along with giving students the language support they may need in the form of an outline or sentence starters. An easy and effective way to do this is through Concept Attainment.
Concept Attainment involves the teacher identifying both "good" and "bad" examples of the intended learning objective. In this case, we use a PowerPoint containing three “good” slides and three “bad” ones (see them at The Best Resources For Teaching Students The Difference Between A Good and a Bad Slide ).
We start by showing students the first example of a “good” or “yes” slide (containing very little text and two images) and saying, “This is a yes.” However, we don’t explain why it is a “yes.” Then we show a “bad” or “no” example of a slide (containing multiple images randomly placed with a very “busy background”), saying, “This is a no” without explaining why. Students are then asked to think about them, and share with a partner why they think one is a "yes" and one is a "no."
At this point, we make a quick chart on a large sheet of paper (students can make individual charts on a piece of paper) and ask students to list the good and bad qualities they have observed so far. For example, under the “Good/Yes” column it might say “Has less words and the background is simple” and under the “Bad/No” column “Has too many pictures and the background is distracting.”
We then show the second “yes” example (containing one image with a short amount of text in a clear font) and the “no” example (containing way too much text and using a less clear font style). Students repeat the “think-pair-share” process and then the class again discusses what students are noticing about the “yes” and “no” examples. Then they add these observations to their chart.
Students repeat the whole process a final time with the third examples. The third “yes” example slide contains one image, minimal text and one bullet point. The third “no” example, on the other hand, contains multiple bullet points.
To reinforce this lesson at a later date, the teacher could show students more examples, or students could look for more “yes” and “no” examples online. They could continue to add more qualities of good and bad slides to their chart. See the Technology Connections section for links to good and bad PowerPoint examples, including the PowerPoint we use for this Concept Attainment lesson.
You can learn more about other presentations that support public speaking, such as home culture presentations, speed dating, talking points, top 5 and PechaKucha Book talks in our book, “ The ELL Teacher’s Toolbox: Hundreds of Practical Ideas to Support Your Students .”

Larry Ferlazzo has taught English Language Learners, mainstream and International Baccalaureate students at Luther Burbank High School in Sacramento for 15 years. He has authored eight books on education, hosts a popular blog for educators, and writes a weekly teacher advice column for Education Week Teacher . He was a community organizer for 19 years prior to becoming a high school teacher.

Katie Hull Sypnieski has worked with English Language Learners at the secondary level for over 20 years. She currently teaches middle school ELA and ELD at Rosa Parks K-8 School in Sacramento, California. She is a teaching consultant with the Area 3 Writing Project at the University of California, Davis and has leads professional development for teachers of ELLs. She is co-author (with Larry Ferlazzo) of The ESL/ELL Teacher’s Survival Guide and Navigating the Common Core with English Language Learners .

How to Prepare an Awesome Presentation in English
By Marie-Anne Duffeler
That was a great introduction your boss just delivered. The room is quiet and now it is your turn…your turn to speak.
Maybe you need to present your team, or deliver a sales pitch, or explain some figures and trends.
One thing is for sure: your presentation has to be awesome!
And your presentation will be in English, of course, as it is the language of communication at work.
Let’s think back: your English is quite decent – you can travel abroad and make yourself understood everywhere, you can communicate on the phone with your English-speaking colleagues and get the message across, and you write so many e-mails every day.
True, but these are familiar situations.
This time, however, you feel anxious because this presentation is formal and you have only one chance to get it right.
Speaking in public has always made you nervous, but speaking in public in English makes you twice as nervous.

What can you do? Simply follow this guide, which will help you step by step to prepare an awesome presentation in English.
First I will introduce the steps to create the oral presentation, and second we will look at some tips for the format and the style.
How to Prepare in 5 Steps
Remember that the stress before giving an oral presentation is normal, and even beneficial – it will give you the energy and motivation to prepare a good presentation, and preparation is key to delivering a memorable speech.
Good preparation will also give you confidence, which in turn will make speaking in front of your audience easier.
The five steps to follow to prepare a good presentation are simple to remember: they are the ‘ BASIS ‘ steps.
B = brainstorm A = audience S = slides I = ideas S = simulate
Let’s look at each step carefully.
1. Brainstorm
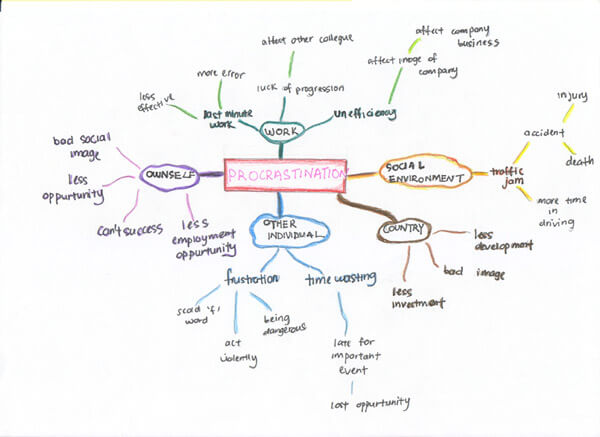
Brainstorming means putting on paper all the ideas that are connected to the topic of your presentation .
You can brainstorm alone or with colleagues. Ideally you should take a sheet of paper and write on it all the information you know and all the ideas you have about this topic.
A common way of brainstorming consists in writing the main topic inside a circle at the center of your page and then jotting down all around it the ideas and information connected to it. You can use arrows to indicate relationships.
2. Audience
Knowing who you will address is vital as it determines what information you need to select from your brainstorming session.
Keep only the information that is important to your audience .
If you add unnecessary information, you will end up losing your audience’s attention and your important message will get lost. So select carefully what to include in your presentation.

Another important reason to know your audience is the degree of formality that you need to use.
For example, how to address the listeners (“Ladies and Gentlemen” as opposed to “Hi everyone”), and whether or not to include humor (but I will come back to that later).
Make sure you find out who your audience will be before preparing your presentation.
Visual aids are key : they help you to remember what to say, and they help the audience to understand your presentation. However they need to be used wisely.
Most presentations will have slides, which can be designed with various software programs (e.g. PowerPoint, Open Office Impress or Prezi ).
Because your slides contain the information about your topic, you do not need to memorize your whole presentation, nor do you need to use clumsy paper notes, and so your hands can move freely during your speech.
When designing your slides remember:
- include an overview at the beginning of your slideshow
- present only the information that you have selected in step 2, only the information that is relevant to your audience
- present only one idea per slide
- write only keywords. Lengthy text will only detract your audience’s attention
- include numbers if necessary: long numbers are easier to grasp when they are written
- include as many pictures (or graphs) as possible – a picture is worth a thousand words.
Now that you have designed your slides, you need to accompany them with explanations. This step is the most difficult one if English is not your native language.

You need to prepare the explanation for each slide. In order to describe the idea in each slide, you need to use precise vocabulary combined with correct grammar – and to deliver both fluently.
So, sit back and look at each slide, then say out loud (or do it mentally if that is not possible) what you will say in front of your audience.
Describe each idea with your own words in the most natural fashion, as if you were explaining it to a friend or to a close colleague .
If you do not know some terms, look them up in a dictionary and write them down.
However, resist the urge to write a script for each slide. Written speeches generally get in the way of effective communication as the speaker ends up reading a script instead of talking to the audience. Only talented speakers can make written speeches sound natural.
Also, rely on what you already know in English. Now is not the appropriate time to venture into grammatical constructions that make you feel uncomfortable. There are many ways to express ideas, so use the words and grammar that you know well.
5. Simulate
The final step is simulating the actual presentation and it is essential to the success of your presentation.
This is what you need to do:
- First, record your presentation with a video camera
- Watch the recording and assess it with a self-assessment grid (I’ve included an example below)
- Film yourself a second time while making the corrections you identified from the self-assessment
- Assess your performance a second time.
You can use a simple video camera, your smart phone, a digital camera or even a webcam.
However, it is important when you film yourself that the camera focuses on the upper part of your body, so that you can assess your body language.
Also, make sure you are standing up. This is important for 3 reasons:
First, because this will likely be your position during the real presentation.
Second, it is a position that opens up your lungs and helps you to breathe better (which is very important to speaking loudly and clearly).
Finally, it allows you to move and to accompany your speech with gestures that emphasize the meaning of your words – and so improves your communication.
How to Self-Assess and Improve your Presentation
After you have filmed yourself, watch your presentation with a critical eye – give yourself both positive and negative criticism.
What did you do well? What do you need to improve?
They are many aspects to oral communication besides the words you say – your voice, body and eyes need to complement your speech.
To evaluate your performance you can use the following self-assessment grid:
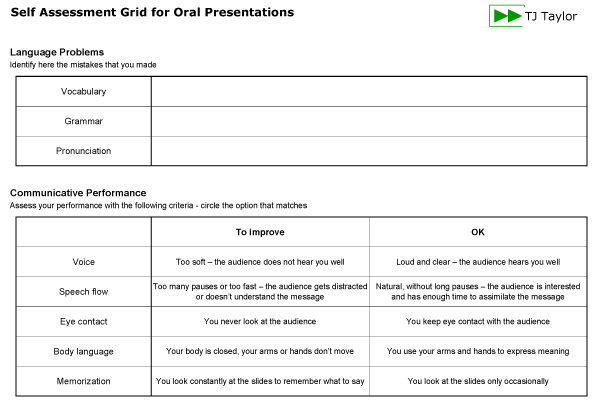
Download the Self-assessment Grid
Once you have used the self-assessment to identify your communication problems, you need to address them: correct the English mistakes, improve your voice or your body language, and film yourself a second time.
Then assess your performance again with the same self-assessment grid. If you are happy with the result, you are ready for the final show.
If not, you can continue to rehearse the presentation until you feel ready.
Depending on time constraints you can choose how many times you practise your speech before the actual performance, but remember that practicing it is not optional: if you want to deliver a good presentation, you have to practise it first .
A final consideration goes to the room where you will give your presentation: if possible, practise in that room, or at least get familiar with it (check where the switches for lights, screens, projectors, etc. are located).
Some Tips on Style and Format
Your oral presentation should have 3 parts : an introduction, a body, and a conclusion.
Your introduction presents the topic and gives an overview of the presentation, the body contains the information, facts or ideas, and the conclusion summarizes the ideas developed previously.
Repetition helps retention : if some information is important and needs to be remembered by your audience, be sure to repeat it. For example, mention it in the body and in the conclusion.
Tell your audience what you are going to tell them Tell them Then tell them what you have told them.
Keep it short and simple : remember that too much information will only result in your audience remembering nothing. Present only the important ideas in your slideshow, and repeat them in the conclusion.
Be credible : avoid spelling mistakes and mispronunciation. You might be a very talented professional but your presentation will be less convincing if it contains errors, spelling mistakes or mispronunciation of English terms.
You need to pay special attention to spelling and pronunciation in titles and keywords as well as in the introduction and the conclusion.
Use spellcheckers to check the spelling of your slides and online dictionaries to listen to the pronunciation of words (such as the Cambridge Dictionary ).
Avoid humor . Although humor can be helpful to defuse tense situations, it can also be dangerous and unpredictable. What makes you smile or laugh might be offensive to a foreigner.
Unless you know your audience well, refrain from using humor in professional presentations as it might lead to the opposite effect.
Engage the audience . When you deliver your speech, you need to establish a relationship between you and your audience.

A good way to do that is to keep eye contact and to ask questions. You can ask direct questions and the audience can answer verbally or physically (by raising their hands, for example) or you can opt for rhetorical questions, which are questions that do not require answers.
For example, you can introduce a slide with the following rhetorical question: “So how can we address this problem?” and then you give the solutions. Or start your conclusion with “What have we learnt so far?” and repeat the important ideas.
Asking questions is a good way to keep your audience attentive and to put rhythm into the presentation.
Ready, Set, Go!
You are now ready to stand up in front of your audience and deliver a memorable speech. Relax, take a deep breath, and just do it.

In conclusion, remember that the more oral presentations you make, the more confident you will be and the easier they will become.
View every opportunity to make a presentation as a challenge and as practice for your next big presentation!
Have you found this article interesting? Which tips will you try out? Tell us by adding your comments below – I look forward to reading your feedback.
Get the latest content first. No spam, ever. Unsubscribe at any time.
Who are TJ Taylor?
TJ Taylor is a language school that organises intensive courses in the UK and Ireland for professionals, and delivers corporate courses in Italy for over 100 companies. Founded in 2003 » Learn more
Module 5: Delivering Your Speech
Vocal aspects of delivery.
Though we speak frequently during the course of a day, a formal speech requires extra attention to detail in preparation of a more formal speech presentation. What can one do in advance to prepare for a speech? The challenge is partly determined by the speaker’s experience, background and sometimes cultural influence and existing habits of speaking. Articulation, Pronunciation, Dialect, Tone, Pitch, and Projection each depends on long-term practice for success. These aspects are like signatures, and should be developed and used by each speaker according to his own persona.
Voice, or vocal sound, is made when controlled air being exhaled from the lungs, passes over the vocal cords causing a controlled vibration. The vibrating air resonates in the body, chest cavity, mouth, and nasal passages. The vibrating air causes a chain reaction with the air in the room. The room’s air, set in motion by the voice, is captured by the listener’s ear. The vibration of the air against the eardrum is transferred to electrical impulses that are interpreted by the listener’s brain. Thus, the sounds we can make are predicated on the breaths that we take.

“Crying baby” by Brazzouk. CC-BY-SA .
Try This! Breathing
Talk without breathing. It cannot be done. So if you are screaming (like a baby), you are also breathing!
The first word of advice on speaking to an audience: BREATHE!
Articulation
We are often judged by how well we speak in general. A measure of perceived intellect or education is how well we articulate . That is: how well and correctly we form our vowels and consonants using our lips, jaw, tongue, and palate to form the sounds that are identified as speech. Diction and enunciation are other terms that refer to the same idea. For instance, saying “going to” instead of “gonna” or “did not” instead of “dint” are examples of good versus poor articulation. Consonant and vowels are spoken with standard accepted precision, and serious students and speakers will strive to practice the clarity of their sounds. Proper diction is as integral to the English language as proper spelling, but it takes practice.
Pronunciation
Proper articulation applied to a given word is that word’s pronunciation . The pronunciation includes how the vowels and consonants are produced as well as which syllable is emphasized. For generations, speakers depended on “markings (such as the International Phonetics Alphabet or similar Dictionary Symbols) to discover or decide how words were officially pronounced. With online dictionaries now readily available, one needs only to “look up” a word and select “play” to hear an audible recording of the official and precise way a word should be pronounced. Now there is no excuse for mispronouncing a word in a speech. A mispronounced word will obliterate a speaker’s credibility, and the audience’s attention will be focused on the fault rather than the message.
Try This! Pronunciation
1. Flip though a book, article or scholarly work until you come to a word that is unfamiliar and you can only guess its pronunciation.
2. Go to the Merriam-Webster Dictionary website, and look up the word.
3. When the definition appears, click the icon of the loudspeaker. The word is audibly pronounced for you.
The online dictionary is useful in both articulation as well as pronunciation.
Accent, Dialect, and Regionalisms

“Iraqi speaker” by Office of United States Rep. Ellen Tauscher. Public domain.
Subtleties in the way we pronounce words and phrase our speech within a given language are evident in accents , regionalisms , and dialects . An accent refers to the degree of prominence of the way syllables are spoken in words, as when someone from Australia says “undah” whereas we say “under.” A regionalism is a type of expression, as when someone says “The dog wants walked,” instead of “the dog wants to go for a walk.” Dialect is a variety of language where one is distinguished from others by grammar and vocabulary. In Pennsylvania you might hear people say that they are going to “red up the room,” which means “to clean the room.”
Those who depend on speaking for a career (broadcasters, politicians, and entertainers) will often strive for unaccented General or Standard English. Listen to most major network newscasters for examples of regionalism-free speech. A given audience may be prejudiced towards or against a speaker with an identifiable accent or dialect. Though we would wish prejudice were not the case, the way we speak implies so much about our education, cultural background, and economic status, that prejudice is inevitable. Any speaker should be aware of how accent, dialect , and regionalisms can be perceived by a given audience. If you speak in a way that the audience might find difficult to understand, make an extra effort to pay attention to the accent and phrasing of your speech. Ask a sympathetic and objective listener to help you when you practice.
We often refuse to accept an idea merely because the tone of voice in which it has been expressed is unsympathetic to us. – Friedrich Nietzsche
Vocal Quality
The quality of the voice, its timbre (distinctive sound) and texture, affects audibility and can affect the articulation. Our voices are unique to each of us. It is a result of our physical vocal instrument, including diaphragm, vocal cords, lungs and body mass. Some examples of vocal quality include warm, clear, soft, scratchy, mellow and breathy. Each speaker should practice at maximizing the vocal effect of his instrument, which can be developed with vocal exercises. There are numerous books, recordings and trainers available to develop one’s vocal quality when needed. The quality of one’s voice is related to its range of pitch.
Try This! Inflection
Your voice goes UP, and then your voice goes d o w n.
Pitch and Inflection
Identical to musical parlance, the pitch is the “highness” or “lowness” of the voice. Each of us has a range of tone . Vocal sounds are actually vibrations sent out from the vocal cords resonating through chambers in the body. The vibrations can literally be measured in terms of audio frequency in the same way music is measured. When the pitch is altered to convey a meaning (like raising the pitch at the end of a sentence that is a question), it is the inflection. Inflections are variations, turns and slides in pitch to achieve the meaning.
In his writing “Poetics,” Aristotle lists “Music” as an element of the Drama. Some scholars interpret that to include the musicalization of the spoken word with dramatic inflection . The meaning and effectiveness of a spoken line is greatly dependent on the “melody” of its inflection.
Though archaic, the study of elocution formalizes the conventions of inflection. In some contemporary cultures, inflection has been minimized because it sounds too “melodramatic” for the taste of the demographic group. It would be sensible to be aware of and avoid both extremes. With good animated inflection, a speaker is more interesting, and the inflection conveys energy and “aliveness” that compels the audience to listen.

“Ice-T” by Tino Jacobs. CC-BY .
When public speaking was known as elocution, sentences were “scored” like music, and spoken using formal rules. Sentences ending as a question went UP at the end. Sentences ending in a period, ended with a base note. And everyone had fun with exclamation points!
For most of music in history, including Opera, Broadway, and early Rock and Roll, songs were written so that the melody (raising and lowering the pitch) was consistent with what would be spoken. Many of today’s songs, notably Rap songs, depend solely on rhythm. There is little if any inflection (melody) to enhance a lyric’s meaning. Certain languages differ in their dependence on inflection. Japanese and German seem monotonic compared to Italian and French, which offer great variety of inflection.
The human voice is the most beautiful instrument of all, but it is the most difficult to play. – Richard Strauss
Even someone one who is not a singer can be expressive with inflection and pitch. Like the “Think System” of Professor Harold Hill in the musical The Music Man. If you THINK varied pitch, you can SPEAK varied pitch. Think of pitch inflections as seasoning spices that can make the speech more interesting. Sing “Happy Birthday.” You do not have to concentrate or analyze how to create the melody in your voice. Your memory and instinct take over. Notice how the pitch also provides an audible version of punctuation, letting the audience know if your sentence has ended, if it is a question, and so on. The melody lets the audience know that there is more to come (a comma) and when the phrase is ended (a period). Remember that in a speech, the audience does not have the written punctuation to follow, so you have to provide the punctuation with your inflection.
Try This! Vocal Variation
Find a listening partner. Using only the sounds of “la” ha,” and “oh,” covey the meaning of the following:1. It’s the biggest thing I’ve ever seen!
2. I’ve fallen and can’t get up!
3. I’ve got a crush on him/her.
4. That soup is disgusting and spoiled.
5. I got an “A” in my Speech Final!
If you cannot relay the meaning with just sounds, try a second time (each) with gestures and facial expressions until the listener understands. Then say the lines with the expressive inflections you have developed using only the sounds.
Those who do not use inflection, or use a range of pitch, are speaking in monotone. And, as the word implies, it can be monotonous, boring, and dull. A balance between melodramatic and monotonous would be preferred. The inflection should have a meaningful and interesting variety. Be careful not to turn a pattern of inflection into a repetitious sound. Think through each phrase and its musicalization separately.
Many speakers have developed the habit of ending each sentence as though it is a question. It may be becoming increasingly common. In the wake of the Valley Girl syndrome of the 1980’s, a bad inflection habit has entered the speech pattern: Some speakers end a declarative sentence with the inflection of a question.
Do you know what I mean?
A word of caution: Inflection and varied pitch must be “organic,” that is to say, natural for the speaker. You cannot fake it, or it sounds artificial and disingenuous. It is a skill that needs to develop over a period of time.
Rate of Speaking
| Table 12.1: Finding the Right Pace for Your Speech | ||
|---|---|---|
| If you speak too quickly… | If you speak too slowly… | |
| the audience might get the impression you have nothing important to say. | the audience might think you are too tired to be presenting. | |
| the audience has a difficult time catching up and comprehending what you are saying. They need time to digest the information. So plan on periodic pauses. | the audience can forget the first part of your sentence by the time you get to the last! (It happens!) And they lose interest. | |
| the audience might think you really do not want to be there. | the audience might think you are wasting their time by taking longer than necessary to relay your message. | |
| As a speaker, you cannot race with the audience, nor drag their attention down. Like Goldilocks, look for the pace that is “just right.” | ||
In order to retain clarity of the speech with articulation and inflection, the speaker must be aware that there is a range of appropriate tempo for speaking. If the tempo is too slow, the speech might resemble a monotonous peal. If it is too fast, the articulation could suffer if consonants or vowels are dropped or rushed to keep up the speed. An audience could become frustrated with either extreme. The tempo needs to be appropriate to the speaker’s style, but neither paced like a Gilbertian Lyric (as in “Gilbert and Sullivan”) patter nor a funereal dirge. A comfortable and clear pace is the best. An ideal speaking rate will allow you to comfortably increase your pace to create a sense of excitement, or slow down to emphasize the seriousness of a topic.
It is simple nonsense to speak of the fixed tempo of any particular vocal phrase. Each voice has its peculiarities. – Anton Seidl
Pauses Versus Vocalized Pauses
A text that is read has punctuation that the reader can see…miniature landmarks to define the text. When spoken, similar punctuation is needed for comprehension, and the speaker’s responsibility is to offer the text with pauses. Space between phrases, properly planted, gives the audience the opportunity to understand the structure of the speaker’s sentences and paragraphs. It also gives time for the audience to “digest” crucial phrases.
Generally, spoken sentences and paragraphs need to be simpler and shorter than what can be comprehended by reading. Pauses can help increase comprehension.
However, pauses that are filled with “uh’s, “um’s,” etc., are called vocalized pauses , or fillers , and should be avoided. They can be distracting and annoying, and give the impression of a lack of preparation if used excessively. Even worse is the use of vernacular phrases like, “y’know” (a contraction of “Do You Know”) which gives the impression of lack of education or lack of concern for the audience. The use of vocalized pauses may be the result of a habit that deserves an effort to be overcome. Avoid using phrases such as “Uh,” “OK?”, “y’know”, “like…, I mean,” “right?”
Vocal Projection
The volume produced by the vocal instrument is projection . Supporting the voice volume with good breathing and energy can be practiced, and helping a speaker develop the correct volume is a main task of a vocal trainer, teacher or coach. Good vocal support with good posture, breathing, and energy should be practiced regularly, long before a speech is delivered. There are numerous exercises devoted to developing projection capabilities.
While there is no need to shout, a speaker should project to be easily heard from the furthest part of the audience. Even if the speech is amplified with a microphone/sound system, one must speak with projection and energy. As with your rate of speech, you should speak at a volume that comfortably allows you to increase the volume of your voice without seeming to shout or decrease the volume of your voice and still be heard by all audience members.
Do not expect to walk up to the podium and have a full voice. Actors spend about a half-hour doing vocal warm-ups, and singers warm up much more. You might not have an opportunity to warm up immediately before your speech, but when you can, warm up with humming, yawning (loudly) or singing scales: all while breathing deeply and efficiently. It will loosen your voice, prevent irritation, and fire up your vocal energy.
Try This! Projection
Go to the room in which you are to speak. Have a friend sit as far away from the podium is possible. Rehearse your speech, talking loudly enough so your friend can hear you comfortably. That is the projection you will need. When you mentally focus on the distant listener, you will tend to project better.
One final note: If public speaking is or will be an important part of your career, it would be sensible to have an evaluation of your voice, articulation and projection done by an objective professional so you can take any remedial action that might be recommended. There are courses of study, private lessons, and professional voice coaches to work with your voice projection, tone, and pitch.
Words mean more than what is set down on paper. It takes the human voice to infuse them with deeper meaning. – Maya Angelou
- Chapter 12 Vocal Aspects of Delivery. Authored by : Victor Capecce, M.F.A.. Provided by : Millersville University, Millersville, PA. Located at : http://publicspeakingproject.org/psvirtualtext.html . Project : The Public Speaking Project. License : CC BY-NC-ND: Attribution-NonCommercial-NoDerivatives
- Crying baby. Authored by : Brazzouk. Provided by : MorgueFile. Located at : https://commons.wikimedia.org/wiki/File:Crying_baby.jpg . License : CC BY-SA: Attribution-ShareAlike
- Ice-T. Authored by : Tino Jacobs. Located at : http://commons.wikimedia.org/wiki/File:Ice-T_(2).jpg . License : CC BY: Attribution
- Iraqi Speaker. Authored by : Office of United States Rep. Ellen Tauscher (D - California). Located at : http://commons.wikimedia.org/wiki/File:Iraqi_Speaker.jpg . License : Public Domain: No Known Copyright

Privacy Policy
- Search entire site
- Search for a course
- Browse study areas
Analytics and Data Science
- Data Science and Innovation
- Postgraduate Research Courses
- Business Research Programs
- Undergraduate Business Programs
- Entrepreneurship
- MBA Programs
- Postgraduate Business Programs
Communication
- Animation Production
- Business Consulting and Technology Implementation
- Digital and Social Media
- Media Arts and Production
- Media Business
- Media Practice and Industry
- Music and Sound Design
- Social and Political Sciences
- Strategic Communication
- Writing and Publishing
- Postgraduate Communication Research Degrees
Design, Architecture and Building
- Architecture
- Built Environment
- DAB Research
- Public Policy and Governance
- Secondary Education
- Education (Learning and Leadership)
- Learning Design
- Postgraduate Education Research Degrees
- Primary Education
Engineering
- Civil and Environmental
- Computer Systems and Software
- Engineering Management
- Mechanical and Mechatronic
- Systems and Operations
- Telecommunications
- Postgraduate Engineering courses
- Undergraduate Engineering courses
- Sport and Exercise
- Palliative Care
- Public Health
- Nursing (Undergraduate)
- Nursing (Postgraduate)
- Health (Postgraduate)
- Research and Honours
- Health Services Management
- Child and Family Health
- Women's and Children's Health
Health (GEM)
- Coursework Degrees
- Clinical Psychology
- Genetic Counselling
- Good Manufacturing Practice
- Physiotherapy
- Speech Pathology
- Research Degrees
Information Technology
- Business Analysis and Information Systems
- Computer Science, Data Analytics/Mining
- Games, Graphics and Multimedia
- IT Management and Leadership
- Networking and Security
- Software Development and Programming
- Systems Design and Analysis
- Web and Cloud Computing
- Postgraduate IT courses
- Postgraduate IT online courses
- Undergraduate Information Technology courses
- International Studies
- Criminology
- International Relations
- Postgraduate International Studies Research Degrees
- Sustainability and Environment
- Practical Legal Training
- Commercial and Business Law
- Juris Doctor
- Legal Studies
- Master of Laws
- Intellectual Property
- Migration Law and Practice
- Overseas Qualified Lawyers
- Postgraduate Law Programs
- Postgraduate Law Research
- Undergraduate Law Programs
- Life Sciences
- Mathematical and Physical Sciences
- Postgraduate Science Programs
- Science Research Programs
- Undergraduate Science Programs
Transdisciplinary Innovation
- Creative Intelligence and Innovation
- Diploma in Innovation
- Postgraduate Research Degree
- Transdisciplinary Learning
Pronunciation tips for presenters
The following page intends to give some useful tips and advice on improving pronunciation whilst giving a presentation.
Get physical
Do some 'warm-ups' to loosen up your mouth and your tongue before you speak. You can move and stretch your mouth, and repeat tricky words and sounds. Doing this will help you to overcome the physical 'stiffness' of the articulators and to speak fluently and clearly.
Get interested in your talk
Focus on your content. Make it sound interesting for your audience. If you speak with meaning and commitment along with healthy interest in the subject you tend to lose your nerves. Ensure you speak clearly so that your pronunciation can be understood.
Give your audience time to take in what you are saying
You can do this by pausing at the end of word groups and information chunks. Pausing is important for listeners because they need time to absorb information. Pausing also gives you, the presenter, time and 'space' to stress key words and use intonation signals.
However, too much pausing, or stopping at the end of each word or couple of words can make you sound hesitant and lacking in confidence. It also makes every word carry too much meaning. English speakers get meaning across by stressing key words in information chunks.
Give your audience a sense of direction
Don't lose your audience in a forest of words. A presentation is like a journey. Take your audience with you.
Signal new topics and sections of your presentation by:
- starting on a higher pitch level
- stressing the key words, such as signposting words and the topic word/s of your new section.
Let your audience know when information is finished or not by using:
Let your audience know what's important
Present key information clearly by stressing key words. In English, new information usually occurs towards the end of a speech chunk or sentence. Keep your voice and breath going so you can stress those crucial words at the end.
Get the basics right
Make sure that you can pronounce key vocabulary, data, names and acronyms clearly and accurately. To do this:
- check the stress in poly-syllabic (multi-syllabic) words - use a dictionary or ask a competent English speaker if you are not sure. Practice difficult vocabulary.
- identify and practise problem sounds, particularly in high frequency words and key words in your presentation.
Rehearse and reflect
Nothing beats practice. It is amazing what mistakes you will find in a presentation as soon as you start rehearsing.
Practise your presentation standing up and imagine yourself in the situation.
- Your opening. When you start your presentation, you're likely to feel nervous so it is helpful to know exactly what you are going to say to open your presentation, where you will pause, what words you will stress, and the intonation you will use.
- Topic sentences and signposting words introducing new topics or making a topic change. This will make your transitions smooth and easy to follow.
- Your conclusion and the body of your presentation, including how you will refer to any visual aids as you move through your presentation.
Video or record yourself if possible and use a checklist to evaluate how effective you are. Better still, get someone to listen to you and give you some feedback.
Back to top
UTS acknowledges the Gadigal people of the Eora Nation, the Boorooberongal people of the Dharug Nation, the Bidiagal people and the Gamaygal people, upon whose ancestral lands our university stands. We would also like to pay respect to the Elders both past and present, acknowledging them as the traditional custodians of knowledge for these lands.

- Cambridge Dictionary +Plus
English pronunciation of presentation
Presentation.
Your browser doesn't support HTML5 audio
(English pronunciations of presentation from the Cambridge Advanced Learner's Dictionary & Thesaurus and from the Cambridge Academic Content Dictionary , both sources © Cambridge University Press)

Word of the Day
to shape something so that it can move as effectively and quickly as possible through a liquid or gas

Fakes and forgeries (Things that are not what they seem to be)

Learn more with +Plus
- Recent and Recommended {{#preferredDictionaries}} {{name}} {{/preferredDictionaries}}
- Definitions Clear explanations of natural written and spoken English English Learner’s Dictionary Essential British English Essential American English
- Grammar and thesaurus Usage explanations of natural written and spoken English Grammar Thesaurus
- Pronunciation British and American pronunciations with audio English Pronunciation
- English–Chinese (Simplified) Chinese (Simplified)–English
- English–Chinese (Traditional) Chinese (Traditional)–English
- English–Dutch Dutch–English
- English–French French–English
- English–German German–English
- English–Indonesian Indonesian–English
- English–Italian Italian–English
- English–Japanese Japanese–English
- English–Norwegian Norwegian–English
- English–Polish Polish–English
- English–Portuguese Portuguese–English
- English–Spanish Spanish–English
- English–Swedish Swedish–English
- Dictionary +Plus Word Lists
- All translations
To add ${headword} to a word list please sign up or log in.
Add ${headword} to one of your lists below, or create a new one.
{{message}}
Something went wrong.
There was a problem sending your report.
Business English
- How to answer the telephone in English
- How to give a company tour in English
- Giving presentations
- Pronunciation of technical vocabulary
- Planning your presentation
- Presentation software
- Phrases for presentations
- English grammar and vocabulary
- English grammar exercises
- B2 level materials
- Listen and read - stories
- Listen and read - grammar
- Practising English podcasts
- Games and activities
Presentations in English: part 1
Using the correct technical language and adequate pronunciation..
Most professionals already know the technical language or English "jargon" relevant to their particular field. However, many may be more proficient with reading technical terminology and could be less familiar with pronouncing the expressions correctly in English.
If the words you need to know are long or complicated to pronounce, you could practise them before giving the presentation in English. One way to do this is by using a free Text to Speech programme (TTS). Insert sentences into the box which include the technical words and select a British English or American English voice if possible. Do not worry that the voice is machine generated. For pronunciation practice of individual words the quality is usually adequate.* You can often download an Mp3 version to keep.
Practice makes perfect
If you want your audience to understand you correctly during the presentation, practise your own pronunciation of technical vocabulary by copying the TTS extracts as mp3 files and then recording and listening to your own voice and comparing the two versions. Keep the TTS extracts on your laptop or mobile device for listening practice on the day the presentation starts, if possible.
Part 2 - Planning your presentation
*Machine generated TTS software often has problems with intonation (rising or falling voice in questions or stressing particular words etc.) but if you use it only for practice of how to pronounce particular words in English, you should find it very useful.
Copyright © 2023 Practising English All rights reserved
This page has been archived and is no longer updated
Effective Oral Presentations
Verbally (and as a general rule), do not write down and memorize or read your full text, because then your presentation will sound like what it is: a recited written text. Instead, memorize the outline of your presentation — that is, a tree structure of main points and subpoints — and speak ex tempore, reinventing the words as you go along. As you do, you will occasionally need to think about what to say next and find the most appropriate words to say it. Instead of using filler words ( um , er , you know , I mean , etc.), simply pause. If you say um , you get about half a second of thinking time and the audience is likely to notice the um and be irritated by it. If you keep silent, you can get up to two or three seconds of thinking time without the audience noticing anything. Even if attendees do notice the silence, they will simply think that you are choosing your words carefully — and there is nothing wrong with that.
Despite pointing often at the screen, Marie nicely faces the audience with her body at all times, keeps her hands down between gestures, and maintains eye contact with the attendees. Transcript Vocally, vary the tone, rate, and volume of your voice as a function of the meaning, complexity, and importance of what you are saying. You need not invent a new intonation pattern: You simply need to amplify your normal pattern.
Visually, control your body. Adopt a stable, confident position; move only when you have a positive reason to do so (for example, move closer to the audience for taking questions), not when your body seems to ask for it. When you make a gesture, make it large and deliberate; between gestures, bring your hands down and do not fidget. Establish eye contact: Engage the audience by looking them straight in the eyes.
At all times, make sure you address the audience. Even if you have slides, tell the audience your story in a stand-alone way; do not just explain your slides. In particular, anticipate your slides. You should know at all times what your next slide is about so you can insert an appropriate transition.
Delivering as a non-native speaker
To keep the audience engaged , Jean-luc emphasizes his points with facial expressions, purposeful gestures, and — especially — a high dynamic range in his vocal delivery. Transcript If you are a non-native speaker of English, you may find it more challenging to speak ex tempore in English than in your native language. Still, even imperfect extemporaneous English is more likely to engage the audience than reciting a more polished, less spontaneous written text. To improve your delivery and overall presentation as a non-native speaker, practice more, pace yourself, and support your spoken discourse with appropriate slides.
While all speakers benefit from practicing their presentations multiple times, consider investing more time in such practice if you are less familiar with the language. Practicing helps you identify missing vocabulary, including key technical terms (which are difficult to circumvent), and express your ideas more fluently. As you practice, you may want to prepare a list of difficult words (to review on the day of your presentation) or write down an occasional complex yet crucial sentence. Still, do not feel bound to what you write down. These notes should be a help, not a constraint.
Practicing in front of an audience (a few colleagues, for example) can help you correct or refine your pronunciation. If you are unsure how to pronounce some words or phrases, you can ask native speakers in advance or check online dictionaries that offer phonetic spelling or audio rendering. Still, you may be unaware of certain words you mispronounce; a practice audience can point these words out to you if you invite it to do so.
During your presentation, pace yourself. As a non-native speaker, you may feel you need to search for your words more often or for a longer time than in your native language, but the mechanism is the same. Do not let this challenge pressure you. Give yourself the time you need to express your ideas clearly. Silence is not your enemy; it is your friend.
Pacing yourself also means speaking more slowly than you otherwise might, especially if you have an accent in English. Accents are common among non-native speakers — and among specific groups of native speakers, too — and they are not a problem as long as they are mild. Often, they are experienced as charming. Still, they take some getting used to. Remember to slow down, especially at the beginning of a presentation, so your audience can get used to your accent, whether native or not.
Handling stage fright and mishaps
Most speakers, even experienced ones, are nervous before or during an oral presentation. Such stage fright is normal and even reassuring: It shows that you care, and you should care if you want to deliver an effective presentation. Accordingly, accept your stage fright rather than feeling guilty about it. Instead of trying to suppress nervousness, strive to focus your nervous energy in your voice, your gestures, and your eye contact. Do not let it dissipate into entropy, such as by using filler words or engaging in nervous mannerisms.
Among the many ways to keep your nerves under control, perhaps the most effective one is to focus constructively on your purpose at all times. Before your presentation, eliminate all the unknowns: Prepare your presentation well, identify (or even meet) your audience, and know the room. During the presentation, do what it takes to get your message across, even if it means doing something differently than you had planned. Have a positive attitude about the presentation at all times: Visualize what you want to achieve, not what you want to avoid.
Even with careful preparation, mishaps can occur. For example, technology may fail, you may forget what you wanted to say, or you may accidentally say the wrong thing. As a rule, do not apologize for what happens — neither in advance nor after the fact. Although well-meant, such apologies provide no benefit to the audience: They are noise. If you can do something about the problem, such as fix the technology or insert what you forgot later in the presentation, concentrate on doing so instead of apologizing. If the problem is out of your control, then there is no need to apologize for it. As a specific example, if you feel your command of English is poor, then do what you can in advance to improve it; in particular, practice your presentation thoroughly. Then, on the day of the presentation, do your best with the command you have, but do not apologize at the beginning of the presentation for what you think is poor English. This apology will not solve anything, and it gives the attendees a negative image of you. Rather, let the attendees judge for themselves whether your command of English is sufficient (perhaps it is, despite what you might think). In other words, focus on delivering results, not excuses.
This page appears in the following eBook
Topic rooms within Scientific Communication

Within this Subject (22)
- Communicating as a Scientist (3)
- Papers (4)
- Correspondence (5)
- Presentations (4)
- Conferences (3)
- Classrooms (3)
Other Topic Rooms
- Gene Inheritance and Transmission
- Gene Expression and Regulation
- Nucleic Acid Structure and Function
- Chromosomes and Cytogenetics
- Evolutionary Genetics
- Population and Quantitative Genetics
- Genes and Disease
- Genetics and Society
- Cell Origins and Metabolism
- Proteins and Gene Expression
- Subcellular Compartments
- Cell Communication
- Cell Cycle and Cell Division
© 2014 Nature Education
- Press Room |
- Terms of Use |
- Privacy Notice |

Visual Browse
- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution

Search form
- Advanced search
- Search responses
- Search blogs
- How to prepare and...
How to prepare and deliver an effective oral presentation
- Related content
- Peer review
- Lucia Hartigan , registrar 1 ,
- Fionnuala Mone , fellow in maternal fetal medicine 1 ,
- Mary Higgins , consultant obstetrician 2
- 1 National Maternity Hospital, Dublin, Ireland
- 2 National Maternity Hospital, Dublin; Obstetrics and Gynaecology, Medicine and Medical Sciences, University College Dublin
- luciahartigan{at}hotmail.com
The success of an oral presentation lies in the speaker’s ability to transmit information to the audience. Lucia Hartigan and colleagues describe what they have learnt about delivering an effective scientific oral presentation from their own experiences, and their mistakes
The objective of an oral presentation is to portray large amounts of often complex information in a clear, bite sized fashion. Although some of the success lies in the content, the rest lies in the speaker’s skills in transmitting the information to the audience. 1
Preparation
It is important to be as well prepared as possible. Look at the venue in person, and find out the time allowed for your presentation and for questions, and the size of the audience and their backgrounds, which will allow the presentation to be pitched at the appropriate level.
See what the ambience and temperature are like and check that the format of your presentation is compatible with the available computer. This is particularly important when embedding videos. Before you begin, look at the video on stand-by and make sure the lights are dimmed and the speakers are functioning.
For visual aids, Microsoft PowerPoint or Apple Mac Keynote programmes are usual, although Prezi is increasing in popularity. Save the presentation on a USB stick, with email or cloud storage backup to avoid last minute disasters.
When preparing the presentation, start with an opening slide containing the title of the study, your name, and the date. Begin by addressing and thanking the audience and the organisation that has invited you to speak. Typically, the format includes background, study aims, methodology, results, strengths and weaknesses of the study, and conclusions.
If the study takes a lecturing format, consider including “any questions?” on a slide before you conclude, which will allow the audience to remember the take home messages. Ideally, the audience should remember three of the main points from the presentation. 2
Have a maximum of four short points per slide. If you can display something as a diagram, video, or a graph, use this instead of text and talk around it.
Animation is available in both Microsoft PowerPoint and the Apple Mac Keynote programme, and its use in presentations has been demonstrated to assist in the retention and recall of facts. 3 Do not overuse it, though, as it could make you appear unprofessional. If you show a video or diagram don’t just sit back—use a laser pointer to explain what is happening.
Rehearse your presentation in front of at least one person. Request feedback and amend accordingly. If possible, practise in the venue itself so things will not be unfamiliar on the day. If you appear comfortable, the audience will feel comfortable. Ask colleagues and seniors what questions they would ask and prepare responses to these questions.
It is important to dress appropriately, stand up straight, and project your voice towards the back of the room. Practise using a microphone, or any other presentation aids, in advance. If you don’t have your own presenting style, think of the style of inspirational scientific speakers you have seen and imitate it.
Try to present slides at the rate of around one slide a minute. If you talk too much, you will lose your audience’s attention. The slides or videos should be an adjunct to your presentation, so do not hide behind them, and be proud of the work you are presenting. You should avoid reading the wording on the slides, but instead talk around the content on them.
Maintain eye contact with the audience and remember to smile and pause after each comment, giving your nerves time to settle. Speak slowly and concisely, highlighting key points.
Do not assume that the audience is completely familiar with the topic you are passionate about, but don’t patronise them either. Use every presentation as an opportunity to teach, even your seniors. The information you are presenting may be new to them, but it is always important to know your audience’s background. You can then ensure you do not patronise world experts.
To maintain the audience’s attention, vary the tone and inflection of your voice. If appropriate, use humour, though you should run any comments or jokes past others beforehand and make sure they are culturally appropriate. Check every now and again that the audience is following and offer them the opportunity to ask questions.
Finishing up is the most important part, as this is when you send your take home message with the audience. Slow down, even though time is important at this stage. Conclude with the three key points from the study and leave the slide up for a further few seconds. Do not ramble on. Give the audience a chance to digest the presentation. Conclude by acknowledging those who assisted you in the study, and thank the audience and organisation. If you are presenting in North America, it is usual practice to conclude with an image of the team. If you wish to show references, insert a text box on the appropriate slide with the primary author, year, and paper, although this is not always required.
Answering questions can often feel like the most daunting part, but don’t look upon this as negative. Assume that the audience has listened and is interested in your research. Listen carefully, and if you are unsure about what someone is saying, ask for the question to be rephrased. Thank the audience member for asking the question and keep responses brief and concise. If you are unsure of the answer you can say that the questioner has raised an interesting point that you will have to investigate further. Have someone in the audience who will write down the questions for you, and remember that this is effectively free peer review.
Be proud of your achievements and try to do justice to the work that you and the rest of your group have done. You deserve to be up on that stage, so show off what you have achieved.
Competing interests: We have read and understood the BMJ Group policy on declaration of interests and declare the following interests: None.
- ↵ Rovira A, Auger C, Naidich TP. How to prepare an oral presentation and a conference. Radiologica 2013 ; 55 (suppl 1): 2 -7S. OpenUrl
- ↵ Bourne PE. Ten simple rules for making good oral presentations. PLos Comput Biol 2007 ; 3 : e77 . OpenUrl PubMed
- ↵ Naqvi SH, Mobasher F, Afzal MA, Umair M, Kohli AN, Bukhari MH. Effectiveness of teaching methods in a medical institute: perceptions of medical students to teaching aids. J Pak Med Assoc 2013 ; 63 : 859 -64. OpenUrl
how to pronounce presentation
/ˌpɹɛzənˈtɛɪʃən/.
audio example by a male speaker
audio example by a female speaker
the above transcription of presentation is a detailed (narrow) transcription according to the rules of the International Phonetic Association; you can find a description of each symbol by clicking the phoneme buttons in the secction below.
presentation is pronounced in four syllables
press buttons with phonetic symbols to learn how to precisely pronounce each sound of presentation
example pitch curve for pronunciation of presentation
Test your pronunciation of presentation.
press the "test" button to check how closely you can replicate the pitch of a native speaker in your pronunciation of presentation
video examples of presentation pronunciation
An example use of presentation in a speech by a native speaker of american english:
“… member said during the presentation we …”
meaning of presentation
Presentation is the act of formally sharing information about something to someone.
presentation frequency in english - B2 level of CEFR
the word presentation occurs in english on average 24.1 times per one million words; this frequency warrants it to be in the study list for B2 level of language mastery according to CEFR, the Common European Framework of Reference.
topics presentation can be related to
it is hard to perfectly classify words into specific topics since each word can have many context of its use, but our machine-learning models believe that presentation can be often used in the following areas:
1) communication, information, and media;
2) people, society, and culture;
words with pronunciation similar to presentation
Words that rhyme with presentation, did this page help you.
How to pronounce "confidence" in American English
Practice the pronunciation of any english word using elsa’s advanced technology., type the word you’d like to practice.
Can you pronounce them?
Step 1 Listen to the word
Step 2 let's hear how you pronounce "oral presentation".
Subscribe to Pro Access
Anybody can speak English with confidence

Join millions of users speaking English more confidently with ELSA

For Individuals
ELSA Subscriptions
For Organizations
English for Schools
English for Companies
Creators Program
Customer Help
Activate Voucher Code
Redeem Promo Code
Current students
- Staff intranet
- Find an event
Oral presentations
- Prepare your presentation
- Structure your presentation
- Audiovisual aids
Speaking tips
A large part of the impact of your presentation is made by your voice and body language, yet people generally focus their preparation on content. Make sure you allocate some time to considering and practising your speaking skills and body language.
Good voice control and clear articulation are essential for an effective oral presentation. You should also think about varying your pitch, volume, pace, expression and intonation to keep the audience engaged.
Your voice can be constricted by tightness in the rest of your body. Try to relax if you notice tension in your body, such as tightness, clenching or locking.
Deep breathing exercises are useful to allow you to relax and develop maximum lung capacity.
If you’re from a language background other than English, making your meaning clear to the audience is more important than correct grammar and pronunciation when you speak. To help clarify your meaning you can:
- repeat your points using different words
- use written text for important keywords, for example on PowerPoint slides
- use visual support such as pictures, tables and diagrams
- use hand gestures
- make a list of the most important technical terms for your presentation, and practise their pronunciation beforehand
- speak more slowly than you would in your first language.
English speech practice course
You can download audio lessons as part of our English speech practice course.
- English speech practice guide (pdf, 6.8MB)
- Lesson 1: stress and rhythm (mp3, 17MB)
- Lesson 2: weak forms (mp3, 9.2MB)
- Lesson 3: contractions (mp3, 6.5MB)
- Lesson 4: link up (mp3, 19.6MB)
- Lesson 5: intonation (mp3, 17.9MB)
- Lesson 6: practice of all features (mp3, 14MB)
- Lesson 7: consonant sounds - fricatives (mp3, 7.2MB)
- Lesson 8: consonant sounds - l, r and n (mp3, 7.1MB)
- Lesson 9: long versus short vowels (mp3, 7.3MB)
- Lesson 10: 'or' and 'er' (mp3, 6.7MB)
- Lesson 11: the 'ugh' sound (mp3, 7.5MB)
Body language
Your body language is important to engage an audience. If you look confident and comfortable while presenting, you will put the audience at ease.
Think about your stance and posture, hand and body movement, and your facial expressions. Try to make eye contact with different people.
When practising you could try recording yourself. You may notice distractions such as frequently saying ‘um’, annoying body or hand movements, speaking too quickly or obscuring visuals.
Managing stress and nerves
It’s normal to feel nervous or anxious before and during giving a presentation in front of an audience.
Stress hormones (such as adrenaline in the short term, or cortisol in the long term) can be useful as they make you feel alert and energetic. They can also have negative effects too, which make it difficult to deliver a presentation.
To reduce effects such as shaking, sweating and difficulty concentrating, you can try the following strategies.
- Avoid stimulants such as coffee or energy drinks.
- Do some vigorous physical exercise a day or two before your presentation. This can lower your levels of stress hormones.
- Use positive self talk (for example, “I’ve done the preparation and I know the topic – it will be okay”).
- Visualise the presentation being successful.
- Think about your strengths as a presenter.
- Focus on the friendly faces in the audience.
- Take some water with you in case your mouth gets dry. It also gives you an excuse to take a little break for a sip of water.
- Wear clothes that you feel comfortable in and that are appropriate for the situation.
- Practise your talk beforehand with a friend.
- Make your notes very simple and easy to read, with keywords instead of sentences.
- Attend a practice workshop on presentations .
- Join a public speaking club, such as Toastmasters .
- If you’ll be presenting in a new place, go there beforehand to become familiar with it.
Find more information on overcoming stress and anxiety .
This material was developed by the Learning Hub (Academic Language and Learning), which offers workshops, face-to-face consultations and resources to support your learning. Find out more about how they can help you develop your communication, research and study skills .
See the handout on Oral presentations (pdf, 3.2MB) .
Related links
- Learning Hub (Academic Language and Learning)
- Learning Hub (Academic Language and Learning) workshops
- Group work and participating in class
- Preparing for exams
- Website feedback
Your feedback has been sent.
Sorry there was a problem sending your feedback. Please try again
You should only use this form to send feedback about the content on this webpage – we will not respond to other enquiries made through this form. If you have an enquiry or need help with something else such as your enrolment, course etc you can contact the Student Centre.
- Find an expert
- Media contacts
Student links
- How to log in to University systems
- Class timetables
- Our rankings
- Faculties and schools
- Research centres
- Campus locations
- Find a staff member
- Careers at Sydney
- Emergencies and personal safety

- Accessibility
You are using an outdated browser. Please upgrade your browser or activate Google Chrome Frame to improve your experience.

How to Prepare for Your Oral Presentation in English
When you stand up for an oral presentation, you want to feel like a rockstar .
Confident. Cool. Ready to blow the audience away.
That is the ideal situation, anyways.
In real life, most people—even native English speakers—feel totally the opposite before an oral presentation.
Nervous. Self-conscious. Scared the audience will fall asleep.
Most of us have been there. Every student and professional, at some point, will have to do an oral presentation . Of course that includes English language learners. In fact, oral presentations might happen more often in an English class because they are a good way for teachers to assess your speaking and writing skills.
This article will provide a six-step example of how to ace your oral presentation in English . We will provide key English phrases, tips and practice techniques you can use for any presentation you have coming up.
Soon you will be presenting in English with the confidence of a rockstar !
Download: This blog post is available as a convenient and portable PDF that you can take anywhere. Click here to get a copy. (Download)
- Thousands of learner friendly videos (especially beginners)
- Handpicked, organized, and annotated by FluentU's experts
- Integrated into courses for beginners

Follow This Example to Rock Your Oral Presentation in English
Every country has different cultural standards for communication. However, there is a general consensus in English-speaking colleges and universities about what makes a good oral presentation.
Below, we will show you how to write a presentation in English that your listeners will love. Then we will show you the English speaking skills and body language you need to present it effectively.
1. Introducing a Presentation in English
Having a strong introduction is extremely important because it sets the tone for the rest of the presentation . If the audience is not interested in your presentation right away, they probably will not pay attention to the rest of it.
To get everyone’s interest, try using attention-grabbing language . If your introduction is engrossing enough, the audience will not care if you have an accent or mispronounce a few words. They will want to learn more about your topic because you did such a great job of making them interested.
Here are some example ideas and phrases you can use in your own presentation introductions:
- Start with a story or personal anecdote , so the audience will be able to relate to your presentation.
“When I was a child…”
- Interactive subtitles: click any word to see detailed examples and explanations
- Slow down or loop the tricky parts
- Show or hide subtitles
- Review words with our powerful learning engine
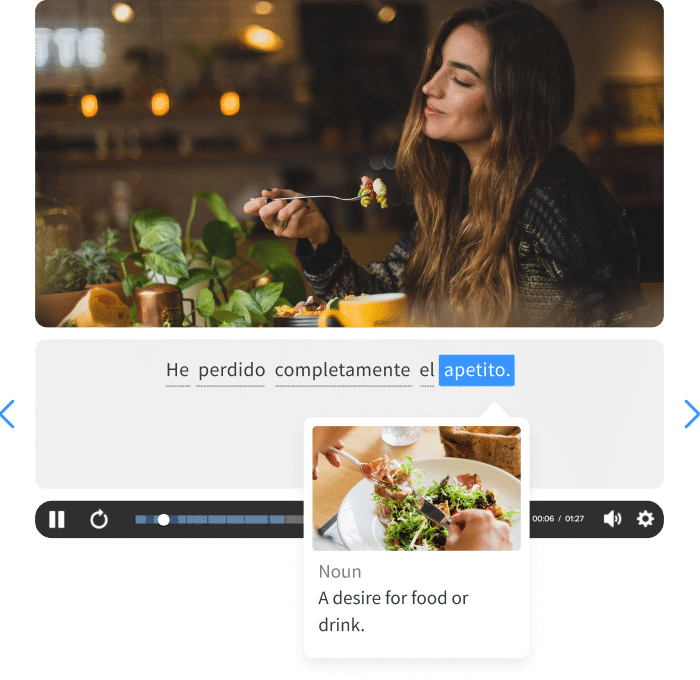
- Mention a startling fact or statistic.
“Did you know the U.S. is the only country that…”
- Have the audience imagine something or describe a vivid scene to them.
“Imagine you are sitting on the beach…”
- Show an interesting picture or video on your presentation screen.
- Introducing yourself can also help make the audience more comfortable. It does not have to be anything fancy.
“My name is John and I am…”
“I became interested in this topic because…”
2. Supporting Your Claims with Evidence
If you have written an essay in English , you have probably had to do some research to provide statistics and other facts to support your thesis (the main point or argument of your essay). Just like those essays, many oral presentations will require you to persuade someone or inform them about a topic.
Your presentation will need background information and evidence . To persuade someone, you will need convincing evidence. No one will be persuaded if you simply say, “We need to stop global warming because it is bad.”
At the same time, it may be hard to express your thoughts or argument if English is not your first language. That is why doing research and finding credible sources is extra important.
- Learn words in the context of sentences
- Swipe left or right to see more examples from other videos
- Go beyond just a superficial understanding

Using information and quoting from sources can make your presentation much stronger. (Of course, always remember to cite your research properly so you do not plagiarize !) If you are not sure how to go about researching or where to look for evidence, the University of North Carolina’s Writing Center provides some excellent examples here .
After you have done research, add a section or a slide that specifically gives facts or evidence for your topic . This should be somewhere in the middle of the presentation, after your introduction but before your conclusion or closing thoughts (basically like the body paragraphs in an essay). This will help keep your ideas logical and make it a really effective presentation.
3. Incorporating Persuasive Language
Specific evidence is crucial for a persuasive argument. But to truly impact your audience, you need to speak persuasively, too .
Need some vocabulary that will catch everyone’s attention? According to Buffer , the five most persuasive words in the English language are surprisingly simple:
- Free (this one is less relevant to oral presentations, since it is used in the context of persuading people to get a product)
Using these words in your introduction and throughout your presentation will help keep the audience engaged.
For example, if giving a persuasive speech, speaking directly to the audience will have a better effect:
“To help lessen the effects of global warming, the planet needs you .”
- FluentU builds you up, so you can build sentences on your own
- Start with multiple-choice questions and advance through sentence building to producing your own output
- Go from understanding to speaking in a natural progression.
4. Using Logical Flow and Transitions
As an English learner, was there ever a conversation that you could not follow because you had no idea what was going on? A language barrier often causes this confusion. However, even if your English is fluent, this can also happen when ideas or information are presented in an order that does not make sense.
This applies to presentations as well. If the sequence is illogical, the audience may become confused. It is important to have a clear sequence of thoughts or events. A distinct beginning, middle and end with logical sequences is needed for your audience to follow along.
As an English language learner, you may not be familiar with certain transitional words or phrases. Below are some example English words and phrases to use as you transition through your oral presentation.
General transitions that show sequence:
- First…
- Next…
- Then…
- In addition/additionally…
When you are nearing the end of your presentation, it is important to let the audience know you are going to finish soon. Abruptly ending the presentation may confuse the audience. Or, the presentation may not seem as effective. Just like with introductions and transitions, there are certain phrases that you can use to bring your presentation to a close.
Phrases to conclude your presentation:
- Images, examples, video examples, and tips
- Covering all the tricky edge cases, eg.: phrases, idioms, collocations, and separable verbs
- No reliance on volunteers or open source dictionaries
- 100,000+ hours spent by FluentU's team to create and maintain
- To conclude/In conclusion…
- To sum everything up…
- Finally…
5. Speaking Clearly and Confidently
You may be self-conscious about your ability to speak clearly if you are not fluent in English or if you have an accent. But let us be honest. Many people do not have long attention spans (the length of time someone can focus on one thing), so you will need to keep their attention during your presentation. And to do this, you will have to enunciate (speak clearly, loudly and confidently).
Do not expect this to just happen on the day of your presentation. You will need to practice ahead of time . Here is how:
Pay attention to how your lips, mouth and tongue move.
Practice saying different sounds and words over and over in front of the mirror, or have a friend watch you. What shapes does your mouth make? When does your tongue raise or flick? How can you change those movements to make each word sound clearer?
Listen to others speak English so you know how it should sound.
FluentU takes authentic videos—like music videos, movie trailers, news and inspiring talks—and turns them into personalized language learning lessons.
You can try FluentU for free for 2 weeks. Check out the website or download the iOS app or Android app.
P.S. Click here to take advantage of our current sale! (Expires at the end of this month.)
Try FluentU for FREE!
Record yourself when you practice your presentation.
This will help you get a better sense of how your mouth moves or how you pronounce words. You will also see what kind of mistakes you made and will be able to correct them.
Practice speaking slowly.
Along with enunciation, it is important to practice speaking slowly . Nerves can make us rush through things, but the audience may not understand you if you speak too quickly. Try reading your presentation for a couple minutes a day to get used to speaking slowing.

6. Making Eye Contact
In American society, it is important to keep eye contact. It is considered rude to not look someone in the eyes when you are speaking with them. Avoiding eye contact (even if it is unintentional or out of embarrassment) might frustrate your audience.
Therefore, when giving your oral presentation, you will want to try to make eye contact with your audience, especially if you are in the U.S. The audience will not feel appreciated if you stare down at your note cards or at the presentation screen. They may become bored. Or, they may think you are not confident in your work—and if you are not confident, they will not be, either!
Here is an example of a speaker demonstrating eye contact during an English presentation . Notice how he is careful to make eye contact with all audience members, looking left, right and forward throughout the presentation.
Following the tips in this article will help make your oral presentation great. Who knows, maybe your teacher or professor will use it as an example for other students!
As an added bonus, all of the skills needed for a good oral presentation are needed in everyday English. Speaking clearly, making eye contact and having a logical flow of ideas will help you communicate better with others when you are speaking with them in English. In addition, knowing how to write an introduction, use attention-grabbing language and provide evidence will help you in English classes. You will be able to get a great grade on your presentation and improve your overall communication skills.
And One More Thing...
If you like learning English through movies and online media, you should also check out FluentU. FluentU lets you learn English from popular talk shows, catchy music videos and funny commercials , as you can see here:
If you want to watch it, the FluentU app has probably got it.
The FluentU app and website makes it really easy to watch English videos. There are captions that are interactive. That means you can tap on any word to see an image, definition, and useful examples.
FluentU lets you learn engaging content with world famous celebrities.
For example, when you tap on the word "searching," you see this:
FluentU lets you tap to look up any word.
Learn all the vocabulary in any video with quizzes. Swipe left or right to see more examples for the word you’re learning.
FluentU helps you learn fast with useful questions and multiple examples. Learn more.
The best part? FluentU remembers the vocabulary that you’re learning. It gives you extra practice with difficult words—and reminds you when it’s time to review what you’ve learned. You have a truly personalized experience.
Start using the FluentU website on your computer or tablet or, better yet, download the FluentU app from the iTunes or Google Play store. Click here to take advantage of our current sale! (Expires at the end of this month.)
Enter your e-mail address to get your free PDF!
We hate SPAM and promise to keep your email address safe


Want to create or adapt books like this? Learn more about how Pressbooks supports open publishing practices.
20 Considerations and Strategies for Oral Presentations
Introduction.
Whether short, informal and require sharing of specific information within a small group, or more lengthy and structured, requiring visual aids and sharing on a specific topic to a larger audience, oral presentations are anxiety producing.

What do you think are the biggest causes of anxiety for your students associated with giving oral presentations? Are there specific anxieties you would associate with the different groups of learners in your class?
Did You Know?
About 25% of the population are anxious and afraid to make presentations. According to neuropsychologist Dr. Theo Tsaousides as outlined in the article “ Why Are We Afraid of Public Speaking ?” there are many factors which contribute to this sense of dread induced by the thought of speaking in public (glossophobia). Four of the main contributing factors are:

- Physiology – the autonomic nervous system is aroused in response to a potentially threatening stimulus which leads to fear – this fear of public speaking can be heightened for some who then worry about how their fear will manifest itself and how they will be perceived.
- Thoughts – personal beliefs about public speaking and about themselves as speakers can lead to inflating the stakes of the speech event. Having personal self-doubt about one’s own ability to communicate effectively can increase anxiety and fear
- lack of experience presenting in the target format,
- fear of being evaluated on the performance,
- status difference – presenting with the professor present or presenting to others perceived to be more competent
- presenting something new – the topic many not be that familiar and there is a fear of being asked related questions that the speaker is unable to answer
- new Audiences – presenting to the unfamiliar people and not knowing what to expect induces fear
- Skills – how well a speaker is able to communicate. Practice helps develop skills, even for naturally good speakers.
If 25% of the population experience fear and anxiety when speaking in public, think about how amplified that likely is for some non native speakers who are speaking publicly in a language that is not their mother tongue. On top of overcoming the nerves many feel while speaking, they also have to be processing their thoughts and converting them to another language with all its nuances, grammar, pronunciation and structures, under pressure to perform according to North American expectations in a given amount of time and be evaluated – now that’s pressure!

It might be time to reflect on how you are actually using presentations in your course and if there might be a better way for your students to showcase their understanding of and ability to apply the course concepts. Based on the principles of socio-cultural constructivism, UDL and CRP, offering options for students to choose from in order to demonstrate their learning can lead to higher levels of engagement and achievement. If the ability to create and make a presentation is not part of your learning outcomes, you may want to consider some alternate dynamic learning ideas and tools outlined in Dr. Matthew Joseph’s post “ Moving to Dynamic Learning ”
Keys to Supporting Your ELLs’ Oral Presentation Success

Create a Linguistic and Culturally Inclusive Space
You have learned about the linguistic and cultural diversity of students in Module 2 and the need to create an equitable and supportive environment in Module 3 . Looking at all teaching and learning through a CRP lens and adhering to UDL principles helps to create inclusive learning spaces.
Determine English Expectations
In the classroom your linguistic expectations are communicated through course outcomes, assignments and rubrics as well as classroom interactions. Your treatment of linguistic diversity signals acceptable attitudes and behaviours within the class towards the sociolinguistic diversity of the ELLs.
Foster Tolerance of Sociolinguistic Diversity

Determine Acceptable Varieties of English
There is no question that clarity of communication is important for both native and non native speakers in your class. For ELLs, this generally means using the grammar and vocabulary appropriate for the context; using the wrong tense or incorrect pronoun can interfere with comprehension. Furthermore, using an unfamiliar word or placing the accent on the wrong syllable making the word unrecognizable can also obscure the clarity of the transaction. However, English grammar and vocabulary usage are not universal. Consider the differences in countries such as the USA, Australia New Zealand, Ireland, South Africa and some Caribbean territories where the official language is English. Do we all speak the same English? Do we have the same way of organizing our thoughts and conveying ideas? Do we all use the same word to describe an object or is it dependent on the geographic location? Do we speak with the same accent? Of course the answer is ‘no.’ Then when we take into account all the countries that use English for transactional purposes around the world, there is even more variety in the English used. Native speakers of English are actually in the minority of English speakers globally. English is used as the language of communication (lingua franca) between populations that do not share a common language and is the universal language used in the science fields, for many jobs such as pilots and is the default choice for transnational communication. This lingua franca is referred to as World English as the varieties and registers do follow standardized patterns of use associated with the profession. While this use of English globally is generally aligned with the accepted practices in certain disciplines, the notion of a standard English in today’s global environment is unrealistic. The term ‘World Englishes’ better captures the reality of English used globally. Localized or indigenized varieties of English have emerged in various contexts globally as a result of contact with local and other languages and usage purpose .
Distinguish Between Pronunciation and Accent
Like grammar, pronunciation can sometimes interfere with others’ ability to comprehend the speaker. Pronunciation of certain consonant or vowel sounds (segmentals) may be strongly influenced by the ELL’s first language. In some languages, there may be words that are the same or very close to the same word in the ELL’s native language; however, the stressed syllable (suprasegmental) may be different, so when spoken aloud, the word may not be recognizable by others in the class. ELLs are not alone in mispronouncing words. It is common for native speakers to incorrectly pronounce unfamiliar words.
- It’s helpful for all learners in your class to see key vocabulary written and provide the pronunciation of the words orally.
- Encourage learners to use text to speech software so they can hear the pronunciation of their pronunciation. MS Word had a screen reader function or you can you a program like Balaboka. Learners can record their presentation script and then read along to practice the pronunciation.
Accents, on the other hand, should not interfere with comprehension. However, it is common for NA students to express frustration at not being able to understand some ELLs in the class or even their teachers who may have a pronounced accent. Linguistics research indicates that decoding accents is possible if learners have the right mentality. Researchers found that students’ personal expectation of how easy or difficult a speaker will be to understand plays a significant role in their ability to understand the speaker’s accent (Stellino, 2019).

Watch the video about strong accents or read the transcript Strong Accent Transcript [Word] S4 Strong Accent Transcript [PDF] . Do you agree with the speaker?
The notion of ‘English’ today needs to be retuned from thinking of it as a single, monolithic entity, a linguistic ‘standard’ and a reference system, to understanding it as a set of related, structurally overlapping, but also distinct varieties, the products of a fundamental “glocalization” process with variable, context-dependent outcomes (Schneider, 2018).

Consider the backgrounds of the students in your class and their purpose for taking the course. The content knowledge is very important for all the learners but is the English standard equally as important? Will your ELLs be working or living in an environment that requires strict adherence to North American ‘standards?’ Or is communicating competently in World Englishes their goal? The weighting and priority you place on oral communication using ‘standard’ English should reflect the realities of global communication.
- Check in with your students and ask them how and where they think they will be using the knowledge and skills covered in your class.
- Ask your students about what their personal goals are for their English oral skills.
Class Engagement During Presentations
In addition to performance anxiety, many learners worry about performing in front of their peers. It is important to collaboratively develop expectations about the responsibilities and expected behaviours of others in the class during a learner’s or group of learners’ presentation. Determine the following:
- Who is managing the class? Is the presenter responsible for the class during the presentation? Should the presenter ensure that the class is paying attention?
- What expectations are there around chatter in the class during the presentation? Do you need to teach learners strategies such as using physical proximity to draw the chatters’ attention? Should the presenter stop talking? Should the presenter ask for attention?
- Should the audience have a specific task associated with each presenter so they are required to actively listen to the presentation to successfully complete the task? Should answers to the task be assessed by the teacher for marks?
- What should happen if the presenter goes over the allotted time?

Stack Exchange Network
Stack Exchange network consists of 183 Q&A communities including Stack Overflow , the largest, most trusted online community for developers to learn, share their knowledge, and build their careers.
Q&A for work
Connect and share knowledge within a single location that is structured and easy to search.
How to correctly pronounce 'presentation'?
I have been confident with my pronunciation of the word presentation , which is /prɛz(ə)nˈteɪʃ(ə)n/ or /ˌprez.ən'teɪ.ʃən/ .
In the Cambridge Advanced Learner's Dictionary, the pronunciation of this word is as above in both AmE and BrE. I also listen to the human pronunciation given by the dictionary and decide that it must be /ˌprez.ən'teɪ.ʃən/ . Also, on http://dictionary.cambridge.org , both AmE and BrE versions are pronounced as /prez.ən'teɪ.ʃən/ .
However, on the Oxford Learner's Dictionaries ( http://www.oxfordlearnersdictionaries.com ), this word is pronounced as BrE /ˌpreznˈteɪʃn/ , AmE /ˌpriːzenˈteɪʃn/ with the human pronunciation being clearly different.
So my questions:
- Why do the two 'reliable' dictionaries give different versions of pronunciation of presentation ?
Are both right? Which is more common?
- american-english
- pronunciation
- british-english
- 2 Isn't it simply a case of "British prez-en-tation , American pree-zen-tation "? – WS2 Commented Jun 8, 2017 at 10:37
- 3 Why are you paying attention to British dictionaries when trying to figure out whether a variant pronunciation in American English pronunciation? All three American dictionaries I checked indicate that both pronunciations are acceptable. – Peter Shor Commented Jun 8, 2017 at 10:43
- 4 I hear both versions, and while the short E version is likely more common (here in the Midwest US), the long E version is fairly common as well. – Hot Licks Commented Jun 8, 2017 at 11:24
- 3 What is your question? Can a word such as "presentation" have two different, correct pronunciations–even in the same country? The answer is yes. As a native speaker of American English, I use /prez/ and the /priz/ variant seems odd, but we don't all speak the same. – Arm the good guys in America Commented Jun 8, 2017 at 11:30
- 1 You guys seem to miss my points. What I mean is learners of English in their non-speaking countries depend on dictionaries to learn English. But Cambridge says only /ˌpreznˈteɪʃn/ for both AmE and BrE while Oxford teaches them that /ˌpreznˈteɪʃn/ is BrE and /ˌpriːzenˈteɪʃn/ is AmE. – Niamh Doyle Commented Jun 8, 2017 at 13:59
2 Answers 2
Some answers to your questions:
Why do the two 'reliable' dictionaries give different versions of pronunciation of presentation?
Who told you that these were "the two 'reliable' dictionaries"? These are both British dictionaries, and both seem to treat American pronunciations more or less as a side issue; they don't always get them correct. They are very reliable for the definitions of the words (at least the British English definitions, which are the same for most words as the American definitions) and the British pronunciations.
Two of the American dictionaries I checked ( Merriam-Webster and American Heritage ), say that both /ˌprezənˈteɪʃən/ and /ˌpriːzənˈteɪʃən/ are acceptable pronunciations. I hear both. So for this particular word, these American dictionaries are batting 1000, while the British dictionaries are not. Unfortunately, neither of them gives pronunciations in IPA.
Which is more common? That may depend on which region of the U.S. that you live in, and I wouldn't want to guess the answer without access to a nation-wide survey.
- 1 For what it’s worth, in a 2007 update for presentation the OED gives Brit. /ˌprɛznˈteɪʃn/, U.S. /ˌprɛznˈteɪʃ(ə)n/, /ˌpriˌzɛnˈteɪʃ(ə)n/, /ˌpriznˈteɪʃ(ə)n/. I can’t say that the /priz/ pronunciation personally rings any bells; I’ve only ever said /prɛz/. I can’t imagine anyone in the world ever saying /prez/ like prays . – tchrist ♦ Commented Feb 17, 2018 at 16:43
Because English speakers pronounce the word in different ways.
Yes, both are right. I don't know which is more common. But if you choose a pronunciation from a reliable dictionary, there should be no problem. If you chat with other speakers and hear them pronounce the word differently from you, then maybe you can change the way you pronounce it. Reliable dictionaries won't give you bad pronunciations. You can also check forvo.com and listen to various native pronunciations.
It is also simply not true that, to quote part of your comment,
learners of English in their non-speaking countries depend on dictionaries to learn English.
Maybe you do. Most learners I know rely on more natural sources, such as podcasts, forvo, speaking with native speakers on Italki, Verbling, GoSpeaky, etc., YouTube videos such as Rachel's English, listening to English radio, watching movies, etc. Yes, you can start by consulting a dictionary, but you should rely more on these other sources if you're truly studying and learning the language. And I haven't even mentioned taking English classes or hiring a tutor. Okay, now I have mentioned them.
Not the answer you're looking for? Browse other questions tagged american-english pronunciation british-english or ask your own question .
- Featured on Meta
- Upcoming sign-up experiments related to tags
Hot Network Questions
- Having friends who are talented is great, but it can also be ___ at times
- "Better break out the weapons" before they leave the ship and explore the planet -- what kind?
- What does "far right tilt" actually mean in the context of the EU in 2024?
- Brake Line no Longer Connected to Brake Lever
- Why isn't "meanwhile" advisable in this sentence? Doesn't it mean "at the same time"?
- Poorly-acted 90s–2000s low-budget sci-fi movie with ill-fated desert miners and a man-eating monster
- In the Unabomber case, was "call Nathan R" really mistakenly written by a New York Times intern?
- Ubuntu Terminal with alternating colours for each line
- what does the word of seven book- length means
- What is the origin of the idiom "say the word"?
- Structure that holds the twin-engine on an aircraft
- What do humans do uniquely, that computers apparently will not be able to?
- Can someone explain the damage distrubution on this aircraft that flew through a hailstorm?
- Quote identification: Alles, was man lernen kann, lohnt sich nicht, gelernt zu werden
- can you roll your own .iso for USB stick?
- Preventing Javascript in a browser from connecting to servers
- What is the point of triggering of a national snap election immediately after losing the EU elections?
- Some congruence problem
- How to explain Hazard Ratio in layperson's terms
- Is the Residue Theorem applied correctly?
- Do reflective warning triangles blow away in wind storms?
- possible to change sub-shortcuts of basic tools like 'move' [bpy.ops.transform.translate]?
- What changes the velocity perpendicular to radius in an elliptical orbit?
- Why did the drywall tape fail in my garage? How can I fix it?
Grand Valley State University
Search people & pages, office of undergraduate research and scholarship.
- Student Ambassadors
- Undergraduate Research Council
- What is Research?
- How to Get Started
- Programs & Grants
- Research/Creative Scholar Transcript Designation
- Student Spotlights
- Engaging Students
- Funding Opportunities
- Faculty Resources
- Outreach Presentation Request
- Chalk Art Symposium
- Undergraduate Research Fair
- Student Scholars Day
- Student Summer Scholars
- Events Calendar

Oral Presentation Tips
An oral presentation is more than just reading a paper or set of slides to an audience. How you deliver your presentation is at least as important in effectively communicating your message as what you say. Use these guidelines to learn simple tools that help you prepare and present an effective presentation, and design PowerPoint slides that support and enhance your talk.
Preparing an Effective Presentation An effective presentation is more than just standing up and giving information. A presenter must consider how best to communicate the information to the audience. Use these tips to create a presentation that is both informative and interesting:
- Organize your thoughts. Start with an outline and develop good transitions between sections. Emphasize the real-world significance of your research.
- Have a strong opening. Why should the audience listen to you? One good way to get their attention is to start with a question, whether or not you expect an answer.
- Define terms early. If you are using terms that may be new to the audience, introduce them early in your presentation. Once an audience gets lost in unfamiliar terminology, it is extremely difficult to get them back on track.
- Finish with a bang. Find one or two sentences that sum up the importance of your research. How is the world better off as a result of what you have done?
- Design PowerPoint slides to introduce important information. Consider doing a presentation without PowerPoint. Then consider which points you cannot make without slides. Create only those slides that are necessary to improve your communication with the audience.
- Time yourself. Do not wait until the last minute to time your presentation. You only have 15 minutes to speak, so you want to know, as soon as possible, if you are close to that limit.
- Create effective notes for yourself. Have notes that you can read. Do not write out your entire talk; use an outline or other brief reminders of what you want to say. Make sure the text is large enough that you can read it from a distance.
- Practice, practice, practice. The more you practice your presentation, the more comfortable you will be in front of an audience. Practice in front of a friend or two and ask for their feedback. Record yourself and listen to it critically. Make it better and do it again.
PowerPoint Tips Microsoft PowerPoint is a tremendous tool for presentations. It is also a tool that is sometimes not used effectively. If you are using PowerPoint, use these tips to enhance your presentation:
- Use a large font. As a general rule, avoid text smaller than 24 point.
- Use a clean typeface. Sans serif typefaces, such as Arial, are generally easier to read on a screen than serif typefaces, such as Times New Roman.
- Use bullet points, not complete sentences. The text on your slide provides an outline to what you are saying. If the entire text of your presentation is on your slides, there is no reason for the audience to listen to you. A common standard is the 6/7 rule: no more than six bulleted items per slide and no more than seven words per item.
- Use contrasting colors. Use a dark text on a light background or a light text on a dark background. Avoid combinations of colors that look similar. Avoid red/green combinations, as this is the most common form of color blindness.
- Use special effects sparingly. Using animations, cool transition effects, sounds and other special effects is an effective way to make sure the audience notices your slides. Unfortunately, that means that they are not listening to what you are saying. Use special effects only when they are necessary to make a point.
Presenting Effectively When you start your presentation, the audience will be interested in what you say. Use these tips to help keep them interested throughout your presentation:
- Be excited. You are talking about something exciting. If you remember to be excited, your audience will feel it and automatically become more interested.
- Speak with confidence. When you are speaking, you are the authority on your topic, but do not pretend that you know everything. If you do not know the answer to a question, admit it. Consider deferring the question to your mentor or offer to look into the matter further.
- Make eye contact with the audience. Your purpose is to communicate with your audience, and people listen more if they feel you are talking directly to them. As you speak, let your eyes settle on one person for several seconds before moving on to somebody else. You do not have to make eye contact with everybody, but make sure you connect with all areas of the audience equally.
- Avoid reading from the screen. First, if you are reading from the screen, you are not making eye contact with your audience. Second, if you put it on your slide, it is because you wanted them to read it, not you.
- Blank the screen when a slide is unnecessary. A slide that is not related to what you are speaking about can distract the audience. Pressing the letter B or the period key displays a black screen, which lets the audience concentrate solely on your words. Press the same key to restore the display.
- Use a pointer only when necessary. If you are using a laser pointer, remember to keep it off unless you need to highlight something on the screen.
- Explain your equations and graphs. When you display equations, explain them fully. Point out all constants and dependent and independent variables. With graphs, tell how they support your point. Explain the x- and y-axes and show how the graph progresses from left to right.
- Pause. Pauses bring audible structure to your presentation. They emphasize important information, make transitions obvious, and give the audience time to catch up between points and to read new slides. Pauses always feel much longer to speakers than to listeners. Practice counting silently to three (slowly) between points.
- Avoid filler words. Um, like, you know, and many others. To an audience, these are indications that you do not know what to say; you sound uncomfortable, so they start to feel uncomfortable as well. Speak slowly enough that you can collect your thoughts before moving ahead. If you really do not know what to say, pause silently until you do.
- Relax. I t is hard to relax when you are nervous, but your audience will be much more comfortable if you are too.
- Breathe. It is fine to be nervous. In fact, you should be all good presenters are nervous every time they are in front of an audience. The most effective way to keep your nerves in check aside from a lot of practice before hand is to remember to breathe deeply throughout your presentation.
- Acknowledge the people who supported your research. Be sure to thank the people who made your research possible, including your mentor, research team, collaborators, and other sources of funding and support.
Sharing your work can help you expand your network of contacts who share your research interests. For undergraduate researcher who intend to complete a graduate degree, presenting can be an invaluable experience. We recommend discussing your interest in sharing your research with your faculty advisor. They can help match your interests with the appropriate venue.
Office of Undergraduate Research and Scholarship Center for Undergraduate Scholar Engagement 230 Mary Idema Pew Library 1 Campus Drive Allendale , Michigan 49401
(616) 331-8100 ours@gvsu.edu
Monday - Friday 8:00 a.m. - 5:00 p.m.
Social Media
https://www.facebook.com/GVSU.OURS https://www.instagram.com/cuse_gvsu/ https://www.linkedin.com/groups/4644589/profile https://twitter.com/CUSE_GVSU https://www.youtube.com/channel/UCyjiHk8_L10kO2QIn9NcoFA
- GVSU is an AA/EO Institution
- Privacy Policy
- HEERF Funding
- Disclosures
- Copyright © 2024 GVSU
- Uncategorized
Importance of Pronunciation
Is Pronunciation important? Does it really affect our communication if we mispronounce any word? Let’s talk about the importance of pronunciation.
Pronunciation plays a major role when we talk about English speaking skills. As a fluent English speaker, one needs to ensure that he makes the right pronunciation if he wants to convey the message effectively and properly.
The correct pronunciation is extremely important especially when you are speaking English. The significant fact about pronunciation is that it is not something that you can learn but it’s a physical skill that you need to practice and master regularly.
Now, the question arises of how to master this. First of all, you need to identify your areas of improvement that means to find out the words which you are mispronouncing and following the right approach to learning the right pronunciation.
Although many language aspirants go for advanced English speaking course online to make sure that they get the right training to fix their all the pronunciation errors. You may join our professional English speaking course if you wish to overcome all your pronunciation issues.
#08 quick tips to improve yourself
Listen to yourself (Record yourself)
Are you also the one who doesn’t like listening to your voice? If yes, then my dear friend, you are not alone, most people feel the same. The fact is that most people don’t like to listen to their voice which is quite funny. But this is the only way you can work on your pronunciation. To know, what words or letters you are mispronouncing, you first need to identify your errors, so that you can accordingly prepare and practice yourself. If you are not aware of the mistakes you are making, how will you be able to correct?
Try to record your voice on your smartphone and make notes of areas you need to brush up on. Another advantage of doing this activity is that it will make you feel confident as you will realize that you don’t speak that badly as you might think. So, go and start doing it from today itself.
Speak Slowly
What do you mean by being fluent? Speaking very fast without taking any pause is fluency! A big NO is an answer. Fluency doesn’t mean that you have to speak very fast. Speaking with the right pauses at right time is fluency. Why are we talking about this here? Let me tell you, when we try to speak very fast just to leave a strong impression on others, we forget that we are committing many errors based on pronunciation. If you want to speak effectively and impressively, you need to slow down your pace. This will not only help you to become a consistent and confident speaker but will also allow you the time to think about what you want to say next.
So, if you are a beginner and make pronunciation errors, this point is highly recommended for you all. Speak slowly and gradually master the art of pronunciation. You may also join our American English speaking course online and British English speaking course for better practice.
Pronunciation is physical exercise
Why do we go to the gym? We do because we want our body in shape. It’s a physical exercise only which helps you to become well-built. The same applies to pronunciation as well. You have to follow the same approach, nothing needs to be done differently. Pronunciation is also a physical skill. How? You need to move and use your mouth muscles differently to learn correct pronunciation. For example, having trouble with ‘th’ sound. For this put your tongue between your teeth and blow air out of your mouth. Feel the air give way the highest of your tongue. For this, you can join English speaking course online and practice the art of communication effectively with right guidance.
Self assess yourself/Mirror technique
How can you do it?
Practice in front of the mirror to see the placement of your tongue, lips, and shape of your mouth when you make certain sounds. Believe me, this is one of the best techniques that you can follow and see the visible changes very soon. Self assessment makes you realize your areas of improvement. Once you know this, you should go for some online English communication classes that provide you the platform to improve yourself.
Listening to English audios
We can’t avoid the fact that we learn English not only by speaking but also by listening. Therefore, this is recommended for all English learners to listen to English programs and watch movies in English. Concentrate on every single word spoken by them. Imitate the same word you are struggling to understand at the same time when you are hearing.
Don’t become Shy: Practice in Isolation
Why do we face pronunciation problems? Because we are too conscious to present ourselves in front of others. First of all, we have to grow our confidence level and need to start speaking to ourselves. Sounds funny? But it’s not. When you practice speaking alone, you become more confident because no one is watching you. How can you do it? You are supposed to create some imaginary situations like meeting your colleagues, friends, doing shopping, etc. Let’s get started with our English communication course online and make learning fun.
How to pronounce long words?
Do you face problems in pronouncing long words? Here, you need to understand the syllables. What is a syllable? Breaking or diving the word into small parts is called syllable.
Each word is made up of syllable. When you break the word into parts (Syllables),it makes the speaker pronounce the word easily. How to know that how many syllables are there in a word? For this, you should place your hand flat under your chin. Then try to say the word slowly. When your chin touches your hand, that’s a syllable. Or you can also write the syllable word and draw a line or leave a space between each syllable.
Pay attention to intonation and emphasize stress
We just don’t speak individual words with individual sounds. Good pronunciation is also about having good command and understanding of your tonality (rise and fall of the voice) and stress (putting emphasis and making different sounds for the powerful words of the sentence). How can you practice doing this? Here comes the role of listening wherein you should listen to various poems, lectures, speeches to have a better understanding of stress and intonation.
Have fun and enjoy reading.
No Comments
Leave a reply cancel reply.
Save my name, email, and website in this browser for the next time I comment.
Recent Posts
- The Significance of Oral Communication in the Digital Age
- Strategies for Enhancing Business Writing
- Unlocking Success: Fueling Motivation and Mastering Strategies Before Learning English Language
- How English Language Holds Immense Importance in Our Lives
- 10 Common Body Language Mistakes to Avoid in Professional Settings
Back to Blog
How to think in english, scan and pay.
You can send money using this QR code. Use any UPI app to make transactions.
Academia.edu no longer supports Internet Explorer.
To browse Academia.edu and the wider internet faster and more securely, please take a few seconds to upgrade your browser .
Enter the email address you signed up with and we'll email you a reset link.
- We're Hiring!
- Help Center

The Impact of English Pronunciation on Effective Oral Communication

2019, International Journal of Science, Engineering and Technology Research (IJSETR)
The title of this paper is the impact of English pronunciation on effective oral communication. Therefore, in this paper, the researcher aims to explore whether the Sudanese secondary EFL learners' weakness in English pronunciation is regarded as a main obstacle for them to speak in English. Consequently, the researcher investigates the relation between pronunciation and oral communication in English and tries to show that pronunciation is one of the most important elements of effective oral communication. In spite of the important of English pronunciation skill in the field of language learning and teaching as one of the most important elements of effective oral communication, English pronunciation is still an area of teaching which is often neglected by English EFL teachers and learners at secondary schools in Sudan. Therefore, learners at secondary schools in Sudan have many problems with pronunciation in English when they try to speak in English. To that end, this paper focuses on areas of pronunciation problems that face learners at secondary school in Sudan, which lead to serious breakdown in oral communication in English and then tries to find solutions to them so as to ensure effective English oral communication. Thus, the researcher describes the causes behind the problems of pronunciation that the Sudanese learners of English at secondary schools face while trying to speak in English and the ways to cope with them. In this paper, the researcher, followed the descriptive analytic method and used a questionnaire as a main tool for data collection. The data gathered from the questionnaire are analyzed qualitatively. The results of the study displayed that the Sudanese secondary EFL learners' weakness in English pronunciation is regarded as a main obstacle for them to speak in English.
Related Papers
English Language and Literature Studies
Elkhair Hassan
Abdelmadjid Benraghda
Majority of Arab EFL students reach the university with generally poor English and bad pronunciation in particular. Students in Arab countries have little opportunities to speak English where Arabic is their native language. Since correct pronunciation is very important to avoid misunderstanding when communicating with others and to build self-confidence when trying to speak in front of other people, the current paper investigates the reasons for Arab EFL students to face problems in English pronunciation. The participants were 10 Preparatory Intensive English (PIE) students of University Malaysia Pahang (UMP). Using an in-depth qualitative methodology which is individual interviews, all interviews were audio-recorded with the participants’ consent. The participants were undergraduate Arab EFL students who completed their high school and they do not have exposure to English language. All recorded interviews were analyzed manually. From the data analyzed some emergent themes were fou...
Mohammad Shariq
BAIUST Academic Journal
The study was conducted to test the impact of factors on English pronunciation competence and oral presentation performance of the tertiary-level EFL learners in Bangladesh. The data was obtained from eighty students and thirty teachers from the departments of English in four Bangladeshi private and public universities. The experiment research was based on the survey of a self-developed Likert scale questionnaire and independent t-test. The results were analyzed using the Statistical Package for Social Science (SPSS). The independent t-test was statistically significant. Therefore, we rejected the null hypothesis. The independent t-test and the survey results showed a significant negative effect of factors on the ESL learners' pronunciation competence and English oral performance. The findings call for further research regarding what methods and techniques can be used to develop learners' pronunciation competence and English oral performance. Cite: Noor, L. (2021). Factors Affecting EFL Learners’ English Pronunciation Competence and Oral Presentation Performance: A Study on Tertiary Level in Bangladesh. BAIUST Academic Journal, 2(1), 174–188.
English Language Teaching and Linguistics Studies
Magdaline Nkongho
The present study aims at investigating the pronunciation components in the prescribed English Language Syllabus for High Schools as well as the pronunciation instruction in some selected High Schools in Moundou, Chad, in order to determine the impact on learners’ production with respect to this phonological feature. The content analysis method was adopted for the investigation. Data were drawn from consultation of the National English Language Syllabus for High Schools in Chad, and 44 English Teachers’ logbooks for the 2021/2022 academic year. These logbooks were collected from 6 Government High Schools in Moundou. Two research questions guided the collection of data which were later on classified, put in numerical data and analysed within the framework of the Factors Affecting Pronunciation Theory. Results of the study revealed a total lack of pronunciation prescription in the English language syllabus and a zero percent representation of pronunciation related lessons in English l...
International Journal of Research in English Education
Meisam Ziafar
Sabri T . S . Ahmed
This study was designed to investigate the spoken communication difficulties encountered by the fourth-year EFL students of some rural faculties of Aden University and the factors that might negatively contribute to their poor acquisition of spoken communication proficiency during their learning of English in these faculties to reach some suggestions and solutions that may help in changing the current situation and enabling the concerned EFL programs produce proficient speakers of English. The data were collected through varied instruments, namely students' questionnaire (n = 120), teachers' questionnaire (n = 23), students' interviews (n = 30), speaking skill teachers' interviews (n = 4), students' spoken communication proficiency assessment rubric (n = 42), classroom observations (n = 6), and an assessment of speaking skill syllabi. The collected data were analyzed by using the SPSS 21 st version and manually. The results revealed that the majority of the students encounter fluency and automaticity related difficulties and lack of communication strategies. Moreover, some students also share one or more difficulties with these two major ones, such as phonological difficulties, comprehension difficulties, sociocultural difficulties, lack of discourse knowledge, affective difficulties, and lexical and grammatical difficulties, respectively. Though the majority of the students seem to have mastered a somehow sufficient level of grammar and vocabulary, many of them cannot put their lexical and grammatical knowledge into use orally. It has also been revealed that several factors are responsible for students' poor acquisition of spoken communication proficiency. These factors can be categorized into five types, namely a. factors related to the students themselves, namely students' lack of language learning strategies to develop spoken English and lack of integrative motivation, b. factors related to student's past education, namely students' poor English level when they joined the concerned faculties in addition to the traditional learning habits they brought from their pre-tertiary education, c. factors related to students' environment, namely lack of exposure to spoken English provided by the environment, d. factors related to teaching methodology, such as lack of communicative activities, majority of the students are rarely engaged in spoken activities, mother tongue use in English classes, no language laboratory or ICTs, low qualifications of some teachers, i.e. a semi-traditional teaching setting is still dominant in most classes, and e. curriculum-related factors, namely lack of time devoted to teaching spoken communication skills and the overuse of non-English subjects. As per the results, this study has provided some suggestions and solutions that will help in changing the current situation and enabling the concerned EFL programs to produce proficient speakers of English. To get a soft copy of the thesis, e-mail me to [email protected]
Asian Journal of Humanities and Social Studies
jalal ahmad
Mr. Ahmed Ismael sefaldin Aldikheri
Abstract This study is an attempt to investigate Errors committed by Sudanese EFL learners in consonants pronunciation. Since pronunciation is a very problematic aspect of language; it is worthy to investigate. The study aimed at identifies errors committed by Sudanese EFL learners in consonants pronunciation as well as to explore the impact of mother-tongue interference and spelling-pronunciation inconsistency. Moreover, to classify errors committed by Sudanese EFL learners in consonants pronunciation into types. The study adopted quantitative and descriptive-analytic approach. The data of the study was gathered by a pronunciation test. The test was applied for (40) second year students majoring in English language at Alzaiem Alazhari University. Simple Random Sampling is the technique to draw a representative from the study population. The study achieved several findings the most important of these are; a certain English consonants are difficult to pronounce for Sudanese EFL learners. The sounds were attributed to errors committed by Sudanese EFL learners are /p/, /b/, /f/, /v/, /t/, /d/, /ʧ/ and/ʤ/. They were also encountered errors by inserting a vowel sounds to break up the consonant clusters. They dropped the last sound that occur in consonant clusters syllable-final position and there were substitution of some sounds. Mother-tongue interference and spelling-pronunciation inconsistency was appeared most frequently and seemed to be major factors that constitute errors in consonants pronunciation. In the light of these findings the study recommended the following: First, teachers should recognize the problematic sounds and teach students how to pronounce those sounds correctly. Second, according to the importance of pronunciation as essential part of learning English, textbooks should included pronunciation lessons and activities to be same as lessons in other skills. Third, tools for practicing pronunciation should be offered at schools, universities and so on. Fourth, Sudanese EFL learners of English should check the dictionary to correct pronunciation of difficult words. At last but not least; as language teachers, it is necessary to teach pronunciation and its problems to students from the early stage of education.
مجلة جامعة كركوك للدراسات الإنسانية
Anmar Saeed
Apart from accuracy, fluency is one of the issues that concern language learners and teachers on parallel grounds. Fluency is generally defined as the rapid, smooth, and effortless use of spoken language. This study aims at suggesting a method and a set of techniques that can be employed in the pronunciation classroom to work on training Iraqi learners of English to learn and use some aspects of connected speech, viz. weak forms, sentence stress, linking and elision, and accordingly developing their fluency skills in speaking English. It basically draws on using a communicative approach with the following characteristics: first, maintaining a learner-centered teaching, encouraging pair/group work, and employing practice through using activities and tasks. The suggested method involves proceeding through three stages of practice: mechanical, meaningful, and communicative. Mechanical practice is a controlled type of practice carried out through repetition and substitution activities; meaningful practice is also a controlled practice through meaningful choices of items; and the last stage, communicative practice involves using language within real communicative contexts. The suggested techniques are also set into three stages in consonance with the stages of the method.
Loading Preview
Sorry, preview is currently unavailable. You can download the paper by clicking the button above.
RELATED PAPERS
Mohamed Mutasim Mohamed Ali
Journal of communication and cultural trends
Tania Ali Khan
Journal of Science and Technology
Zahara Mustafa
English Language Teaching
A. Majid Hayati
Abbas Badawi
RELATED TOPICS
- We're Hiring!
- Help Center
- Find new research papers in:
- Health Sciences
- Earth Sciences
- Cognitive Science
- Mathematics
- Computer Science
- Academia ©2024
- help_outline help
iRubric: ESL Oral Presentation Evaluation rubric
| Rubric Code: By Draft Public Rubric Subject: Type: Grade Levels: (none) |
| Oral Presentation Evaluation | ||||
| | ||||
| | ||||
| | ||||
| | ||||
| | ||||
| | ||||
| | ||||
- Oral presentation evaluation
- Communication
- Presentation
Episode 109: Pronunciation Advice - MB Words The Déclic Anglais Podcast
- Language Learning
In this episode, we are exploring the link between English spelling (orthography) and pronunciation - especially in words that end with the letters -MB (e.g. climb, numb, thumb etc.). Tom also shares some practical tips for improving your pronunciation in general. Happy listening! Cliquez ici pour télécharger la transcription. ------------------------- The Déclic Anglais Podcast est le podcast idéal pour progresser rapidement et efficacement en anglais. Avec un nouvel épisode tous les mardis, améliorez votre compréhension orale en anglais, débarrassez-vous de vos croyances limitantes sur l'apprentissage de l'anglais, découvrez des astuces pour ne plus faire les erreurs les plus fréquentes et apprenez-en plus sur la culture britannique ! ------------------------- Vous voulez pratiquer votre anglais à l'oral pour gagner en aisance et ne plus avoir peur de parler anglais ? Nous organisons deux fois par mois des cours de conversation en anglais en ligne : le lieu idéal pour parler anglais en toute décontraction dans un petit groupe motivé ! Prochaines dates, tarifs et inscriptions ici : https://www.declicanglais.com/ateliers ------------------------- Vous souhaitez aller plus loin et améliorer votre anglais sous tous ses aspects mais ne savez pas par où commencer, quelles ressources utiliser ? En vous abonnant au Déclic Anglais Club, ayez accès chaque mois à 4 nouvelles leçons exclusives pré-enregistrées pour nos membres, avec des exercices fun et interactifs basés sur les sujets abordés dans notre podcast. Ils vous permettront d'améliorer progressivement votre écrit, prononciation, compréhension en anglais. Et avec la version Boost, les deux cours de conversation mensuels sont inclus, ainsi qu'un cours d'anglais en groupe dont le sujet est choisi chaque mois par les membres du Club ! Infos, tarifs et inscriptions ici : https://www.declicanglais.com/presentation-declic-anglais-club ------------------------- Pour toute question ou remarque, n'hésitez pas à nous écrire à l'adresse [email protected] Notre site : www.declicanglais.com Retrouvez-nous aussi sur : Facebook / Instagram ------------------------ Soundtrack credits: Kevin Hartnell: Podcast Theme, album: CC BY-SA 4.0, Overlook Hotel Records. Hébergé par Acast. Visitez acast.com/privacy pour plus d'informations.
- Episode Website
- More Episodes
- Thomas George
Watch Reimagine Education and learn what's new with responsible AI in education >
- Career readiness skills
- Published Feb 23, 2023
Strengthen presentation skills with Speaker Coach

- Content Type
- Learning accelerators
- Microsoft Teams
Concise and clear speaking is an essential communication skill that employers value. According to the National Association of Colleges and Employers , approximately 96 percent of businesses view oral and written communication as an essential competency but only 42 percent of employees are proficient at these skills. In classrooms there is always someone presenting information. Whether students are engaging in discussion or giving reports, or educators are presenting new material or conferencing with students, presentation skills are critical.
Speaker Coach , a Microsoft Learning Accelerator , enhances both foundational and workplace skills. With a focus on public speaking, Speaker Coach uses innovative technology that automatically analyzes a speaker’s communication style and helps them present information more effectively. Whether they are a native speaker or an English Language Learner, any student will benefit from the practical feedback and custom reports that highlight areas of strength and ways to improve delivery. Speaker Coach helps students not only build critical skills, but they also develop confidence in their speaking abilities while practicing.
Designed for student-driven learning and educator freedom
Intelligent, automatic help is one of many reasons why educators choose to use Speaker Coach with their students. Like other Learning Accelerators, Speaker Coach offers:
- Real-time coaching: Immediate personalized coaching and practice for students
- Inclusive design: Students can practice where they feel most comfortable and use built-in accessibility tools that increase access
- Time-saving efficiency: Learning Accelerators are built into Microsoft 365 and offer technology-assisted learning instead of teacher-directed instruction
- Actionable insights: Help track progress, display where students are challenged and excelling, and provide a more holistic view of student progress
More importantly, Speaker Coach incorporates good pedagogy. Students oversee their own learning by following detailed, pragmatic feedback that can be applied to the task at hand: improving communication clarity and enhancing public speaking skills. The analysis also enables students to learn whenever and wherever it is most convenient without needing someone else to participate.
From individual support to following a curriculum pacing guide, educators have more teaching commitments than time to complete all that is required of them. That makes Speaker Coach a valuable tool for educators; it frees time for other tasks because students can use Speaker Coach’s automatic feedback instead of direct teacher intervention. Mike Thomas, Senior Digital Learning Analyst for Springfield Public Schools, believes that Speaker Coach and other Learning Accelerators positively impact teaching in his district:
School is 180 days at six hours a day, and we must prioritize what is being taught. By having these tools available, it puts less focus trying to always deliver. It gives students the ability to learn for themselves. With tools like Reading Progress, Immersive Reader, and now Speaker Coach, I think we can take that student empowerment to the next level.” Mike Thomas, Senior Digital Learning Analyst for Springfield Public Schools
Perfect your next PowerPoint presentation
Get feedback on your presentation
Speaker Coach in PowerPoint provides suggestions while a student speaks, and it delivers a tailored, final report that is packed with ways to improve their delivery. Using a computer’s camera and microphone, Speaker Coach analyzes key qualities of effective communication:
- Pace: Monitor speed and receive pacing suggestions that increase audience recall and comprehension
- Filler Words: Highlight frequently used filler words like “um” and “you know”
- Pitch: Listen for monotone pitch that can negatively affect messaging
- Pronunciation: Isolate indistinct pronunciation or mispronunciation
- Originality: Call out instances when reading directly from a slide
- Inclusiveness: Discover when language is not inclusive in areas like disability, gender, and race
- Speech Refinements: Spot speech problems like wordiness and euphemisms
- Repetitive Words: Count words and phrases that are repeatedly spoken
- Body Language: Analyze physical mannerisms, posture, and eye contact
In addition to automatic, real-time suggestions while practicing, Speaker Coach generates a personalized report that can be used as a learning tool. Each report is customized to the speaking session, contains interactive feedback and analysis, and is privately shared so that students can securely review suggestions without the pressure of having another person listen and critique.
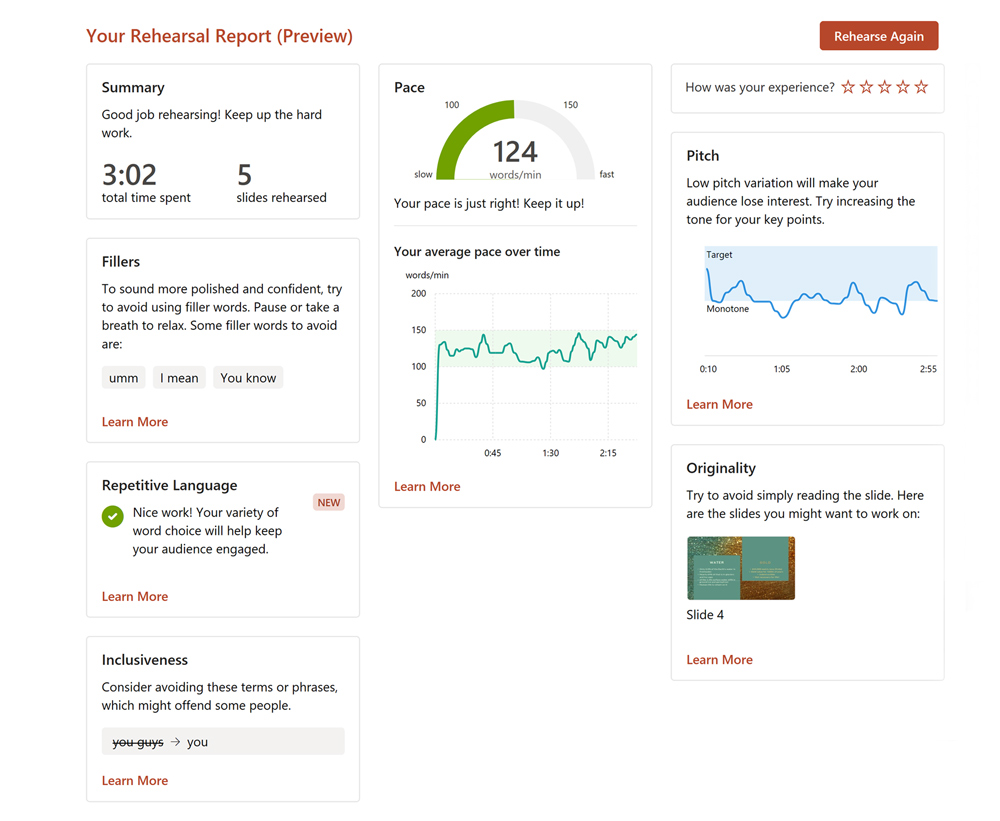
Rehearsal report summarizes your performance and provides actionable feedback
Because Speaker Coach automatically listens and analyzes multiple aspects of a presentation, students can use it at school or at home without someone else participating. Speaker Coach is like a teaching assistant who is always available and willing to offer constructive feedback in a judgement-free environment.
Students in Spring Public Schools in Springfield, MA have been using Speaker Coach to practice and prepare for classroom presentations. Educators are already seeing improvements in how ideas are communicated.
Speaker Coach is giving the students the confidence to be able to present in front of others and share their voice.” Melissa Zeitz, Digital Literacy and Computer Science Teacher for Springfield Public Schools
Rehearse with Speaker Coach
To start using Speaker Coach in PowerPoint:
- Open PowerPoint
- Add information to the slide show
- Go to Slide Show in the ribbon
- Select Rehearse with Coach
- Allow PowerPoint to use the camera and microphone
Coaching suggestions during live meetings
Students and educators can also use Speaker Coach during scheduled Teams meetings and live events. The Teams integration delivers private speaking suggestions and guidance during roundtable discussions or while sharing content from a PowerPoint or Word Document. Unobtrusive tips appear at the top of the Teams window and are only visible to the speaker. When you leave the meeting, Speaker Coach generates a timestamped report with actionable insights for improving speaking clarity, word choice, inclusiveness, and information. This report lives inside of Teams and is available to review and compare over time or throughout the year.
Follow a few simple steps to use Speaker Coach in Teams for Education:
- Schedule a Teams Meeting with a peer or colleague. Speaker Coach is unavailable in Meet Now meetings.
- Select More .
- Select Turn on Speaker Coach and follow any additional prompts. Contact your IT Administrator if you do not see this option as some clients need preview features enabled .
What are you waiting for? Say goodbye to standing in front of a mirror and talking to yourself. Instead, let Speaker Coach analyze your communication skills and reduce anxiety associated with public speaking.
Related Posts

Inspiring students during Women’s History Month 2024

Stay ahead with 8 new updates from Microsoft Education

- Customer stories
Streamline messaging with Dynamics 365 Customer Insights
Ai in education brings opportunity to life.
Watch Reimagine Education
Connect with us on social

Subscribe to our newsletter
Stay up to date with monthly newsletters from Microsoft Education.
School stories
Get inspired by stories from Microsoft Education customers.
Microsoft Learn Educator Center
Expand possibilities with educator training and professional development resources.
Contact sales
Connect with a Microsoft Education sales specialist to explore solutions for your school.
Discover a collection of resources to support a variety of educational topics.
Biora Therapeutics Announces Presentation on the BioJet™ Systemic Oral Delivery Platform at the Next Gen Peptide Formulation & Delivery Summit 2024
SAN DIEGO, June 11, 2024 (GLOBE NEWSWIRE) -- Biora Therapeutics, Inc. (Nasdaq: BIOR), the biotech company that is reimagining therapeutic delivery, today announced that it will present preclinical data from the BioJet™ Systemic Oral Delivery Platform at the Next Gen Peptide Formulation & Delivery Summit, which will take place June 18–20, 2024 in Boston.
Details of the presentation are as follows:
| Empowering Peptide Self Administration with Needle-Free Smart Capsules | |
| Pioneering Devices to Increase the Reach of Peptide APIs | |
| Wednesday, June 19, 2024 from 2:15 to 2:45 PM Eastern time | |
| Sharat Singh, PhD, Head of Research, Biora Therapeutics, Inc. |
Dr. Singh will also participate in a panel discussion on June 19 at 8:45 AM titled “Uncovering Innovations in Peptide Drug Delivery & Formulation for Improved Bioavailability,” alongside representatives from Eli Lilly, Merck, and Novo Nordisk.
Selections from the presentation will be made available on the Biora Therapeutics website following the event.
About Sharat Singh, PhD Sharat Singh brings over 25 years of experience as a dynamic and innovative scientific leader to his role at Biora Therapeutics, where he is focused on development of the company’s ingestible drug-device combination product candidates. Dr. Singh previously held scientific leadership roles at Aclara Biosciences, Prometheus Laboratories, and Nestle Health Sciences, where he established partnerships with leading biotech/pharmaceutical companies and conducted multiple phase I/II clinical trials in both oncology and gastrointestinal disease. As Chief Scientific Officer at Prometheus Laboratories, Sharat worked on topical formulation of budesonide for both Crohn’s disease (marketed as Endocort) and ulcerative colitis. He is the key inventor of multiple platform technologies (CEER, ANSER, eTag, and LOCI) and has authored over 100 patents. In his academic career, Sharat conducted inter-disciplinary research as a postdoctoral fellow at Columbia University. He holds a PhD in Chemistry from IISc, Bangalore, and has authored over 100 manuscripts and presentations.
About the BioJet™ Systemic Oral Delivery Platform Biora's BioJet systemic oral therapeutics platform uses an ingestible capsule for needle-free, oral delivery of large molecules designed to achieve systemic bioavailability and replace injection for better management of chronic diseases.
The BioJet platform uses an ingestible device designed to transit through the digestive system and activate in the small intestine, where liquid jets deliver a drug directly into the small intestine for uptake into systemic circulation. The BioJet device is approximately the size of a multivitamin and is designed to autonomously deliver a wide range of large molecules, such as proteins, peptides, and nucleic acids, in liquid formulation at multi-milligram doses, without requiring complex reformulation.
Biora holds a comprehensive patent position for the BioJet systemic oral delivery platform, with approximately 12 issued patents and 31 pending applications that cover its delivery platform and methods for using the platform to treat a disease or condition in a patient using liquid jet delivery of a wide range of drugs.
About Biora Therapeutics Biora Therapeutics is reimagining therapeutic delivery. By creating innovative smart pills designed for targeted drug delivery to the GI tract, and systemic, needle-free delivery of biotherapeutics, the company is developing therapies to improve patients’ lives.
Biora is focused on development of two therapeutics platforms: the NaviCap™ targeted oral delivery platform, which is designed to improve outcomes for patients with inflammatory bowel disease through treatment at the site of disease in the gastrointestinal tract, and the BioJet™ systemic oral delivery platform, which is designed to replace injection for better management of chronic diseases through needle-free, oral delivery of large molecules.
For more information, visit bioratherapeutics.com or follow the company on LinkedIn or Twitter.
Safe Harbor Statement or Forward-Looking Statements This press release contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, which statements are subject to substantial risks and uncertainties and are based on estimates and assumptions. All statements, other than statements of historical facts included in this press release, including statements concerning the progress and future expectations and goals of our research and development, preclinical and clinical trial activities, and partnering and collaboration efforts with third parties, are forward-looking statements. In some cases, you can identify forward-looking statements by terms such as “may,” “might,” “will,” “objective,” “intend,” “should,” “could,” “can,” “would,” “expect,” “anticipate,” “forward,” “believe,” “design,” “estimate,” “predict,” “potential,” “plan,” “target,” or the negative of these terms, and similar expressions intended to identify forward-looking statements. These statements reflect our plans, estimates, and expectations, as of the date of this press release. These statements involve known and unknown risks, uncertainties and other factors that could cause our actual results to differ materially from the forward-looking statements expressed or implied in this press release. Such risks, uncertainties, and other factors include, among others, our ability to innovate in the field of therapeutics, our ability to make future FDA filings and initiate and execute clinical trials on expected timelines or at all, our ability to obtain and maintain regulatory approval or clearance of our products on expected timelines or at all, our plans to research, develop, and commercialize new products, the unpredictable relationship between preclinical study results and clinical study results, our expectations regarding allowed patents or intended grants to result in issued or granted patents, our expectations regarding opportunities with current or future pharmaceutical collaborators or partners, our ability to raise sufficient capital to achieve our business objectives, and those risks described in “Risk Factors” and “Management’s Discussion and Analysis of Financial Condition and Results of Operations” in our Annual Report on Form 10-K for the year ended December 31, 2023 filed with the SEC and other subsequent documents, including Quarterly Reports, that we file with the SEC.
Biora Therapeutics expressly disclaims any obligation to update any forward-looking statements whether as a result of new information, future events or otherwise, except as required by law.
Investor Contact Chuck Padala Managing Director, LifeSci Advisors [email protected] (646) 627-8390
Media Contact [email protected]

Related Stocks

- Our Company
- Sustainability Approach
- Access to Medicines
- Sustainability Disclosures
- Transparency Disclosures
- Corporate Giving
- Governance Strategy
- Governance Structure
- Charters and Reports
- Areas of Focus
- Research and Development
- Our Products
- Manufacturing
- Clinical Trials
- Our Stories
- Press Releases
- Media Resources
- Financial Results
- Investor Events
- Investor Contact
Takeda’s TAK-861 Phase 2b Late-Breaking Data Presentations at SLEEP 2024 Demonstrate Clinically Meaningful Impact of Oral Orexin Agonist in Narcolepsy Type 1 Compared to Placebo
Phase 2b Trial Demonstrated Statistically Significant and Clinically Meaningful Improvements Across Primary and all Secondary Endpoints up to 8 Weeks
TAK-861 is the First Oral Orexin Receptor 2 Agonist to Potentially Address the Underlying Pathophysiology of NT1
Safety Results Indicated TAK-861 is Generally Safe and Well Tolerated
Phase 3 Trials of TAK-861 to be Initiated in 1H FY2024
OSAKA, Japan and CAMBRIDGE, Massachusetts, June 3, 2024 – Takeda ( TSE: 4502/NYSE:TAK ) will present today positive results from its Phase 2b trial of TAK-861 in narcolepsy type 1 (NT1) as late-breaking data presentations at SLEEP 2024, the 38th annual meeting of the American Academy of Sleep Medicine and the Sleep Research Society. TAK-861 is an investigational oral orexin receptor 2 (OX2R) agonist and, based on the results, has the potential to provide transformative efficacy in addressing the overall disease burden in people with NT1. The randomized, double-blind, placebo-controlled, multiple dose trial, TAK-861-2001 ( NCT05687903 Go to https://classic.clinicaltrials.gov/ct2/show/NCT05687903?term=TAK-861&draw=2&rank=3 ), in 112 patients with NT1 demonstrated statistically significant and clinically meaningful improvements across primary and secondary endpoints, with efficacy sustained over 8 weeks of treatment.*
NT1 is a chronic, rare neurological central disorder of hypersomnolence caused by a significant loss of orexin neurons, resulting in low levels of orexin neuropeptides in the brain and cerebrospinal fluid. No currently approved treatments target the underlying pathophysiology of NT1. People with NT1 suffer from excessive daytime sleepiness (EDS), cataplexy (sudden loss of muscle tone), disrupted nighttime sleep, hypnagogic and hypnopompic hallucinations and sleep paralysis. These debilitating symptoms lead to a markedly reduced quality of life and can severely impact job performance, academic achievement and personal relationships. TAK-861 is designed to address the orexin deficiency in NT1 by selectively stimulating the orexin receptor 2.
The presentation highlights results from the Phase 2b trial including:
The primary endpoint demonstrated statistically significant and clinically meaningful increased sleep latency on the Maintenance of Wakefulness Test (MWT) versus placebo across all doses (LS mean difference versus placebo all p ≤0.001). Improvements were sustained over 8 weeks.
Consistent results were achieved in the key secondary endpoints including the Epworth Sleepiness Scale (ESS) and Weekly Cataplexy Rate (WCR), demonstrating significantly improved subjective measures of sleepiness and cataplexy (sudden loss of muscle tone) frequency versus placebo that were also sustained over 8 weeks.
The majority of NT1 patients in the trial were found to be within normative ranges for MWT and ESS by the end of the 8-week treatment period as a result of these sustained improvements.
The majority of the participants who completed the trial enrolled in the long-term extension (LTE) study with some patients reaching one year of treatment.
The trial also included additional exploratory endpoints that showed meaningful improvements in narcolepsy symptoms and functioning according to most participants. These data will also be presented in poster presentations at SLEEP and at future scientific congresses.
The dataset showed that TAK-861 was generally safe and well tolerated during the study, with no treatment-related serious treatment-emergent adverse events (TEAEs) or discontinuations due to TEAEs.
No cases of hepatotoxicity or visual disturbances were reported in the Phase 2b trial or in the ongoing LTE study. The most common TEAEs were insomnia, urinary urgency and frequency, and salivary hypersecretion. Most TEAEs were mild to moderate in severity, and most started within 1-2 days of treatment and were transient.
“In this trial, TAK-861's profile balanced efficacy and safety with the potential to establish a new standard of care for people with NT1,” said Sarah Sheikh, M.D., M.Sc., B.M., B.Ch., MRCP, Head, Neuroscience Therapeutic Area Unit and Head, Global Development at Takeda. “We are dedicated to investigating the full potential of orexin biology and advancing TAK-861 to late-stage clinical trials, with the ultimate goal of delivering a potential first-in-class treatment that can make a meaningful difference for patients.”
Based on these results, and in consultation with global health authorities, Takeda plans to initiate global Phase 3 trials of TAK-861 in NT1 in the first half of its fiscal year 2024. The Phase 2b data also supported the recent Breakthrough Therapy designation for TAK-861 for the treatment of EDS in NT1 from the U.S. Food and Drug Administration (FDA). Breakthrough Therapy designation is a process designed to expedite the development and review of a drug that is intended to treat a serious or life-threatening condition, for which preliminary clinical evidence indicates that the drug may demonstrate substantial improvement over available therapies on at least one clinically significant endpoint.
Takeda will be hosting a call to discuss these data this evening, June 3, at 7:30 p.m. CT for investors and analysts. Presentation slides and a virtual meeting link will be available here .
Additional presentations on TAK-861 will be shared during the SLEEP 2024 poster presentation session on Tuesday, June 4, from 10:00 to 11:45 a.m. CT, assessing function and health-related quality of life in individuals with NT1, as well as patient satisfaction with TAK-861 treatment. There is no change in Takeda’s full year consolidated forecast for the fiscal year ending March 31, 2025 (FY2024), announced on May 9, 2024.
About Takeda’s Orexin Franchise
Takeda is advancing the field of orexin therapeutics with a multi-asset franchise offering tailored treatments to unlock the full potential of orexin. Orexin is a key regulator of the sleep-wake cycle and is involved in other essential functions, including respiration and metabolism. TAK-861 is the leading program in this franchise. The company is also progressing multiple orexin agonists in patient populations with normal levels of orexin neuropeptides and other indications where orexin biology is implicated. This includes TAK-360, an oral OX2R agonist being investigated for narcolepsy type 2 and idiopathic hypersomnia, which recently initiated a Phase 1 trial and received Fast Track designation from the U.S. FDA, and danavorexton (TAK-925), an intravenously administered OX2R agonist being investigated in a Phase 2 trial in patients with moderate to severe obstructive sleep apnea undergoing general anesthesia.
About Takeda
Takeda is focused on creating better health for people and a brighter future for the world. We aim to discover and deliver life-transforming treatments in our core therapeutic and business areas, including gastrointestinal and inflammation, rare diseases, plasma-derived therapies, oncology, neuroscience and vaccines. Together with our partners, we aim to improve the patient experience and advance a new frontier of treatment options through our dynamic and diverse pipeline. As a leading values-based, R&D-driven biopharmaceutical company headquartered in Japan, we are guided by our commitment to patients, our people and the planet. Our employees in approximately 80 countries and regions are driven by our purpose and are grounded in the values that have defined us for more than two centuries. For more information, visit www.takeda.com .
* The topline results were announced on February 8, 2024, via a press release, “Takeda Intends to Rapidly Initiate the First Global Phase 3 Trials of TAK-861, an Oral Orexin Agonist, in Narcolepsy Type 1 in First Half of Fiscal Year 2024."
Media Contacts:
Japanese media.
Yuko Yoneyama
[email protected]
+81 70-2610-6609
U.S. and International Media
Rachel Wallace
Important Notice
For the purposes of this notice, “press release” means this document, any oral presentation, any question-and-answer session and any written or oral material discussed or distributed by Takeda Pharmaceutical Company Limited (“Takeda”) regarding this release. This press release (including any oral briefing and any question-and-answer in connection with it) is not intended to, and does not constitute, represent or form part of any offer, invitation or solicitation of any offer to purchase, otherwise acquire, subscribe for, exchange, sell or otherwise dispose of, any securities or the solicitation of any vote or approval in any jurisdiction. No shares or other securities are being offered to the public by means of this press release. No offering of securities shall be made in the United States except pursuant to registration under the U.S. Securities Act of 1933, as amended, or an exemption therefrom. This press release is being given (together with any further information which may be provided to the recipient) on the condition that it is for use by the recipient for information purposes only (and not for the evaluation of any investment, acquisition, disposal or any other transaction). Any failure to comply with these restrictions may constitute a violation of applicable securities laws. The companies in which Takeda directly and indirectly owns investments are separate entities. In this press release, “Takeda” is sometimes used for convenience where references are made to Takeda and its subsidiaries in general. Likewise, the words “we”, “us” and “our” are also used to refer to subsidiaries in general or to those who work for them. These expressions are also used where no useful purpose is served by identifying the particular company or companies.
Forward-Looking Statements
This press release and any materials distributed in connection with this press release may contain forward-looking statements, beliefs or opinions regarding Takeda’s future business, future position and results of operations, including estimates, forecasts, targets and plans for Takeda. Without limitation, forward-looking statements often include words such as “targets”, “plans”, “believes”, “hopes”, “continues”, “expects”, “aims”, “intends”, “ensures”, “will”, “may”, “should”, “would”, “could”, “anticipates”, “estimates”, “projects” or similar expressions or the negative thereof. These forward-looking statements are based on assumptions about many important factors, including the following, which could cause actual results to differ materially from those expressed or implied by the forward-looking statements: the economic circumstances surrounding Takeda’s global business, including general economic conditions in Japan and the United States; competitive pressures and developments; changes to applicable laws and regulations, including global health care reforms; challenges inherent in new product development, including uncertainty of clinical success and decisions of regulatory authorities and the timing thereof; uncertainty of commercial success for new and existing products; manufacturing difficulties or delays; fluctuations in interest and currency exchange rates; claims or concerns regarding the safety or efficacy of marketed products or product candidates; the impact of health crises, like the novel coronavirus pandemic, on Takeda and its customers and suppliers, including foreign governments in countries in which Takeda operates, or on other facets of its business; the timing and impact of post-merger integration efforts with acquired companies; the ability to divest assets that are not core to Takeda’s operations and the timing of any such divestment(s); and other factors identified in Takeda’s most recent Annual Report on Form 20-F and Takeda’s other reports filed with the U.S. Securities and Exchange Commission, available on Takeda’s website at: https://www.takeda.com/investors/sec-filings-and-security-reports/ or at www.sec.gov Go to https://www.sec.gov . Takeda does not undertake to update any of the forward-looking statements contained in this press release or any other forward-looking statements it may make, except as required by law or stock exchange rule. Past performance is not an indicator of future results and the results or statements of Takeda in this press release may not be indicative of, and are not an estimate, forecast, guarantee or projection of Takeda’s future results.
Medical Information
This press release contains information about products that may not be available in all countries, or may be available under different trademarks, for different indications, in different dosages, or in different strengths. Nothing contained herein should be considered a solicitation, promotion or advertisement for any prescription drugs including the ones under development.

IMAGES
VIDEO
COMMENTS
Research confirms that in order for ELLs to acquire English they must engage in oral language practice and be given the opportunity to use language in meaningful ways for social and academic purposes (Williams & Roberts, 2011). Teaching students to design effective oral presentations has also been found to support thinking development as "the ...
Guide to preparing oral presentations in English. Read the 5 steps to creating and delivering an English presentation with confidence, with downloadable assessment. ... Use spellcheckers to check the spelling of your slides and online dictionaries to listen to the pronunciation of words (such as the Cambridge Dictionary).
Voice, or vocal sound, is made when controlled air being exhaled from the lungs, passes over the vocal cords causing a controlled vibration. The vibrating air resonates in the body, chest cavity, mouth, and nasal passages. The vibrating air causes a chain reaction with the air in the room. The room's air, set in motion by the voice, is ...
The following page intends to give some useful tips and advice on improving pronunciation whilst giving a presentation. Get physical Do some 'warm-ups' to loosen up your mouth and your tongue before you speak. You can move and stretch your mouth, and repeat tricky words and sounds. Doing this will help you to overcome the physical 'stiffness' of the articulators and to speak fluently and clearly.
How to pronounce PRESENTATION. How to say PRESENTATION. Listen to the audio pronunciation in the Cambridge English Dictionary. Learn more.
If the words you need to know are long or complicated to pronounce, you could practise them before giving the presentation in English. One way to do this is by using a free Text to Speech programme (TTS). Insert sentences into the box which include the technical words and select a British English or American English voice if possible. Do not ...
Personal online tutoring. EnglishScore Tutors is the British Council's one-to-one tutoring platform for 13- to 17-year-olds. Giving an oral presentation as part of a speaking exam can be quite scary, but we're here to help you. Watch two students giving presentations and then read the tips carefully.
Delivering effective oral presentations involves three components: what you say ( verbal ), how you say it with your voice ( vocal ), and everything the audience can see about you ( visual ). For ...
Delivery. It is important to dress appropriately, stand up straight, and project your voice towards the back of the room. Practise using a microphone, or any other presentation aids, in advance. If you don't have your own presenting style, think of the style of inspirational scientific speakers you have seen and imitate it.
presentation frequency in english - B2 level of CEFR the word presentation occurs in english on average 24.1 times per one million words; this frequency warrants it to be in the study list for B2 level of language mastery according to CEFR, the Common European Framework of Reference.
Practice pronunciation of the word oral presentation with ELSA advanced technology and say oral presentation like Americans. Product Elsa Speak Speech Analyzer Redeem Promo Code Learn English ... Let's hear how you pronounce "oral presentation" oral presentation Tap then say the word Subscribe to Pro Access. Learn More About ELSA Pro Anybody ...
Here are 4 tips that should help you perfect your pronunciation of "oral presentation":. Break "oral presentation" down into sounds: [AW] + [RUHL PREZ] + [UHN] + [TAY] + [SHUHN] - say it out loud and exaggerate the sounds until you can consistently produce them.; Record yourself saying "oral presentation" in full sentences, then watch yourself and listen.
A large part of the impact of your presentation is made by your voice and body language, yet people generally focus their preparation on content. Make sure you allocate some time to considering and practising your speaking skills and body language. Voice. Good voice control and clear articulation are essential for an effective oral presentation.
Pronunciation of 'presentation' American English pronunciation British English pronunciation American English: pr i zɛnt e ...
Using these words in your introduction and throughout your presentation will help keep the audience engaged. For example, if giving a persuasive speech, speaking directly to the audience will have a better effect: "To help lessen the effects of global warming, the planet needs you.". Stop memorizing words.
Presentation skills are the abilities and qualities necessary for creating and delivering a compelling presentation that effectively communicates information and ideas. They encompass what you say, how you structure it, and the materials you include to support what you say, such as slides, videos, or images. You'll make presentations at various ...
20 Considerations and Strategies for Oral Presentations Introduction. Whether short, informal and require sharing of specific information within a small group, or more lengthy and structured, requiring visual aids and sharing on a specific topic to a larger audience, oral presentations are anxiety producing. ... pronunciation and structures ...
Yes, both are right. I don't know which is more common. But if you choose a pronunciation from a reliable dictionary, there should be no problem. If you chat with other speakers and hear them pronounce the word differently from you, then maybe you can change the way you pronounce it. Reliable dictionaries won't give you bad pronunciations.
An oral presentation is more than just reading a paper or set of slides to an audience. How you deliver your presentation is at least as important in effectively communicating your message as what you say. Use these guidelines to learn simple tools that help you prepare and present an effective presentation, and design PowerPoint slides that ...
ORAL PRESENTATION EVALUATION CRITERIA AND CHECKLIST. talk was well-prepared. topic clearly stated. structure & scope of talk clearly stated in introduction. topic was developed in order stated in introduction. speaker summed up main points in conclusion. speaker formulated conclusions and discussed implications. was in control of subject matter.
The correct pronunciation is extremely important especially when you are speaking English. The significant fact about pronunciation is that it is not something that you can learn but it's a physical skill that you need to practice and master regularly. Now, the question arises of how to master this. First of all, you need to identify your ...
The findings call for further research regarding what methods and techniques can be used to develop learners' pronunciation competence and English oral performance. Cite: Noor, L. (2021). Factors Affecting EFL Learners' English Pronunciation Competence and Oral Presentation Performance: A Study on Tertiary Level in Bangladesh.
ESL Oral Presentation Evaluation. Evaluates the key components of an effective oral presentation for ESL students. This is a holistic assessment. Rubric Code: R3ABX9. By amygporter. Draft. Public Rubric. Subject: Communication. Type: Presentation.
In this episode, we are exploring the link between English spelling (orthography) and pronunciation - especially in words that end with the letters -MB (e.g. climb, numb, thumb etc.). Tom also shares some practical tips for improving your pronunciation in general. Happy listening! Cliquez ici pour télécharger la transcription.
Concise and clear speaking is an essential communication skill that employers value. According to the National Association of Colleges and Employers, approximately 96 percent of businesses view oral and written communication as an essential competency but only 42 percent of employees are proficient at these skills.In classrooms there is always someone presenting information.
SAN DIEGO, June 11, 2024 (GLOBE NEWSWIRE) -- Biora Therapeutics, Inc. (Nasdaq: BIOR), the biotech company that is reimagining therapeutic delivery, today announced that it will present preclinical data from the BioJet™ Systemic Oral Delivery Platform at the Next Gen Peptide Formulation & Delivery Summit, which will take place June 18-20, 2024 in Boston.
The presentations will also be available on Akero's website following the meeting. A late-breaking oral presentation will feature 96-week data from HARMONY, a Phase 2b study evaluating the efficacy and safety of EFX in patients with metabolic dysfunction-associated steatohepatitis (MASH), fibrosis stage 2 or 3 (F2-F3).
Biora is focused on development of two therapeutics platforms: the NaviCap™ targeted oral delivery platform, which is designed to improve outcomes for patients with inflammatory bowel disease ...
Additional presentations on TAK-861 will be shared during the SLEEP 2024 poster presentation session on Tuesday, June 4, from 10:00 to 11:45 a.m. CT, assessing function and health-related quality of life in individuals with NT1, as well as patient satisfaction with TAK-861 treatment.
Actinium Announces Results of Actimab-A + CLAG-M Combination Trial Highlighted in Oral Presentation at the 2024 Society of Nuclear Medicine & Molecular Imaging Annual Meeting - read this article along with other careers information, tips and advice on BioSpace. Actinium Pharmaceuticals, Inc. (NYSE AMERICAN: ATNM) (Actinium or the Company) today ...