- Campus News
- Exams and Results
- Physiotherapy
- Study Abroad/Scholarships
- universities
- Andaman And Nicobar Islands
- Andhra Pradesh
- Arunachal Pradesh
- Chattisgarh
- Dadra And Nagar Haveli
- Daman And Diu
- Himachal Pradesh
- Jammu & Kashmir
- Lakshadweep
- Madhya Pradesh
- Maharashtra
- Uttar Pradesh
- West Bengal

Thesis protocol in DNB Training: NBE issues Notice highlighting new evaluation process
New Delhi: Thesis submission is an important part of DNB training. National Board of Examinations (NBE) informs that the thesis protocol evaluated by subject matter experts and the outcome of the protocol is thereafter communicated to the concerned candidate.
NBE informs the concerned DNB candidates undergoing DNB training in an accredited hospital that they have to submit his/her thesis protocol to NBE within 3 months of commencement of the DNB training.
In its notice, the NBE stated that from 2019 January admission session, the competent authority of NBE has decided to do away with the evaluation of the thesis protocol at the level of NBE. From now onwards, the concerned NBE accredited hospital shall be required to evaluate the thesis protocol at its own level through Institutional Ethics Committee (IEC) and Scientific Research Committee (SRC) and approve the thesis protocol for carrying out the research work. The constitution of IEC & SRC has to be in accordance with the guidelines prescribed by NBE and are at Annexure 1 & 2 respectively.
Read Also: DNB Post MBBS 2019: NBE releases Vacant Indicative Seat Matrix, List of Eligible Candidates for Final Round In person Counselling
After the thesis protocol has been assessed and evaluated by IEC & SRC and has been approved for carrying out the research work, the same has to be communicated to NBE in the prescribed format which is at Annexure 3 which contains Thesis Protocol Approval Letter (Institutional Ethics Committee & Scientific Research Committee and its Composition).
This Thesis Protocol Approval Letter has to be submitted to NBE within 3 months of joining of DNB candidate in the concerned hospital for DNB training.
The DNB coordinators / academic in charges/head of the institutions have also been requested to comply with the above and adhere with the prescribed timelines so that candidates get ample time to do meaningful research work.
Candidates can also view the different annexures regarding notice on the link given below:
Click here for Annexure 1 Click here for Annexure 2 Click here for Annexure 3
Read ALso: NBE publishes Vacant Indicative Seat Matrix, List of Eligible Candidates for Final Round In person Counselling for DNB Post Diploma 2019
for more details click on the link mentioned below.
http://natboard.edu.in/
http://natboard.edu.in/pdoof/pbnotice2019/protocol/THESIS PROTOCOL IEC AND SRC.pdf?h88233erhx90wsd0q2wk2s2ks2weuigfu3g77844jh89de
Disclaimer: This site is primarily intended for healthcare professionals. Any content/information on this website does not replace the advice of medical and/or health professionals and should not be construed as medical/diagnostic advice/endorsement or prescription. Use of this site is subject to our terms of use , privacy policy , advertisement policy . © 2020 Minerva Medical Treatment Pvt Ltd
Garima joined Medical Dialogues in the year 2017 and is currently working as a Senior Editor. She looks after all the Healthcare news pertaining to Medico-legal cases, MCI/DCI decisions, Medical Education issues, government policies as well as all the news and updates concerning Medical and Dental Colleges in India. She is a graduate from Delhi University. She can be contacted at [email protected] Contact no. 011-43720751 To know about our editorial team click here


DNB Thesis Submission – Uploading Thesis – Complete overview

- December 29, 2023
- No Comments

Whatsapp me

Kerala University of Health Sciences Academic Integrity and Plagiarism Policy
- May 17, 2024

The Process of Submitting a Modified DNB Thesis
- April 20, 2024

GUIDELINES FOR SUBMISSION OF PH.D THESIS AND CD (KERALA UNIVERSITY)
- March 25, 2024

The Influence of Social Marketing on Business Entrepreneurs
- February 3, 2024

How to Register and Develop Your Business on Google My Business

How Digital Marketing Can Improve Business in Thiruvananthapuram

Understanding Digital Marketing and Its Benefits for Entrepreneurs

Checklist for Ph.D Thesis Submission and Instructions to candidates in Kerala University

Exploring the Different Types of Websites and Their Associated Costs
- January 26, 2024

The Importance of a Website for a Business Firm

How Digital Marketing Helps the Growth of a Business in the Modern World
University of kerala – prescribed format of ph.d. thesis.
- January 7, 2024


Understanding Plagiarism in Research: Guidelines and Limits
- December 11, 2023
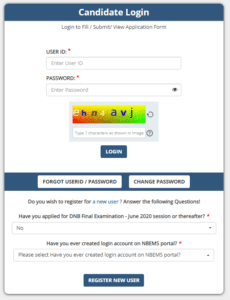
How to apply for DNB/DrNB Examination 2023?
- September 18, 2023

Instructions to Fill Online DNB/ DrNB Final Application Form 2023
- September 12, 2023

KUHS Thesis Submission Notification 2022-23
- January 5, 2023

How to use a reference management system while writing a KUHS / DNB thesis?
- December 14, 2021
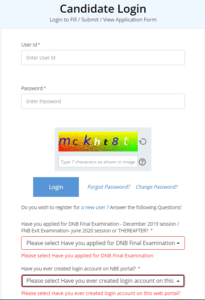
How to apply for DNB Exam 2021?
- November 5, 2021
GUIDELINES FOR DNB THESIS WRITING & SUBMISSION
- September 3, 2021
- One Comment

Guidelines for submitting Post Graduate thesis (MD/MS/MDS courses) and free download PG Thesis Template
- August 29, 2021
Leave a Reply Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
Table of Contents
Discontinuation of hard copy of thesis protocol and thesis
Putting together your final year DNB thesis is a meticulous process that needs to conform to your program’s guidelines. Here’s a general outline on the contents, formatting, and submission of your thesis. However, this is a broad guide, and you should verify the specific requirements with your program.
Kind attn: All DNB/DrNB trainees and Accredited Institutions/Hospitals.
- As per norms, submission of Thesis protocol and Thesis is essential for all DNB/DrNB trainees, towards the partial fulfilment of eligibility for Diplomate of National Board (DNB)/Doctorate of National Board (DrNB) degree.
- The process of submission of Thesis protocol and Thesis to NBEMS is done online through the NBEMS Thesis Portal and the candidates are required to submit Thesis Protocol and Thesis as per the timelines specified in the Thesis Protocol & Thesis submission
- The portal for Thesis Protocol & Thesis Submission can be accessed by clicking on below mentioned link: https://www.natboard.edu.in/thesisonline/index.php
After submitting the online Thesis Protocol and Thesis form, candidates are no longer required to send the Thesis Protocol and the hard bound thesis to NBEMS by post.

- For any query related to Thesis, please contact to NBEMS at 011- 45593000 or write at NBEMS communication web portal by clicking on the following link: https://exam.natboard.edu.in/communication.php?page=main
Soft copy to be uploaded In NBE website
- Photograph (jpg Less than 100 kb)
- Signature (jpg Les than 100 kb)
- Full thesis duly signed by the concerned authorities (pdf file less than 5 mb)
- Declaration Cum undertaking duly signed by Guide, Co-guide, Head of institution and candidate (pdf file less than 1 mb)
- Institutional Ethical Committee Approval letter (pdf file less than 1 mb)
- Thesis Application form duly signed by the concerned Authority (can be downloaded from NBE website after filling the details(pdf file <3 mb))
- Annexure Publication (pdf file less than 5 mb)
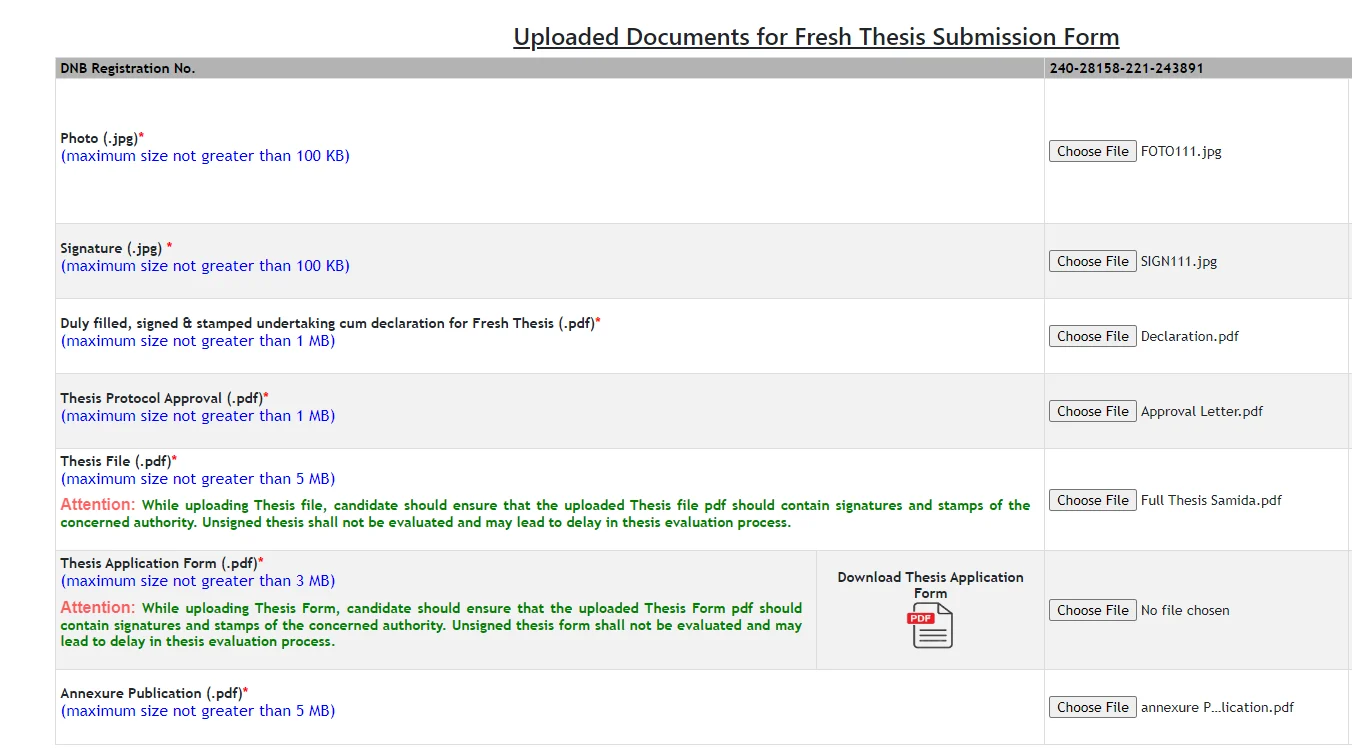
Annexure Publication
Declaration cum Undertaking for Fresh Thesis
Contents of DNB Thesis:
- Title page: Clearly state the title, your name, the program (DNB), institution, and year.
- Declaration: A statement that the work is original, followed by your signature.
- Certificate: From your thesis guide/supervisor and head of the department or institution.
- Acknowledgements: Briefly thank those who assisted you.
- Table of Contents: List of chapters with page numbers.
- List of Tables/Figures: If applicable, with titles and page numbers.
- Introduction: Describe the problem, your hypothesis, and objectives.
- Literature Review: Current knowledge/scholarly context of your work.
- Materials and Methods: Detailed enough to be reproducible.
- Results: Presentation of data; can include tables, charts, etc.
- Discussion : Interpretation of results
- Summary and Conclusion: Summary, Conclusion, implications, and potential for future work.
- References/Bibliography: Cited works using a standard citation style.
- Appendices: Any supplementary material – Ethics Committee Approval, patient consent forms, etc.
Hard copies need not to be sent to National Board of Examinations. The soft copy should be uploaded in the NBE website.
Downloadable stuffs for PG Medical Students

UDYAM Reg No : UDYAM-KL-12-0069167

- Web Development
- Ph.D Thesis
- KUHS Thesis
- Kerala University
- Digital Marketing
- [email protected]
- [email protected]
- Privacy Policy
- Refund Policy
- Terms and Conditions
WhatsApp us
- Upload File
- Most Popular
- Art & Photos
NBE Guideline for Thesis Protocol-2013 _NEW
Upload venu238
Embed Size (px) 344 x 292 429 x 357 514 x 422 599 x 487
Citation preview
7/26/2019 NBE Guideline for Thesis Protocol-2013 _NEW
1 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
NBE GUIDELINES
FOR PREPRATION & SUBMISSION
OF THESIS PROTOCOL
2 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
DESCRIPTION
1 INTRODUCTION 3
2 CONTENTS OF A THESIS PROTOCOL 57
3 ETHICS COMMITTEE 8 10
4PROTOCOL SIZE 11
3 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
INTRODUCTION
Research is defined as a systematic methodological scientific approach for basicfacts in order to find solutions based on these facts. Research investigations may be
carried out in one of two ways: interventional studies (experiments) or non
interventional surveys of naturally occurring phenomenon (descriptive andanalytical studies).
It is assumed that the protocol for a research proposal is a study plan, designed todescribe the objectives, background, methodology, organization, the participants,
interventional procedures and assessment tools of the trial. One may begin to write
a clinical protocol after many discussions among numerous individuals. Hence, we
can say that the protocol is a selfcontained document or the operating manual torefer to while conducting the research related activities.
The style used in writing research protocols depends on the skills and personality of
the candidate writing the protocol. He should refer to the Contents of a ThesisProtocol in the (NBE Guidelines) before the writing phase commences. The minimal
writing requirements are that the language should be clear, concise, precise andconsistent without excessive adjectives or adverbs and long sentences. There should
not be any redundancy in the presentation.
After the development of the first draft of the protocol he should read and rewrite as
often as necessary before the copies are submitted to other individuals for review.In reviewing ones own draft, it is helpful to question whether (1) each section is
placed in the appropriate order, (2) each paragraph within a section is placed in the
appropriate order to carry ideas and statements forward smoothly and without
gaps or sudden changes in direction or logic and (3) each sentence is having definitemeaning and is required for inclusion in the protocol. Introductory phrases at the
start of sentences that do not add meaning to the sentence should be deleted. Checkfor colloquialisms or clichs and replace them with more appropriate terms. Finally,
read each sentence by itself, for its connotations as well as denotations, before
deciding whether it conveys the intended meaning and should remain in the
Scientific writing is often made confusing when different terms are used but they
have the same or approximately the same definition. Thus a concept such as adversereactions should be written using the same single term throughout the protocol and
not sometimes as adverse experiences, adverse events, medicine reactions, adversemedicine reactions, side effects or untoward effects, unless different definitions are
intended. Other examples of when a single word or term should be used throughout
the protocol (unless differences are intended and are defined) include (1) medicine
or drug, (2) volunteers, patients or subjects (3) clinical study or clinical trial (4)disease, condition or problem (5) case report form or data collection form.
4 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
The most obvious technique for improving the quality of the protocol as alreadystated is reviewing of the protocol as critically as possible by the student. Another
established method for improving the quality of the protocol lies with the Thesis
Guide and Co Guide. It is their responsibility to proof read the protocol in terms ofboth the content and format. Finally the protocols have to be approved by the
research committee of the hospital attached and the Ethics Committee.
The development or preparation of the Thesis Protocol by the candidate will help
him in understanding the ongoing research activities in his area of interest. Further
it helps in creating practical exposure to research and hence it bridges the
connectivity between clinical practice and biomedical research. Such researchexposure will be helpful in improving problem solving capacity, updating with
ongoing research and implementing these findings in clinical practice.
5 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
The general information should be provided on the Title Page as:
i) A good title should be short, accurate, informative, and concise; it should avoid
abbreviations. It should also reflect the details of the study undertaken e.g., whethera prospective or a retrospective study / whether a crosssectional or a randomized
trial / whether an equivalence or superiority or inferiority trial etc.? [Titles like: A
study on prevalence on asthma in North India (hospital based) should not beencouraged]
ii) The name and title of the investigator who is responsible for conducting the
iii) The address and telephone numbers of the site
The summary should give a clear idea to the reader regarding the central questionthat the research is intended to answer and also its justification. It should specify
the hypothesis (if applicable) and the research objectives. In addition, the summary
should briefly describe the methods and procedures laid out in the methodology.The anticipated outcome of the study must also be mentioned.
This is critical in any protocol. It familiarizes the readers with the background of the
issue at hand. It is crucial that this is handled well. It must reflect why the issue istopical and its current importance in the vast sea of research being done globally.
This is another crucial area in a protocol. The review must be precise and concise.Unrelated articles in a review only serve to make the thesis bulky. This is against the
trend nowadays. Most universities specify the number of pages the thesis is
restricted to. An excellent review of literature brings out the lacunae in literatureand helps in generating a research question for the candidate to work on.
This is the most critical aspect to be tackled while selecting a topic. This reflects the
candidates diligence in reviewing the topic at hand. Only after the topic is reviewedwell does the candidate or researcher find out the areas in the selected topic which
need answers or are inadequately researched.
6 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
2(f) RESEARCHQUESTION
This is vital to any research proposal. Unless a valid question has been formulated
by the candidate which seeks a cogent answer the whole exercise may become
redundant. ONLY VALID QUESTIONS NEED TO BE RESEARCHED FOR SEEKING THERIGHT ANSWERS.
2(g) AIMSANDOBJECTIVES
The Aims refers to what would be achieved by this study or how this study would
address a bigger question / issue. The Objectives of the research stem from the
research question formulated and should at least include participants, intervention,evaluation, design. There should be ideally a primary objective which is the main
focus of the research proposal. The secondary objectives are the other aspects in theresearch proposal which need answers. These may include the adverse effects or the
adverse events.
METHODS: This section should include the following:
(i) Study area: The patient recruitment area (outpatient and / or inpatient of a
department in the hospital) must be listed out.
(ii) Study population: The target population to be enrolled in the study must bedefined and then patients are selected from the target population as per the
listed out inclusion and exclusion criteria.
Sample size: The number of subjects to be recruited into the study must belisted out.
Study design: The designs are experimental, descriptive or analytical. A
typical experimental design must include whether it is an open label / single
blind / double blind study, whether it is an active or placebo controlled and
whether it is an crossover or parallel design (e.g., double blind, placebocontrolled, parallel design).
A description of the measures taken to minimize / avoid bias includingrandomization and blinding, maintenance of randomization codes and
procedures for breaking codes must also be listed out.
Study intervention if any should be listed out in detail.
(vi) Studyduration: A description regarding duration of subject participationincluding followup if any and description of discontinuation criteria must
be listed out.
7 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
(vii) Methodofmeasurementofoutcomeof interest: The outcome variable
(primary and secondary) and its measurements must be defined clearly byavoiding all possible biases. The visits at which these measurements are to
be assessed and recorded must be listed out.
Methods:All the definitions of the variables and the quality
control issues regarding them must be mentioned.
Forms: All the data pertaining to the research (including
the medical history, medication history and physical examination) must be
entered onto to the data collection forms directly or transcribed from
laboratory or other forms.
2(i) STATISTICALMETHODS
This is also a critical aspect of research. The section on statistics should include thefollowing parts, namely, statistical hypothesis and sample size determination,
definition of analysis set, analysis of demographic data and baseline characteristics,analysis of efficacy and safety parameters. The statistics should also define the
analysis sets clearly from which conclusions of the study are to be derived. It is
important that the candidate decides on a sample size using the statistical formulae.Even if a sample size of convenience is used ultimately (due to time constraints in
thesis) this should be reflected in the work.
CONSIDERATION
It must be noted that the clearance of the research proposal by the EthicsCommittee is compulsory for all the studies including the ones without
interventions.
REFERENCES:
Relevant references must be listed out in Vancouver style. The habit of listing crossreferences without even reading them must be discouraged; sometimes foreign
language cross references is quoted. This is wrong. It must be seen that the
references are topical and current. The thesis with majority references being veryold shows that the candidate has not reviewed the topic well.
The Patient Information Sheet and the Informed Consent Form in English andVernacular Languages, Questionnaires, Measurement tools, Data Collection Forms,
etc. should be enclosed in the Annexure
8 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
3(a) It is mandatory to have ethics committee approval before initiation of theresearch work.
3(b) The researcher should submit an appropriate application to the ethicscommittee in a prescribed format of the ethics committee concerned. The following
documents should be submitted for review:
Protocol:The research protocol prepared having all the contents
required for a Thesis Protocol as per the NBE Guidelines.
(ii) Patient Information Sheet (PIS) in English and / or vernacular languages.
(iii) The protocol must be accompanied by the PIS addressed to the subject thathe is being asked to take part in a research study. The information provided
to him should be in simple language which he can read and understand sothat he can decide to take part in the study or refuse to take part in the study
after discussing with his family doctor or his family members. The PIS mustinclude information on the following and a copy of this must be given to the
1. What is the background to and purpose of the study?
2. Do I have to take part?3.
What will happen to me if I take part?
What do I have to do?
5. What are the possible side effects, risks and discomforts of taking part?
What are the possible benefits of taking part?7. What if new information becomes available?
8. What are the costs of taking part?9.
How will my personal data be used?
Will there be provision for free treatment for research related injury?
11. Will compensation be paid to the subjects if disability or death results from
such injury?12.
Whom should I contact if I need more information or help?
(iv) Informed Consent Form (ICF) in English and / or vernacular languages:
The subjects informed consent should be taken on the form that he or hislegally acceptable representative can read and understand. The format of the
ICF must be as follows:
9 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
Subject identification number for this trial __________________________________
Title of the Project: _____________________________________________________
_____________________________________________________
Name of the Principal Investigator ____________________ Tel. No.____________
I have received the information sheet on the above study and have read and / orunderstood the written information.
I have been given the chance to discuss the study and ask questions.
I consent to take part in the study and I am aware that my participation is
I understand that I may withdraw at any time without this affecting my future care.
I understand that the information collected about me from my participation in this
research and sections of any of my medical notes may be looked at by responsiblepersons (ethics committee members / regulatory authorities). I give access to these
individuals to have access to my records.
I understand I will receive a copy of the patient information sheet and the informedconsent form.
___________________________ __________________
Signature / Thumb Impression of subject Date of signature
______________________________
Printed name of the subject in capitals
___________________________ __________________Signature / Thumb Impression of legally Date of signature
accepted representative
10 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
signature should be added if the subject / legally acceptable representative is unable
to read or write and consent should be obtained in his presence.>>
_______________________________________________Printed name of legally acceptable representative in capitals
______________________________________________________
Relationship of legally accepted representative to subject in capitals
_______________________________________ __________________Signature of the person conducting the Date of Signature
informed consent discussion
_________________________________________
Printed name of the person conducting the
Informed consent discussion in capitals
________________________________________ __________________
Signature of impartial witness Date of signature
________________________________________
Printed name of the impartial witness in capitals
11 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013
Researchers Current CV
Any Amendment(s) to the Protocol / PIS / ICF: The amendment (s) made to
the protocol / PIS / ICF can be incorporated in the research plan only after
getting the ethics committee approval.
The thesis protocol should be restricted to about 1215 pages. The suggested
format should include:
4(a) Title and details 1 page
4(b) Synopsis 1/2 1 page
4(c) Introduction and Background 12 pages
4(d) Review of literature and lacunae 23 pages
4(e) Research question and Aims and Objectives 1 page
4(f) Material and Methods 23 pages
4(g) References 23 pages
4(h) Data Collection Forms, PIS, ICF & mandatory 46 pages
certificates

Harry potter aur_paras_patthar_(hindi)_new

Best Practices _NEW

Dswd Research Agenda _new Version

ITALAMP_2011_CATALOGUE_CULT_EDITION_01 (1)_new

BRIDGE INSPECTION FIELD MANUAL - wisconsindot.gov · 145 Masonry Arch LF NBE 58 146 Timber Arch LF NBE 49 147 Steel Main Cables LF NBE 19 148 Steel Secondary Cables EA NBE 19 152

POSSONI_2012_GENERAL_CATALOGUE_N46_01 (1)_new

Primavera Verano 2011 (2)_new

DATASHEET - Microcomsite.microcom.us/nbem2400.pdf · High-Performance airMAX ® Bridge Models: NBE-M5-19, NBE-M5-16, NBE-M2-400, NBE-M5-400, NBE-M5-300 Uniform Beamwidth Maximizes

NBE - schmid-energy.ch

NBE Directives

how_law_enforcement_utilizes_design_thinking_(2)_new (1).pptx

Santo Ki Holi March 1963 _NEW

Nbe rcausalpredictionv111 lecture2

NBE FLAMENCO
![nbe guidelines for thesis protocol 2020 Pauta Certificación 2009[1]_new](https://img.dokumen.tips/doc/110x75/5571fb15497959916993e83f/pauta-certificacion-20091new.jpg)
Pauta Certificación 2009[1]_new

RockIT _New

Nbe Book Final

NanoBeam NBE-M2-400 / NBE-M5-400 Quick Start Guide

Exercitii Seminar Psihologia Educatiei (1)_new

NBE-Corporate Capabilities

NBE Directive39 45

10 - Matematicas NBE - Programa

NBE Prescription

Dental Nbe Medental_nbe_meritlist.pdfritlist

_NEW page 46-64.indd

shah_2 (1)_new

Chapitre 1 - Configuration électroniqueChapitre 1 – Configuration électronique 9 Elément Symbol Z Nbe de protons Nbe de neutrons Nbe d’électrons MW Abondance nat. (%) Hydrogène

YNIQ _new edition

2012 TPCA presentation _New process

GRUNDFOS TPE, NBE, NKE · grundfos tpe, nbe, nke single-stage solutions for applications in commercial buildings and industry grundfos tpe, nbe, nke

IMAGES
VIDEO
COMMENTS
Online Web Portal for submission of Thesis Protocol reg. 2022-10-31. Last date for submission of Thesis for candidate scheduled to appear in DNB Final Theory Examination - March/April 2023. 2022-06-30. Extension of last date for submission of Thesis. 2022-04-16. Extension of last date for submission of thesis. 2022-01-06.
3. Refer to any one of the thesis protocol given in the Handout-2, and discuss in the group and list out the followings: Good /positive points in the write up. Deficiencies / negative points in the write up. 4. Considering the title you have finalised (refer to the handout-12,, write a draft thesis protocol.
10 Submission guidelines 23 a) NBE - General Instructions 24 b) NBE - Form for Thesis Submission 25 c) NBE - Format for summary of DNB ... It will be wise idea to take help of statistician while writing thesis protocol. 5. Sampling Procedure- The method by which the subset is selected from a larger pool is known as sampling ...
for such candidates w.e.f. 16th of December, 2020. 6. The last date for submission of Thesis Protocol documents, which is 90 days from the date of admission, is also extended till the 15th of December, 2020. 7. This issues with the approval of the competent authority. NBE
New Delhi: Thesis submission is an important part of DNB training. National Board of Examinations (NBE) informs that the thesis protocol evaluated by subject matter experts and the outcome of the protocol is thereafter communicated to the concerned candidate. NBE informs the concerned DNB candidates undergoing DNB training in an accredited ...
The protocol for a research proposal (including thesis) is a study plan, designed to describe the background, research question, aim and objectives, and detailed methodology of the study. In other words, the protocol is the 'operating manual' to refer to while conducting a particular study.
New Delhi : Through a recent notice, NBE (National Board of Examination) has informed DNB/DrNB trainees and all Accredited Institutions/Hospitals for Online Web Portal for submission of Thesis Protocol.As per NBEMS norms, Thesis Protocols is essential for all DNB/DrNB trainees towards the writing of thesis dissertation according to thesis protocols timelines.The candidates are required to...
The candidate should refer to the NBEMS guidelines for preparation and submission of thesis protocol before the writing phase commences. The minimum writing requirements are that the language should be clear, concise, precise and consistent without excessive adjectives or adverbs and long sentences. There should not be any redundancy in the ...
3 . T h e co n ce rn e d NB E a ccre d i t e d h o sp i t a l sh a l l b e re q u i re d t o e va l u a t e t h e t h e si s p ro t o co l a t
Nbe Thesis Protocol - Free download as PDF File (.pdf), Text File (.txt) or read online for free.
Delhi: The National Board of Examinations (NBE) has notified about the discontinuation of the hard copy of the thesis protocol and thesis for DNB, DrDNB Trainees.As per norms, submission of the Thesis protocol and Thesis is essential for all DNB/DrNB trainees towards the partial fulfilment of eligibility for the Diplomate of National Board (DNB)/Doctorate of National Board (DrNB)...
Maulana Azad Medical College Campus, 2, Bahadur Shah Zafar Marg, near Delhi Gate, Balmiki Basti, New Delhi, Delhi, 110002 011-23239279 011-23239271
Nbe Guidelines for Thesis Writing - Free download as PDF File (.pdf), Text File (.txt) or read online for free. nbe guidelines for thesis writing
Any change in your correspondence address after submission of thesis Protocol should be intimated to NBE at email -thesis@natboard edu.in , [email protected] so as to ... He should refer to the Contents of a Thesis Protocol in the (NBE Guidelines) before the writing phase commences. The minimal writing requirements are that the language ...
Discontinuation of hard copy of thesis protocol and thesis Putting together your final year DNB thesis is a meticulous process that needs to conform to your program's guidelines. Here's a general outline on the contents, formatting, and submission of your thesis. However, this is a broad guide, and you should verify the specific requirements with your program.
THESIS PROTOCOL & THESIS The candidates are required to submit a thesis at the end of Three/Six years of training as per the rules and regulations of NBE. Guidelines for Submission of Thesis Protocol & Thesis by candidates Research shall form an integral part of the education programme of all candidates registered for DNB degrees of NBE.
G UI DE L I NE S F O R T HE S I S W RI T I NG & S UBMI S S I O N. G UI DE L I NE S F O R T HE S I S W RI T I NG & S UBMI S S I O N. 1 . T h e p ro p o se d st u d y mu st b e a p p ro ve d b y t h e i n st i t u t i o n a l e t h i cs co mmi t t e e a n d sci e n t i f i c re se a rch co mmi t t e e . 2 .
PGI May 2019 6. FMGE 2019 7. UPSC June 2019 Surgery Sixer for NBE RECENT PATTERN QUESTIONS 2020 Disclaimer: The author and the publishers, hereby announcing that the questions discussed below are neither NEET PG 2020 questions asked in the exam nor the pattern of questions asked in the exam. If there are any similarities in the question; such ...
Nbe Thesis Protocol Submission Form - Free download as PDF File (.pdf), Text File (.txt) or read online for free.
7/26/2019 NBE Guideline for Thesis Protocol-2013 _NEW. 1/11. 1 NBE: NBE Guidelines for Preparation & Submission of Thesis Protocol 2013. NBE GUIDELINES
Guidelines for Thesis Protocol. The protocol for a research proposal (including thesis) is a study plan, designed to describe the background, research question, aim and objectives, and detailed methodology of the study. In other words, the protocol is the 'operating manual' to refer to while conducting a particular study.