An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- v.9(8); 2017 Aug


Case Reports, Case Series – From Clinical Practice to Evidence-Based Medicine in Graduate Medical Education
Jerry w sayre.
1 Family Medicine, North Florida Regional Medical Center
Hale Z Toklu
2 Graduate Medical Education, North Florida Regional Medical Center
Joseph Mazza
3 Department of Clinical Research, Marshfield Clinic Research Foundation
Steven Yale
4 Internal Medicine, University of Central Florida College of Medicine
Case reports and case series or case study research are descriptive studies that are prepared for illustrating novel, unusual, or atypical features identified in patients in medical practice, and they potentially generate new research questions. They are empirical inquiries or investigations of a patient or a group of patients in a natural, real-world clinical setting. Case study research is a method that focuses on the contextual analysis of a number of events or conditions and their relationships. There is disagreement among physicians on the value of case studies in the medical literature, particularly for educators focused on teaching evidence-based medicine (EBM) for student learners in graduate medical education. Despite their limitations, case study research is a beneficial tool and learning experience in graduate medical education and among novice researchers. The preparation and presentation of case studies can help students and graduate medical education programs evaluate and apply the six American College of Graduate Medical Education (ACGME) competencies in the areas of medical knowledge, patient care, practice-based learning, professionalism, systems-based practice, and communication. A goal in graduate medical education should be to assist residents to expand their critical thinking, problem-solving, and decision-making skills. These attributes are required in the teaching and practice of EBM. In this aspect, case studies provide a platform for developing clinical skills and problem-based learning methods. Hence, graduate medical education programs should encourage, assist, and support residents in the publication of clinical case studies; and clinical teachers should encourage graduate students to publish case reports during their graduate medical education.
Introduction
Case reports and case series or case study research are descriptive studies to present patients in their natural clinical setting. Case reports, which generally consist of three or fewer patients, are prepared to illustrate features in the practice of medicine and potentially create new research questions that may contribute to the acquisition of additional knowledge in the literature. Case studies involve multiple patients; they are a qualitative research method and include in-depth analyses or experiential inquiries of a person or group in their real-world setting. Case study research focuses on the contextual analysis of several events or conditions and their relationships [ 1 ]. In addition to their teaching value for students and graduate medical education programs, case reports provide a starting point for novice investigators, which may prepare and encourage them to seek more contextual writing experiences for future research investigation. It may also provide senior physicians with clues about emerging epidemics or a recognition of previously unrecognized syndromes. Limitations primarily involve the lack of generalizability and implications in clinical practice, which are factors extraneous to the learning model (Table (Table1 1 ).
| Advantages | Disadvantages |
| One case to initiate a signal (case report) | No control (uncontrolled) |
| Provide stronger evidence with multiple cases (cases series) | Difficult to compare different cases |
| Observational | Cases may not be generalizable |
| Educational | Selection bias |
| Easy to do (fast and no financial support needed) | Unknown future outcome/follow-up |
| Identify rare manifestations of a disease or drug |
There is disagreement among physicians on the value of case reports in the medical literature and in evidence-based medicine (EBM) [ 2 ]. EBM aims to optimize decision-making by using evidence from well-conducted research. Therefore, not all data has the same value as the evidence. The pyramid (Figure (Figure1) 1 ) classifies publications based on their study outlines and according to the power of evidence they provide [ 2 - 3 ]. In the classical pyramid represented below, systematic reviews and a meta-analysis are expected to provide the strongest evidence. However, a recent modification of the pyramid was suggested by Murad et al. [ 2 ]: the meta-analysis and systematic reviews are removed from the pyramid and are suggested to be a lens through which evidence is viewed (Figure 1 ).

Modified from Murad et al. [ 2 ]
Because case reports do not rank highly in the hierarchy of evidence and are not frequently cited, as they describe the clinical circumstances of single patients, they are seldom published by high-impact medical journals. However, case reports are proposed to have significant educational value because they advance medical knowledge and constitute evidence for EBM. In addition, well-developed publication resources can be difficult to find, especially for medical residents; those that do exist vary in quality and may not be suitable for the aim and scope of the journals. Over the last several years, a number (approximately 160) of new peer-reviewed journals that focus on publishing case reports have emerged. These are mostly open-access journals with considerably high acceptance rates [ 4 ]. Packer et al. reported a 6% publication rate for case reports [ 5 ]; however, they did not disclose the number of papers submitted but rejected and neither did they state whether any of the reported cases were submitted to open-access journals.
The development of open-access journals has created a new venue for students and faculty to publish. In contrast to subscription-based and peer-reviewed e-journals, many of these new case report journals are not adequately reviewed and, instead, have a questionably high acceptance rate [ 4 ]. There, however, remains the issue of the fee-based publication of case reports in open-access journals without proper peer reviews, which increases the burden of scientific literature. Trainees should be made aware of the potential for academic dilution, particularly with some open-access publishers. While case reports with high-quality peer reviews are associated with a relatively low acceptance rate, this rigorous process introduces trainees to the experience and expectations of peer reviews and addresses other issues or flaws not considered prior to submission. We believe that these are important skills that should be emphasized and experienced during training, and authors should seek these journals for the submission of their manuscripts.
Importance of Case Reports and Case Series in Graduate Medical Education
The Accreditation Council for Graduate Medical Education (ACGME) has challenged faculties to adapt teaching methodologies to accommodate the different learning modalities of the next generation of physicians. As evidenced by its implementation by ACGME, competency-based medical education is rapidly gaining international acceptance, moving from classic didactic lectures to self-directed learning opportunities with experiential learning aids in the development of critical cognitive and scholarly skills. As graduate medical educators, we are in agreement with Packer et al. about the value of the educational benefits resulting from student-generated case reports [ 5 ]. Case study assignments help residents develop a variety of key skills, as previously described. EBM is an eventual decision-making process for executing the most appropriate treatment approach by using the tools that are compatible with the national health policy, medical evidence, and the personal factors of physician and patient (Figure (Figure2). The 2 ). The practice of identifying and developing a case study creates a learning opportunity for listening skills and appreciation for the patient’s narrative as well as for developing critical learning and thinking skills that are directly applicable to the practice of EBM. This critically important process simultaneously enhances both the medical and the humanistic importance of physician-patient interaction. In addition, case-based learning is an active learner-centered approach for medical students and residents. It serves as a curricular context, which can promote the retention of information and evidence-based thinking.

Modified from Toklu et al. 2015 [ 3 ]
The value of case studies in the medical literature is controversial among physicians. Despite their limitations, clinical case reports and case series are beneficial tools in graduate medical education. The preparation and presentation of case studies can help students and residents acquire and apply clinical competencies in the areas of medical knowledge, practice-based learning, systems-based practice, professionalism, and communication. In this aspect, case studies provide a tool for developing clinical skills through problem-based learning methods. As a result, journals should encourage the publication of clinical case studies from graduate medical education programs through a commonly applied peer-review process, and clinical teachers should promote medical residents to publish case reports during their graduate medical education.
The content published in Cureus is the result of clinical experience and/or research by independent individuals or organizations. Cureus is not responsible for the scientific accuracy or reliability of data or conclusions published herein. All content published within Cureus is intended only for educational, research and reference purposes. Additionally, articles published within Cureus should not be deemed a suitable substitute for the advice of a qualified health care professional. Do not disregard or avoid professional medical advice due to content published within Cureus.
The authors have declared that no competing interests exist.

Health Case Studies
(29 reviews)
Glynda Rees, British Columbia Institute of Technology
Rob Kruger, British Columbia Institute of Technology
Janet Morrison, British Columbia Institute of Technology
Copyright Year: 2017
Publisher: BCcampus
Language: English
Formats Available
Conditions of use.
Learn more about reviews.
Reviewed by Jessica Sellars, Medical assistant office instructor, Blue Mountain Community College on 10/11/23
This is a book of compiled and very well organized patient case studies. The author has broken it up by disease patient was experiencing and even the healthcare roles that took place in this patients care. There is a well thought out direction and... read more
Comprehensiveness rating: 5 see less
This is a book of compiled and very well organized patient case studies. The author has broken it up by disease patient was experiencing and even the healthcare roles that took place in this patients care. There is a well thought out direction and plan. There is an appendix to refer to as well if you are needing to find something specific quickly. I have been looking for something like this to help my students have a base to do their project on. This is the most comprehensive version I have found on the subject.
Content Accuracy rating: 5
This is a book compiled of medical case studies. It is very accurate and can be used to learn from great care and mistakes.
Relevance/Longevity rating: 5
This material is very relevant in this context. It also has plenty of individual case studies to utilize in many ways in all sorts of medical courses. This is a very useful textbook and it will continue to be useful for a very long time as you can still learn from each study even if medicine changes through out the years.
Clarity rating: 5
The author put a lot of thought into the ease of accessibility and reading level of the target audience. There is even a "how to use this resource" section which could be extremely useful to students.
Consistency rating: 5
The text follows a very consistent format throughout the book.
Modularity rating: 5
Each case study is individual broken up and in a group of similar case studies. This makes it extremely easy to utilize.
Organization/Structure/Flow rating: 5
The book is very organized and the appendix is through. It flows seamlessly through each case study.
Interface rating: 5
I had no issues navigating this book, It was clearly labeled and very easy to move around in.
Grammatical Errors rating: 5
I did not catch any grammar errors as I was going through the book
Cultural Relevance rating: 5
This is a challenging question for any medical textbook. It is very culturally relevant to those in medical or medical office degrees.
I have been looking for something like this for years. I am so happy to have finally found it.
Reviewed by Cindy Sun, Assistant Professor, Marshall University on 1/7/23
Interestingly, this is not a case of ‘you get what you pay for’. Instead, not only are the case studies organized in a fashion for ease of use through a detailed table of contents, the authors have included more support for both faculty and... read more
Interestingly, this is not a case of ‘you get what you pay for’. Instead, not only are the case studies organized in a fashion for ease of use through a detailed table of contents, the authors have included more support for both faculty and students. For faculty, the introduction section titled ‘How to use this resource’ and individual notes to educators before each case study contain application tips. An appendix overview lists key elements as issues / concepts, scenario context, and healthcare roles for each case study. For students, learning objectives are presented at the beginning of each case study to provide a framework of expectations.
The content is presented accurately and realistic.
The case studies read similar to ‘A Day In the Life of…’ with detailed intraprofessional communications similar to what would be overheard in patient care areas. The authors present not only the view of the patient care nurse, but also weave interprofessional vantage points through each case study by including patient interaction with individual professionals such as radiology, physician, etc.
In addition to objective assessment findings, the authors integrate standard orders for each diagnosis including medications, treatments, and tests allowing the student to incorporate pathophysiology components to their assessments.
Each case study is arranged in the same framework for consistency and ease of use.
This compilation of eight healthcare case studies focusing on new onset and exacerbation of prevalent diagnoses, such as heart failure, deep vein thrombosis, cancer, and chronic obstructive pulmonary disease advancing to pneumonia.
Each case study has a photo of the ‘patient’. Simple as this may seem, it gives an immediate mental image for the student to focus.
Interface rating: 4
As noted by previous reviewers, most of the links do not connect active web pages. This may be due to the multiple options for accessing this resource (pdf download, pdf electronic, web view, etc.).
Grammatical Errors rating: 4
A minor weakness that faculty will probably need to address prior to use is regarding specific term usages differences between Commonwealth countries and United States, such as lung sound descriptors as ‘quiet’ in place of ‘diminished’ and ‘puffers’ in place of ‘inhalers’.
The authors have provided a multicultural, multigenerational approach in selection of patient characteristics representing a snapshot of today’s patient population. Additionally, one case study focusing on heart failure is about a middle-aged adult, contrasting to the average aged patient the students would normally see during clinical rotations. This option provides opportunities for students to expand their knowledge on risk factors extending beyond age.
This resource is applicable to nursing students learning to care for patients with the specific disease processes presented in each case study or for the leadership students focusing on intraprofessional communication. Educators can assign as a supplement to clinical experiences or as an in-class application of knowledge.
Reviewed by Stephanie Sideras, Assistant Professor, University of Portland on 8/15/22
The eight case studies included in this text addressed high frequency health alterations that all nurses need to be able to manage competently. While diabetes was not highlighted directly, it was included as a potential comorbidity. The five... read more
The eight case studies included in this text addressed high frequency health alterations that all nurses need to be able to manage competently. While diabetes was not highlighted directly, it was included as a potential comorbidity. The five overarching learning objectives pulled from the Institute of Medicine core competencies will clearly resonate with any faculty familiar with Quality and Safety Education for Nurses curriculum.
The presentation of symptoms, treatments and management of the health alterations was accurate. Dialogue between the the interprofessional team was realistic. At times the formatting of lab results was confusing as they reflected reference ranges specific to the Canadian healthcare system but these occurrences were minimal and could be easily adapted.
The focus for learning from these case studies was communication - patient centered communication and interprofessional team communication. Specific details, such as drug dosing, was minimized, which increases longevity and allows for easy individualization of the case data.
While some vocabulary was specific to the Canadian healthcare system, overall the narrative was extremely engaging and easy to follow. Subjective case data from patient or provider were formatted in italics and identified as 'thoughts'. Objective and behavioral case data were smoothly integrated into the narrative.
The consistency of formatting across the eight cases was remarkable. Specific learning objectives are identified for each case and these remain consistent across the range of cases, varying only in the focus for the goals for each different health alterations. Each case begins with presentation of essential patient background and the progress across the trajectory of illness as the patient moves from location to location encountering different healthcare professionals. Many of the characters (the triage nurse in the Emergency Department, the phlebotomist) are consistent across the case situations. These consistencies facilitate both application of a variety of teaching methods and student engagement with the situated learning approach.
Case data is presented by location and begins with the patient's first encounter with the healthcare system. This allows for an examination of how specific trajectories of illness are manifested and how care management needs to be prioritized at different stages. This approach supports discussions of care transitions and the complexity of the associated interprofessional communication.
The text is well organized. The case that has two levels of complexity is clearly identified
The internal links between the table of contents and case specific locations work consistently. In the EPUB and the Digital PDF the external hyperlinks are inconsistently valid.
The grammatical errors were minimal and did not detract from readability
Cultural diversity is present across the cases in factors including race, ethnicity, socioeconomic status, family dynamics and sexual orientation.
The level of detail included in these cases supports a teaching approach to address all three spectrums of learning - knowledge, skills and attitudes - necessary for the development of competent practice. I also appreciate the inclusion of specific assessment instruments that would facilitate a discussion of evidence based practice. I will enjoy using these case to promote clinical reasoning discussions of data that is noticed and interpreted with the resulting prioritizes that are set followed by reflections that result from learner choices.
Reviewed by Chris Roman, Associate Professor, Butler University on 5/19/22
It would be extremely difficult for a book of clinical cases to comprehensively cover all of medicine, and this text does not try. Rather, it provides cases related to common medical problems and introduces them in a way that allows for various... read more
Comprehensiveness rating: 4 see less
It would be extremely difficult for a book of clinical cases to comprehensively cover all of medicine, and this text does not try. Rather, it provides cases related to common medical problems and introduces them in a way that allows for various learning strategies to be employed to leverage the cases for deeper student learning and application.
The narrative form of the cases is less subject to issues of accuracy than a more content-based book would be. That said, the cases are realistic and reasonable, avoiding being too mundane or too extreme.
These cases are narrative and do not include many specific mentions of drugs, dosages, or other aspects of clinical care that may grow/evolve as guidelines change. For this reason, the cases should be “evergreen” and can be modified to suit different types of learners.
Clarity rating: 4
The text is written in very accessible language and avoids heavy use of technical language. Depending on the level of learner, this might even be too simplistic and omit some details that would be needed for physicians, pharmacists, and others to make nuanced care decisions.
The format is very consistent with clear labeling at transition points.
The authors point out in the introductory materials that this text is designed to be used in a modular fashion. Further, they have built in opportunities to customize each cases, such as giving dates of birth at “19xx” to allow for adjustments based on instructional objectives, etc.
The organization is very easy to follow.
I did not identify any issues in navigating the text.
The text contains no grammatical errors, though the language is a little stiff/unrealistic in some cases.
Cases involve patients and members of the care team that are of varying ages, genders, and racial/ethnic backgrounds
Reviewed by Trina Larery, Assistant Professor, Pittsburg State University on 4/5/22
The book covers common scenarios, providing allied health students insight into common health issues. The information in the book is thorough and easily modified if needed to include other scenarios not listed. The material was easy to understand... read more
The book covers common scenarios, providing allied health students insight into common health issues. The information in the book is thorough and easily modified if needed to include other scenarios not listed. The material was easy to understand and apply to the classroom. The E-reader format included hyperlinks that bring the students to subsequent clinical studies.
Content Accuracy rating: 4
The treatments were explained and rationales were given, which can be very helpful to facilitate effective learning for a nursing student or novice nurse. The case studies were accurate in explanation. The DVT case study incorrectly identifies the location of the clot in the popliteal artery instead of in the vein.
The content is relevant to a variety of different types of health care providers and due to the general nature of the cases, will remain relevant over time. Updates should be made annually to the hyperlinks and to assure current standard of practice is still being met.
Clear, simple and easy to read.
Consistent with healthcare terminology and framework throughout all eight case studies.
The text is modular. Cases can be used individually within a unit on the given disease process or relevant sections of a case could be used to illustrate a specific point providing great flexibility. The appendix is helpful in locating content specific to a certain diagnosis or a certain type of health care provider.
The book is well organized, presenting in a logical clear fashion. The appendix allows the student to move about the case study without difficulty.
The interface is easy and simple to navigate. Some links to external sources might need to be updated regularly since those links are subject to change based on current guidelines. A few hyperlinks had "page not found".
Few grammatical errors were noted in text.
The case studies include people of different ethnicities, socioeconomic status, ages, and genders to make this a very useful book.
I enjoyed reading the text. It was interesting and relevant to today's nursing student. There are roughly 25 broken online links or "pages not found", care needs to be taken to update at least annually and assure links are valid and utilizing the most up to date information.
Reviewed by Benjamin Silverberg, Associate Professor/Clinician, West Virginia University on 3/24/22
The appendix reviews the "key roles" and medical venues found in all 8 cases, but is fairly spartan on medical content. The table of contents at the beginning only lists the cases and locations of care. It can be a little tricky to figure out what... read more
Comprehensiveness rating: 3 see less
The appendix reviews the "key roles" and medical venues found in all 8 cases, but is fairly spartan on medical content. The table of contents at the beginning only lists the cases and locations of care. It can be a little tricky to figure out what is going on where, especially since each case is largely conversation-based. Since this presents 8 cases (really 7 with one being expanded upon), there are many medical topics (and venues) that are not included. It's impossible to include every kind of situation, but I'd love to see inclusion of sexual health, renal pathology, substance abuse, etc.
Though there are differences in how care can be delivered based on personal style, changing guidelines, available supplies, etc, the medical accuracy seems to be high. I did not detect bias or industry influence.
Relevance/Longevity rating: 4
Medications are generally listed as generics, with at least current dosing recommendations. The text gives a picture of what care looks like currently, but will be a little challenging to update based on new guidelines (ie, it can be hard to find the exact page in which a medication is dosed/prescribed). Even if the text were to be a little out of date, an instructor can use that to point out what has changed (and why).
Clear text, usually with definitions of medical slang or higher-tier vocabulary. Minimal jargon and there are instances where the "characters" are sorting out the meaning as well, making it accessible for new learners, too.
Overall, the style is consistent between cases - largely broken up into scenes and driven by conversation rather than descriptions of what is happening.
There are 8 (well, again, 7) cases which can be reviewed in any order. Case #2 builds upon #1, which is intentional and a good idea, though personally I would have preferred one case to have different possible outcomes or even a recurrence of illness. Each scene within a case is reasonably short.
Organization/Structure/Flow rating: 4
These cases are modular and don't really build on concepts throughout. As previously stated, case #2 builds upon #1, but beyond that, there is no progression. (To be sure, the authors suggest using case #1 for newer learners and #2 for more advanced ones.) The text would benefit from thematic grouping, a longer introduction and debriefing for each case (there are learning objectives but no real context in medical education nor questions to reflect on what was just read), and progressively-increasing difficulty in medical complexity, ethics, etc.
I used the PDF version and had no interface issues. There are minimal photographs and charts. Some words are marked in blue but those did not seem to be hyperlinked anywhere.
No noticeable errors in grammar, spelling, or formatting were noted.
I appreciate that some diversity of age and ethnicity were offered, but this could be improved. There were Canadian Indian and First Nations patients, for example, as well as other characters with implied diversity, but there didn't seem to be any mention of gender diverse or non-heterosexual people, or disabilities. The cases tried to paint family scenes (the first patient's dog was fairly prominently mentioned) to humanize them. Including more cases would allow for more opportunities to include sex/gender minorities, (hidden) disabilities, etc.
The text (originally from 2017) could use an update. It could be used in conjunction with other Open Texts, as a compliment to other coursework, or purely by itself. The focus is meant to be on improving communication, but there are only 3 short pages at the beginning of the text considering those issues (which are really just learning objectives). In addition to adding more cases and further diversity, I personally would love to see more discussion before and after the case to guide readers (and/or instructors). I also wonder if some of the ambiguity could be improved by suggesting possible health outcomes - this kind of counterfactual comparison isn't possible in real life and could be really interesting in a text. Addition of comprehension/discussion questions would also be worthwhile.
Reviewed by Danielle Peterson, Assistant Professor, University of Saint Francis on 12/31/21
This text provides readers with 8 case studies which include both chronic and acute healthcare issues. Although not comprehensive in regard to types of healthcare conditions, it provides a thorough look at the communication between healthcare... read more
This text provides readers with 8 case studies which include both chronic and acute healthcare issues. Although not comprehensive in regard to types of healthcare conditions, it provides a thorough look at the communication between healthcare workers in acute hospital settings. The cases are primarily set in the inpatient hospital setting, so the bulk of the clinical information is basic emergency care and inpatient protocol: vitals, breathing, medication management, etc. The text provides a table of contents at opening of the text and a handy appendix at the conclusion of the text that outlines each case’s issue(s), scenario, and healthcare roles. No index or glossary present.
Although easy to update, it should be noted that the cases are taking place in a Canadian healthcare system. Terms may be unfamiliar to some students including “province,” “operating theatre,” “physio/physiotherapy,” and “porter.” Units of measurement used include Celsius and meters. Also, the issue of managed care, health insurance coverage, and length of stay is missing for American students. These are primary issues that dictate much of the healthcare system in the US and a primary job function of social workers, nurse case managers, and medical professionals in general. However, instructors that wish to add this to the case studies could do so easily.
The focus of this text is on healthcare communication which makes it less likely to become obsolete. Much of the clinical information is stable healthcare practice that has been standard of care for quite some time. Nevertheless, given the nature of text, updates would be easy to make. Hyperlinks should be updated to the most relevant and trustworthy sources and checked frequently for effectiveness.
The spacing that was used to note change of speaker made for ease of reading. Although unembellished and plain, I expect students to find this format easy to digest and interesting, especially since the script is appropriately balanced with ‘human’ qualities like the current TV shows and songs, the use of humor, and nonverbal cues.
A welcome characteristic of this text is its consistency. Each case is presented in a similar fashion and the roles of the healthcare team are ‘played’ by the same character in each of the scenarios. This allows students to see how healthcare providers prioritize cases and juggle the needs of multiple patients at once. Across scenarios, there was inconsistency in when clinical terms were hyperlinked.
The text is easily divisible into smaller reading sections. However, since the nature of the text is script-narrative format, if significant reorganization occurs, one will need to make sure that the communication of the script still makes sense.
The text is straightforward and presented in a consistent fashion: learning objectives, case history, a script of what happened before the patient enters the healthcare setting, and a script of what happens once the patient arrives at the healthcare setting. The authors use the term, “ideal interactions,” and I would agree that these cases are in large part, ‘best case scenarios.’ Due to this, the case studies are well organized, clear, logical, and predictable. However, depending on the level of student, instructors may want to introduce complications that are typical in the hospital setting.
The interface is pleasing and straightforward. With exception to the case summary and learning objectives, the cases are in narrative, script format. Each case study supplies a photo of the ‘patient’ and one of the case studies includes a link to a 3-minute video that introduces the reader to the patient/case. One of the highlights of this text is the use of hyperlinks to various clinical practices (ABG, vital signs, transfer of patient). Unfortunately, a majority of the links are broken. However, since this is an open text, instructors can update the links to their preference.
Although not free from grammatical errors, those that were noticed were minimal and did not detract from reading.
Cultural Relevance rating: 4
Cultural diversity is visible throughout the patients used in the case studies and includes factors such as age, race, socioeconomic status, family dynamics, and sexual orientation. A moderate level of diversity is noted in the healthcare team with some stereotypes: social workers being female, doctors primarily male.
As a social work instructor, I was grateful to find a text that incorporates this important healthcare role. I would have liked to have seen more content related to advance directives, mediating decision making between the patient and care team, emotional and practical support related to initial diagnosis and discharge planning, and provision of support to colleagues, all typical roles of a medical social worker. I also found it interesting that even though social work was included in multiple scenarios, the role was only introduced on the learning objectives page for the oncology case.
Reviewed by Crystal Wynn, Associate Professor, Virginia State University on 7/21/21
The text covers a variety of chronic diseases within the cases; however, not all of the common disease states were included within the text. More chronic diseases need to be included such as diabetes, cancer, and renal failure. Not all allied... read more
The text covers a variety of chronic diseases within the cases; however, not all of the common disease states were included within the text. More chronic diseases need to be included such as diabetes, cancer, and renal failure. Not all allied health care team members are represented within the case study. Key terms appear throughout the case study textbook and readers are able to click on a hyperlink which directs them to the definition and an explanation of the key term.
Content is accurate, error-free and unbiased.
The content is up-to-date, but not in a way that will quickly make the text obsolete within a short period of time. The text is written and/or arranged in such a way that necessary updates will be relatively easy and straightforward to implement.
The text is written in lucid, accessible prose, and provides adequate context for any jargon/technical terminology used
The text is internally consistent in terms of terminology and framework.
The text is easily and readily divisible into smaller reading sections that can be assigned at different points within the course. Each case can be divided into a chronic disease state unit, which will allow the reader to focus on one section at a time.
Organization/Structure/Flow rating: 3
The topics in the text are presented in a logical manner. Each case provides an excessive amount of language that provides a description of the case. The cases in this text reads more like a novel versus a clinical textbook. The learning objectives listed within each case should be in the form of questions or activities that could be provided as resources for instructors and teachers.
Interface rating: 3
There are several hyperlinks embedded within the textbook that are not functional.
The text contains no grammatical errors.
Cultural Relevance rating: 3
The text is not culturally insensitive or offensive in any way. More examples of cultural inclusiveness is needed throughout the textbook. The cases should be indicative of individuals from a variety of races and ethnicities.
Reviewed by Rebecca Hillary, Biology Instructor, Portland Community College on 6/15/21
This textbook consists of a collection of clinical case studies that can be applicable to a wide range of learning environments from supplementing an undergraduate Anatomy and Physiology Course, to including as part of a Medical or other health... read more
This textbook consists of a collection of clinical case studies that can be applicable to a wide range of learning environments from supplementing an undergraduate Anatomy and Physiology Course, to including as part of a Medical or other health care program. I read the textbook in E-reader format and this includes hyperlinks that bring the students to subsequent clinical study if the book is being used in a clinical classroom. This book is significantly more comprehensive in its approach from other case studies I have read because it provides a bird’s eye view of the many clinicians, technicians, and hospital staff working with one patient. The book also provides real time measurements for patients that change as they travel throughout the hospital until time of discharge.
Each case gave an accurate sense of the chaos that would be present in an emergency situation and show how the conditions affect the practitioners as well as the patients. The reader gets an accurate big picture--a feel for each practitioner’s point of view as well as the point of view of the patient and the patient’s family as the clock ticks down and the patients are subjected to a number of procedures. The clinical information contained in this textbook is all in hyperlinks containing references to clinical skills open text sources or medical websites. I did find one broken link on an external medical resource.
The diseases presented are relevant and will remain so. Some of the links are directly related to the Canadian Medical system so they may not be applicable to those living in other regions. Clinical links may change over time but the text itself will remain relevant.
Each case study clearly presents clinical data as is it recorded in real time.
Each case study provides the point of view of several practitioners and the patient over several days. While each of the case studies covers different pathology they all follow this same format, several points of view and data points, over a number of days.
The case studies are divided by days and this was easy to navigate as a reader. It would be easy to assign one case study per body system in an Anatomy and Physiology course, or to divide them up into small segments for small in class teaching moments.
The topics are presented in an organized way showing clinical data over time and each case presents a large number of view points. For example, in the first case study, the patient is experiencing difficulty breathing. We follow her through several days from her entrance to the emergency room. We meet her X Ray Technicians, Doctor, Nurses, Medical Assistant, Porter, Physiotherapist, Respiratory therapist, and the Lab Technicians running her tests during her stay. Each practitioner paints the overall clinical picture to the reader.
I found the text easy to navigate. There were not any figures included in the text, only clinical data organized in charts. The figures were all accessible via hyperlink. Some figures within the textbook illustrating patient scans could have been helpful but I did not have trouble navigating the links to visualize the scans.
I did not see any grammatical errors in the text.
The patients in the text are a variety of ages and have a variety of family arrangements but there is not much diversity among the patients. Our seven patients in the eight case studies are mostly white and all cis gendered.
Some of the case studies, for example the heart failure study, show clinical data before and after drug treatments so the students can get a feel for mechanism in physiological action. I also liked that the case studies included diet and lifestyle advice for the patients rather than solely emphasizing these pharmacological interventions. Overall, I enjoyed reading through these case studies and I plan to utilize them in my Anatomy and Physiology courses.
Reviewed by Richard Tarpey, Assistant Professor, Middle Tennessee State University on 5/11/21
As a case study book, there is no index or glossary. However, medical and technical terms provide a useful link to definitions and explanations that will prove useful to students unfamiliar with the terms. The information provided is appropriate... read more
As a case study book, there is no index or glossary. However, medical and technical terms provide a useful link to definitions and explanations that will prove useful to students unfamiliar with the terms. The information provided is appropriate for entry-level health care students. The book includes important health problems, but I would like to see coverage of at least one more chronic/lifestyle issue such as diabetes. The book covers adult issues only.
Content is accurate without bias
The content of the book is relevant and up-to-date. It addresses conditions that are prevalent in today's population among adults. There are no pediatric cases, but this does not significantly detract from the usefulness of the text. The format of the book lends to easy updating of data or information.
The book is written with clarity and is easy to read. The writing style is accessible and technical terminology is explained with links to more information.
Consistency is present. Lack of consistency is typically a problem with case study texts, but this book is consistent with presentation, format, and terminology throughout each of the eight cases.
The book has high modularity. Each of the case studies can be used independently from the others providing flexibility. Additionally, each case study can be partitioned for specific learning objectives based on the learning objectives of the course or module.
The book is well organized, presenting students conceptually with differing patient flow patterns through a hospital. The patient information provided at the beginning of each case is a wonderful mechanism for providing personal context for the students as they consider the issues. Many case studies focus on the problem and the organization without students getting a patient's perspective. The patient perspective is well represented in these cases.
The navigation through the cases is good. There are some terminology and procedure hyperlinks within the cases that do not work when accessed. This is troubling if you intend to use the text for entry-level health care students since many of these links are critical for a full understanding of the case.
There are some non-US variants of spelling and a few grammatical errors, but these do not detract from the content of the messages of each case.
The book is inclusive of differing backgrounds and perspectives. No insensitive or offensive references were found.
I like this text for its application flexibility. The book is useful for non-clinical healthcare management students to introduce various healthcare-related concepts and terminology. The content is also helpful for the identification of healthcare administration managerial issues for students to consider. The book has many applications.
Reviewed by Paula Baldwin, Associate Professor/Communication Studies, Western Oregon University on 5/10/21
The different case studies fall on a range, from crisis care to chronic illness care. read more
The different case studies fall on a range, from crisis care to chronic illness care.
The contents seems to be written as they occurred to represent the most complete picture of each medical event's occurence.
These case studies are from the Canadian medical system, but that does not interfere with it's applicability.
It is written for a medical audience, so the terminology is mostly formal and technical.
Some cases are shorter than others and some go in more depth, but it is not problematic.
The eight separate case studies is the perfect size for a class in the quarter system. You could combine this with other texts, videos or learning modalities, or use it alone.
As this is a case studies book, there is not a need for a logical progression in presentation of topics.
No problems in terms of interface.
I have not seen any grammatical errors.
I did not see anything that was culturally insensitive.
I used this in a Health Communication class and it has been extraordinarily successful. My studies are analyzing the messaging for the good, the bad, and the questionable. The case studies are widely varied and it gives the class insights into hospital experiences, both front and back stage, that they would not normally be able to examine. I believe that because it is based real-life medical incidents, my students are finding the material highly engaging.
Reviewed by Marlena Isaac, Instructor, Aiken Technical College on 4/23/21
This text is great to walk through patient care with entry level healthcare students. The students are able to take in the information, digest it, then provide suggestions to how they would facilitate patient healing. Then when they are faced with... read more
This text is great to walk through patient care with entry level healthcare students. The students are able to take in the information, digest it, then provide suggestions to how they would facilitate patient healing. Then when they are faced with a situation in clinical they are not surprised and now how to move through it effectively.
The case studies provided accurate information that relates to the named disease.
It is relevant to health care studies and the development of critical thinking.
Cases are straightforward with great clinical information.
Clinical information is provided concisely.
Appropriate for clinical case study.
Presented to facilitate information gathering.
Takes a while to navigate in the browser.
Cultural Relevance rating: 1
Text lacks adequate representation of minorities.
Reviewed by Kim Garcia, Lecturer III, University of Texas Rio Grande Valley on 11/16/20
The book has 8 case studies, so obviously does not cover the whole of medicine, but the cases provided are descriptive and well developed. Cases are presented at different levels of difficulty, making the cases appropriate for students at... read more
The book has 8 case studies, so obviously does not cover the whole of medicine, but the cases provided are descriptive and well developed. Cases are presented at different levels of difficulty, making the cases appropriate for students at different levels of clinical knowledge. The human element of both patient and health care provider is well captured. The cases are presented with a focus on interprofessional interaction and collaboration, more so than teaching medical content.
Content is accurate and un-biased. No errors noted. Most diagnostic and treatment information is general so it will remain relevant over time. The content of these cases is more appropriate for teaching interprofessional collaboration and less so for teaching the medical care for each diagnosis.
The content is relevant to a variety of different types of health care providers (nurses, radiologic technicians, medical laboratory personnel, etc) and due to the general nature of the cases, will remain relevant over time.
Easy to read. Clear headings are provided for sections of each case study and these section headings clearly tell when time has passed or setting has changed. Enough description is provided to help set the scene for each part of the case. Much of the text is written in the form of dialogue involving patient, family and health care providers, making it easy to adapt for role play. Medical jargon is limited and links for medical terms are provided to other resources that expound on medical terms used.
The text is consistent in structure of each case. Learning objectives are provided. Cases generally start with the patient at home and move with the patient through admission, testing and treatment, using a variety of healthcare services and encountering a variety of personnel.
The text is modular. Cases could be used individually within a unit on the given disease process or relevant sections of a case could be used to illustrate a specific point. The appendix is helpful in locating content specific to a certain diagnosis or a certain type of health care provider.
Each case follows a patient in a logical, chronologic fashion. A clear table of contents and appendix are provided which allows the user to quickly locate desired content. It would be helpful if the items in the table of contents and appendix were linked to the corresponding section of the text.
The hyperlinks to content outside this book work, however using the back arrow on your browser returns you to the front page of the book instead of to the point at which you left the text. I would prefer it if the hyperlinks opened in a new window or tab so closing that window or tab would leave you back where you left the text.
No grammatical errors were noted.
The text is culturally inclusive and appropriate. Characters, both patients and care givers are of a variety of races, ethnicities, ages and backgrounds.
I enjoyed reading the cases and reviewing this text. I can think of several ways in which I will use this content.
Reviewed by Raihan Khan, Instructor/Assistant Professor, James Madison University on 11/3/20
The book contains several important health issues, however still missing some chronic health issues that the students should learn before they join the workforce, such as diabetes-related health issues suffered by the patients. read more
The book contains several important health issues, however still missing some chronic health issues that the students should learn before they join the workforce, such as diabetes-related health issues suffered by the patients.
The health information contained in the textbook is mostly accurate.
I think the book is written focusing on the current culture and health issues faced by the patients. To keep the book relevant in the future, the contexts especially the culture/lifestyle/health care modalities, etc. would need to be updated regularly.
The language is pretty simple, clear, and easy to read.
There is no complaint about consistency. One of the main issues of writing a book, consistency was well managed by the authors.
The book is easy to explore based on how easy the setup is. Students can browse to the specific section that they want to read without much hassle of finding the correct information.
The organization is simple but effective. The authors organized the book based on what can happen in a patient's life and what possible scenarios students should learn about the disease. From that perspective, the book does a good job.
The interface is easy and simple to navigate. Some links to external sources might need to be updated regularly since those links are subject to change that is beyond the author's control. It's frustrating for the reader when the external link shows no information.
The book is free of any major language and grammatical errors.
The book might do a little better in cultural competency. e.g. Last name Singh is mainly for Sikh people. In the text Harj and Priya Singh are Muslim. the authors can consult colleagues who are more familiar with those cultures and revise some cultural aspects of the cases mentioned in the book.
The book is a nice addition to the open textbook world. Hope to see more health issues covered by the book.
Reviewed by Ryan Sheryl, Assistant Professor, California State University, Dominguez Hills on 7/16/20
This text contains 8 medical case studies that reflect best practices at the time of publication. The text identifies 5 overarching learning objectives: interprofessional collaboration, client centered care, evidence-based practice, quality... read more
This text contains 8 medical case studies that reflect best practices at the time of publication. The text identifies 5 overarching learning objectives: interprofessional collaboration, client centered care, evidence-based practice, quality improvement, and informatics. While the case studies do not cover all medical conditions or bodily systems, the book is thorough in conveying details of various patients and medical team members in a hospital environment. Rather than an index or glossary at the end of the text, it contains links to outside websites for more information on medical tests and terms referenced in the cases.
The content provided is reflective of best practices in patient care, interdisciplinary collaboration, and communication at the time of publication. It is specifically accurate for the context of hospitals in Canada. The links provided throughout the text have the potential to supplement with up-to-date descriptions and definitions, however, many of them are broken (see notes in Interface section).
The content of the case studies reflects the increasingly complex landscape of healthcare, including a variety of conditions, ages, and personal situations of the clients and care providers. The text will require frequent updating due to the rapidly changing landscape of society and best practices in client care. For example, a future version may include inclusive practices with transgender clients, or address ways medical racism implicitly impacts client care (see notes in Cultural Relevance section).
The text is written clearly and presents thorough, realistic details about working and being treated in an acute hospital context.
The text is very straightforward. It is consistent in its structure and flow. It uses consistent terminology and follows a structured framework throughout.
Being a series of 8 separate case studies, this text is easily and readily divisible into smaller sections. The text was designed to be taken apart and used piece by piece in order to serve various learning contexts. The parts of each case study can also be used independently of each other to facilitate problem solving.
The topics in the case studies are presented clearly. The structure of each of the case studies proceeds in a similar fashion. All of the cases are set within the same hospital so the hospital personnel and service providers reappear across the cases, giving a textured portrayal of the experiences of the various service providers. The cases can be used individually, or one service provider can be studied across the various studies.
The text is very straightforward, without complex charts or images that could become distorted. Many of the embedded links are broken and require updating. The links that do work are a very useful way to define and expand upon medical terms used in the case studies.
Grammatical errors are minimal and do not distract from the flow of the text. In one instance the last name Singh is spelled Sing, and one patient named Fred in the text is referred to as Frank in the appendix.
The cases all show examples of health care personnel providing compassionate, client-centered care, and there is no overt discrimination portrayed. Two of the clients are in same-sex marriages and these are shown positively. It is notable, however, that the two cases presenting people of color contain more negative characteristics than the other six cases portraying Caucasian people. The people of color are the only two examples of clients who smoke regularly. In addition, the Indian client drinks and is overweight, while the First Nations client is the only one in the text to have a terminal diagnosis. The Indian client is identified as being Punjabi and attending a mosque, although there are only 2% Muslims in the Punjab province of India. Also, the last name Singh generally indicates a person who is a Hindu or Sikh, not Muslim.
Reviewed by Monica LeJeune, RN Instructor, LSUE on 4/24/20
Has comprehensive unfolding case studies that guide the reader to recognize and manage the scenario presented. Assists in critical thinking process. read more
Has comprehensive unfolding case studies that guide the reader to recognize and manage the scenario presented. Assists in critical thinking process.
Accurately presents health scenarios with real life assessment techniques and patient outcomes.
Relevant to nursing practice.
Clearly written and easily understood.
Consistent with healthcare terminology and framework
Has a good reading flow.
Topics presented in logical fashion
Easy to read.
No grammatical errors noted.
Text is not culturally insensitive or offensive.
Good book to have to teach nursing students.
Reviewed by april jarrell, associate professor, J. Sargeant Reynolds Community College on 1/7/20
The text is a great case study tool that is appropriate for nursing school instructors to use in aiding students to learn the nursing process. read more
The text is a great case study tool that is appropriate for nursing school instructors to use in aiding students to learn the nursing process.
The content is accurate and evidence based. There is no bias noted
The content in the text is relevant, up to date for nursing students. It will be easy to update content as needed because the framework allows for addition to the content.
The text is clear and easy to understand.
Framework and terminology is consistent throughout the text; the case study is a continual and takes the student on a journey with the patient. Great for learning!
The case studies can be easily divided into smaller sections to allow for discussions, and weekly studies.
The text and content progress in a logical, clear fashion allowing for progression of learning.
No interface issues noted with this text.
No grammatical errors noted in the text.
No racial or culture insensitivity were noted in the text.
I would recommend this text be used in nursing schools. The use of case studies are helpful for students to learn and practice the nursing process.
Reviewed by Lisa Underwood, Practical Nursing Instructor, NTCC on 12/3/19
The text provides eight comprehensive case studies that showcase the different viewpoints of the many roles involved in patient care. It encompasses the most common seen diagnoses seen across healthcare today. Each case study comes with its own... read more
The text provides eight comprehensive case studies that showcase the different viewpoints of the many roles involved in patient care. It encompasses the most common seen diagnoses seen across healthcare today. Each case study comes with its own set of learning objectives that can be tweaked to fit several allied health courses. Although the case studies are designed around the Canadian Healthcare System, they are quite easily adaptable to fit most any modern, developed healthcare system.
Content Accuracy rating: 3
Overall, the text is quite accurate. There is one significant error that needs to be addressed. It is located in the DVT case study. In the study, a popliteal artery clot is mislabeled as a DVT. DVTs are located in veins, not in arteries. That said, the case study on the whole is quite good. This case study could be used as a learning tool in the classroom for discussion purposes or as a way to test student understanding of DVTs, on example might be, "Can they spot the error?"
At this time, all of the case studies within the text are current. Healthcare is an ever evolving field that rests on the best evidence based practice. Keeping that in mind, educators can easily adapt the studies as the newest evidence emerges and changes practice in healthcare.
All of the case studies are well written and easy to understand. The text includes several hyperlinks and it also highlights certain medical terminology to prompt readers as a way to enhance their learning experience.
Across the text, the language, style, and format of the case studies are completely consistent.
The text is divided into eight separate case studies. Each case study may be used independently of the others. All case studies are further broken down as the focus patient passes through each aspect of their healthcare system. The text's modularity makes it possible to use a case study as individual work, group projects, class discussions, homework or in a simulation lab.
The case studies and the diagnoses that they cover are presented in such a way that educators and allied health students can easily follow and comprehend.
The book in itself is free of any image distortion and it prints nicely. The text is offered in a variety of digital formats. As noted in the above reviews, some of the hyperlinks have navigational issues. When the reader attempts to access them, a "page not found" message is received.
There were minimal grammatical errors. Some of which may be traced back to the differences in our spelling.
The text is culturally relevant in that it includes patients from many different backgrounds and ethnicities. This allows educators and students to explore cultural relevance and sensitivity needs across all areas in healthcare. I do not believe that the text was in any way insensitive or offensive to the reader.
By using the case studies, it may be possible to have an open dialogue about the differences noted in healthcare systems. Students will have the ability to compare and contrast the Canadian healthcare system with their own. I also firmly believe that by using these case studies, students can improve their critical thinking skills. These case studies help them to "put it all together".
Reviewed by Melanie McGrath, Associate Professor, TRAILS on 11/29/19
The text covered some of the most common conditions seen by healthcare providers in a hospital setting, which forms a solid general base for the discussions based on each case. read more
The text covered some of the most common conditions seen by healthcare providers in a hospital setting, which forms a solid general base for the discussions based on each case.
I saw no areas of inaccuracy
As in all healthcare texts, treatments and/or tests will change frequently. However, everything is currently up-to-date thus it should be a good reference for several years.
Each case is written so that any level of healthcare student would understand. Hyperlinks in the text is also very helpful.
All of the cases are written in a similar fashion.
Although not structured as a typical text, each case is easily assigned as a stand-alone.
Each case is organized clearly in an appropriate manner.
I did not see any issues.
I did not see any grammatical errors
The text seemed appropriately inclusive. There are no pediatric cases and no cases of intellectually-impaired patients, but those types of cases introduce more advanced problem-solving which perhaps exceed the scope of the text. May be a good addition to the text.
I found this text to be an excellent resource for healthcare students in a variety of fields. It would be best utilized in inter professional courses to help guide discussion.
Reviewed by Lynne Umbarger, Clinical Assistant Professor, Occupational Therapy, Emory and Henry College on 11/26/19
While the book does not cover every scenario, the ones in the book are quite common and troublesome for inexperienced allied health students. The information in the book is thorough enough, and I have found the cases easy to modify for educational... read more
While the book does not cover every scenario, the ones in the book are quite common and troublesome for inexperienced allied health students. The information in the book is thorough enough, and I have found the cases easy to modify for educational purposes. The material was easily understood by the students but challenging enough for classroom discussion. There are no mentions in the book about occupational therapy, but it is easy enough to add a couple words and make inclusion simple.
Very nice lab values are provided in the case study, making it more realistic for students.
These case studies focus on commonly encountered diagnoses for allied health and nursing students. They are comprehensive, realistic, and easily understood. The only difference is that the hospital in one case allows the patient's dog to visit in the room (highly unusual in US hospitals).
The material is easily understood by allied health students. The cases have links to additional learning materials for concepts that may be less familiar or should be explored further in a particular health field.
The language used in the book is consistent between cases. The framework is the same with each case which makes it easier to locate areas that would be of interest to a particular allied health profession.
The case studies are comprehensive but well-organized. They are short enough to be useful for class discussion or a full-blown assignment. The students seem to understand the material and have not expressed that any concepts or details were missing.
Each case is set up like the other cases. There are learning objectives at the beginning of each case to facilitate using the case, and it is easy enough to pull out material to develop useful activities and assignments.
There is a quick chart in the Appendix to allow the reader to determine the professions involved in each case as well as the pertinent settings and diagnoses for each case study. The contents are easy to access even while reading the book.
As a person who attends carefully to grammar, I found no errors in all of the material I read in this book.
There are a greater number of people of different ethnicities, socioeconomic status, ages, and genders to make this a very useful book. With each case, I could easily picture the person in the case. This book appears to be Canadian and more inclusive than most American books.
I was able to use this book the first time I accessed it to develop a classroom activity for first-year occupational therapy students and a more comprehensive activity for second-year students. I really appreciate the links to a multitude of terminology and medical lab values/issues for each case. I will keep using this book.
Reviewed by Cindy Krentz, Assistant Professor, Metropolitan State University of Denver on 6/15/19
The book covers eight case studies of common inpatient or emergency department scenarios. I appreciated that they had written out the learning objectives. I liked that the patient was described before the case was started, giving some... read more
The book covers eight case studies of common inpatient or emergency department scenarios. I appreciated that they had written out the learning objectives. I liked that the patient was described before the case was started, giving some understanding of the patient's background. I think it could benefit from having a glossary. I liked how the authors included the vital signs in an easily readable bar. I would have liked to see the labs also highlighted like this. I also felt that it would have been good written in a 'what would you do next?' type of case study.
The book is very accurate in language, what tests would be prudent to run and in the day in the life of the hospital in all cases. One inaccuracy is that the authors called a popliteal artery clot a DVT. The rest of the DVT case study was great, though, but the one mistake should be changed.
The book is up to date for now, but as tests become obsolete and new equipment is routinely used, the book ( like any other health textbook) will need to be updated. It would be easy to change, however. All that would have to happen is that the authors go in and change out the test to whatever newer, evidence-based test is being utilized.
The text is written clearly and easy to understand from a student's perspective. There is not too much technical jargon, and it is pretty universal when used- for example DVT for Deep Vein Thrombosis.
The book is consistent in language and how it is broken down into case studies. The same format is used for highlighting vital signs throughout the different case studies. It's great that the reader does not have to read the book in a linear fashion. Each case study can be read without needing to read the others.
The text is broken down into eight case studies, and within the case studies is broken down into days. It is consistent and shows how the patient can pass through the different hospital departments (from the ER to the unit, to surgery, to home) in a realistic manner. The instructor could use one or more of the case studies as (s)he sees fit.
The topics are eight different case studies- and are presented very clearly and organized well. Each one is broken down into how the patient goes through the system. The text is easy to follow and logical.
The interface has some problems with the highlighted blue links. Some of them did not work and I got a 'page not found' message. That can be frustrating for the reader. I'm wondering if a glossary could be utilized (instead of the links) to explain what some of these links are supposed to explain.
I found two or three typos, I don't think they were grammatical errors. In one case I think the Canadian spelling and the United States spelling of the word are just different.
This is a very culturally competent book. In today's world, however, one more type of background that would merit delving into is the trans-gender, GLBTQI person. I was glad that there were no stereotypes.
I enjoyed reading the text. It was interesting and relevant to today's nursing student. Since we are becoming more interprofessional, I liked that we saw what the phlebotomist and other ancillary personnel (mostly different technicians) did. I think that it could become even more interdisciplinary so colleges and universities could have more interprofessional education- courses or simulations- with the addition of the nurse using social work, nutrition, or other professional health care majors.
Reviewed by Catherine J. Grott, Interim Director, Health Administration Program, TRAILS on 5/5/19
The book is comprehensive but is specifically written for healthcare workers practicing in Canada. The title of the book should reflect this. read more
The book is comprehensive but is specifically written for healthcare workers practicing in Canada. The title of the book should reflect this.
The book is accurate, however it has numerous broken online links.
Relevance/Longevity rating: 3
The content is very relevant, but some links are out-dated. For example, WHO Guidelines for Safe Surgery 2009 (p. 186) should be updated.
The book is written in clear and concise language. The side stories about the healthcare workers make the text interesting.
The book is consistent in terms of terminology and framework. Some terms that are emphasized in one case study are not emphasized (with online links) in the other case studies. All of the case studies should have the same words linked to online definitions.
Modularity rating: 3
The book can easily be parsed out if necessary. However, the way the case studies have been written, it's evident that different authors contributed singularly to each case study.
The organization and flow are good.
Interface rating: 1
There are numerous broken online links and "pages not found."
The grammar and punctuation are correct. There are two errors detected: p. 120 a space between the word "heart" and the comma; also a period is needed after Dr (p. 113).
I'm not quite sure that the social worker (p. 119) should comment that the patient and partner are "very normal people."
There are roughly 25 broken online links or "pages not found." The BC & Canadian Guidelines (p. 198) could also include a link to US guidelines to make the text more universal . The basilar crackles (p. 166) is very good. Text could be used compare US and Canadian healthcare. Text could be enhanced to teach "soft skills" and interdepartmental communication skills in healthcare.
Reviewed by Lindsey Henry, Practical Nursing Instructor, Fletcher on 5/1/19
I really appreciated how in the introduction, five learning objectives were identified for students. These objectives are paramount in nursing care and they are each spelled out for the learner. Each Case study also has its own learning... read more
I really appreciated how in the introduction, five learning objectives were identified for students. These objectives are paramount in nursing care and they are each spelled out for the learner. Each Case study also has its own learning objectives, which were effectively met in the readings.
As a seasoned nurse, I believe that the content regarding pathophysiology and treatments used in the case studies were accurate. I really appreciated how many of the treatments were also explained and rationales were given, which can be very helpful to facilitate effective learning for a nursing student or novice nurse.
The case studies are up to date and correlate with the current time period. They are easily understood.
I really loved how several important medical terms, including specific treatments were highlighted to alert the reader. Many interventions performed were also explained further, which is great to enhance learning for the nursing student or novice nurse. Also, with each scenario, a background and history of the patient is depicted, as well as the perspectives of the patient, patients family member, and the primary nurse. This really helps to give the reader a full picture of the day in the life of a nurse or a patient, and also better facilitates the learning process of the reader.
These case studies are consistent. They begin with report, the patient background or updates on subsequent days, and follow the patients all the way through discharge. Once again, I really appreciate how this book describes most if not all aspects of patient care on a day to day basis.
Each case study is separated into days. While they can be divided to be assigned at different points within the course, they also build on each other. They show trends in vital signs, what happens when a patient deteriorates, what happens when they get better and go home. Showing the entire process from ER admit to discharge is really helpful to enhance the students learning experience.
The topics are all presented very similarly and very clearly. The way that the scenarios are explained could even be understood by a non-nursing student as well. The case studies are very clear and very thorough.
The book is very easy to navigate, prints well on paper, and is not distorted or confusing.
I did not see any grammatical errors.
Each case study involves a different type of patient. These differences include race, gender, sexual orientation and medical backgrounds. I do not feel the text was offensive to the reader.
I teach practical nursing students and after reading this book, I am looking forward to implementing it in my classroom. Great read for nursing students!
Reviewed by Leah Jolly, Instructor, Clinical Coordinator, Oregon Institute of Technology on 4/10/19
Good variety of cases and pathologies covered. read more
Good variety of cases and pathologies covered.
Content Accuracy rating: 2
Some examples and scenarios are not completely accurate. For example in the DVT case, the sonographer found thrombus in the "popliteal artery", which according to the book indicated presence of DVT. However in DVT, thrombus is located in the vein, not the artery. The patient would also have much different symptoms if located in the artery. Perhaps some of these inaccuracies are just typos, but in real-life situations this simple mistake can make a world of difference in the patient's course of treatment and outcomes.
Good examples of interprofessional collaboration. If only it worked this way on an every day basis!
Clear and easy to read for those with knowledge of medical terminology.
Good consistency overall.
Broken up well.
Topics are clear and logical.
Would be nice to simply click through to the next page, rather than going through the table of contents each time.
Minor typos/grammatical errors.
No offensive or insensitive materials observed.
Reviewed by Alex Sargsyan, Doctor of Nursing Practice/Assistant Professor , East Tennessee State University on 10/8/18
Because of the case study character of the book it does not have index or glossary. However it has summary for each health case study outlining key elements discussed in each case study. read more
Because of the case study character of the book it does not have index or glossary. However it has summary for each health case study outlining key elements discussed in each case study.
Overall the book is accurately depicting the clinical environment. There are numerous references to external sites. While most of them are correct, some of them are not working. For example Homan’s test link is not working "404 error"
Book is relevant in its current version and can be used in undergraduate and graduate classes. That said, the longevity of the book may be limited because of the character of the clinical education. Clinical guidelines change constantly and it may require a major update of the content.
Cases are written very clearly and have realistic description of an inpatient setting.
The book is easy to read and consistent in the language in all eight cases.
The cases are very well written. Each case is subdivided into logical segments. The segments reflect different setting where the patient is being seen. There is a flow and transition between the settings.
Book has eight distinct cases. This is a great format for a book that presents distinct clinical issues. This will allow the students to have immersive experiences and gain better understanding of the healthcare environment.
Book is offered in many different formats. Besides the issues with the links mentioned above, overall navigation of the book content is very smooth.
Book is very well written and has no grammatical errors.
Book is culturally relevant. Patients in the case studies come different cultures and represent diverse ethnicities.
Reviewed by Justin Berry, Physical Therapist Assistant Program Director, Northland Community and Technical College, East Grand Forks, MN on 8/2/18
This text provides eight patient case studies from a variety of diagnoses, which can be utilized by healthcare students from multiple disciplines. The cases are comprehensive and can be helpful for students to determine professional roles,... read more
This text provides eight patient case studies from a variety of diagnoses, which can be utilized by healthcare students from multiple disciplines. The cases are comprehensive and can be helpful for students to determine professional roles, interprofessional roles, when to initiate communication with other healthcare practitioners due to a change in patient status, and treatment ideas. Some additional patient information, such as lab values, would have been beneficial to include.
Case study information is accurate and unbiased.
Content is up to date. The case studies are written in a way so that they will not be obsolete soon, even with changes in healthcare.
The case studies are well written, and can be utilized for a variety of classroom assignments, discussions, and projects. Some additional lab value information for each patient would have been a nice addition.
The case studies are consistently organized to make it easy for the reader to determine the framework.
The text is broken up into eight different case studies for various patient diagnoses. This design makes it highly modular, and would be easy to assign at different points of a course.
The flow of the topics are presented consistently in a logical manner. Each case study follows a patient chronologically, making it easy to determine changes in patient status and treatment options.
The text is free of interface issues, with no distortion of images or charts.
The text is not culturally insensitive or offensive in any way. Patients are represented from a variety of races, ethnicities, and backgrounds
This book would be a good addition for many different health programs.
Reviewed by Ann Bell-Pfeifer, Instructor/Program Director, Minnesota State Community and Technical College on 5/21/18
The book gives a comprehensive overview of many types of cases for patient conditions. Emergency Room patients may arrive with COPD, heart failure, sepsis, pneumonia, or as motor vehicle accident victims. It is directed towards nurses, medical... read more
The book gives a comprehensive overview of many types of cases for patient conditions. Emergency Room patients may arrive with COPD, heart failure, sepsis, pneumonia, or as motor vehicle accident victims. It is directed towards nurses, medical laboratory technologists, medical radiology technologists, and respiratory therapists and their roles in caring for patients. Most of the overview is accurate. One suggestion is to provide an embedded radiologist interpretation of the exams which are performed which lead to the patients diagnosis.
Overall the book is accurate. Would like to see updates related to the addition of direct radiography technology which is commonly used in the hospital setting.
Many aspects of medicine will remain constant. The case studies seem fairly accurate and may be relevant for up to 3 years. Since technology changes so quickly in medicine, the CT and x-ray components may need minor updates within a few years.
The book clarity is excellent.
The case stories are consistent with each scenario. It is easy to follow the structure and learn from the content.
The book is quite modular. It is easy to break it up into cases and utilize them individually and sequentially.
The cases are listed by disease process and follow a logical flow through each condition. They are easy to follow as they have the same format from the beginning to the end of each case.
The interface seems seamless. Hyperlinks are inserted which provide descriptions and references to medical procedures and in depth definitions.
The book is free of most grammatical errors. There is a place where a few words do not fit the sentence structure and could be a typo.
The book included all types of relationships and ethnic backgrounds. One type which could be added is a transgender patient.
I think the book was quite useful for a variety of health care professionals. The authors did an excellent job of integrating patient cases which could be applied to the health care setting. The stories seemed real and relevant. This book could be used to teach health care professionals about integrated care within the emergency department.
Reviewed by Shelley Wolfe, Assistant Professor, Winona State University on 5/21/18
This text is comprised of comprehensive, detailed case studies that provide the reader with multiple character views throughout a patient’s encounter with the health care system. The Table of Contents accurately reflected the content. It should... read more
This text is comprised of comprehensive, detailed case studies that provide the reader with multiple character views throughout a patient’s encounter with the health care system. The Table of Contents accurately reflected the content. It should be noted that the authors include a statement that conveys that this text is not like traditional textbooks and is not meant to be read in a linear fashion. This allows the educator more flexibility to use the text as a supplement to enhance learning opportunities.
The content of the text appears accurate and unbiased. The “five overarching learning objectives” provide a clear aim of the text and the educator is able to glean how these objectives are captured into each of the case studies. While written for the Canadian healthcare system, this text is easily adaptable to the American healthcare system.
Overall, the content is up-to-date and the case studies provide a variety of uses that promote longevity of the text. However, not all of the blue font links (if using the digital PDF version) were still in working order. I encountered links that led to error pages or outdated “page not found” websites. While the links can be helpful, continued maintenance of these links could prove time-consuming.
I found the text easy to read and understand. I enjoyed that the viewpoints of all the different roles (patient, nurse, lab personnel, etc.) were articulated well and allowed the reader to connect and gain appreciation of the entire healthcare team. Medical jargon was noted to be appropriate for the intended audience of this text.
The terminology and organization of this text is consistent.
The text is divided into 8 case studies that follow a similar organizational structure. The case studies can further be divided to focus on individual learning objectives. For example, the case studies could be looked at as a whole for discussing communication or could be broken down into segments to focus on disease risk factors.
The case studies in this text follow a similar organizational structure and are consistent in their presentation. The flow of individual case studies is excellent and sets the reader on a clear path. As noted previously, this text is not meant to be read in a linear fashion.
This text is available in many different forms. I chose to review the text in the digital PDF version in order to use the embedded links. I did not encounter significant interface issues and did not find any images or features that would distract or confuse a reader.
No significant grammatical errors were noted.
The case studies in this text included patients and healthcare workers from a variety of backgrounds. Educators and students will benefit from expanding the case studies to include discussions and other learning opportunities to help develop culturally-sensitive healthcare providers.
I found the case studies to be very detailed, yet written in a way in which they could be used in various manners. The authors note a variety of ways in which the case studies could be employed with students; however, I feel the authors could also include that the case studies could be used as a basis for simulated clinical experiences. The case studies in this text would be an excellent tool for developing interprofessional communication and collaboration skills in a variety healthcare students.
Reviewed by Darline Foltz, Assistant Professor, University of Cincinnati - Clermont College on 3/27/18
This book covers all areas listed in the Table of Contents. In addition to the detailed patient case studies, there is a helpful section of "How to Use this Resource". I would like to note that this resource "aligns with the open textbooks... read more
This book covers all areas listed in the Table of Contents. In addition to the detailed patient case studies, there is a helpful section of "How to Use this Resource". I would like to note that this resource "aligns with the open textbooks Clinical Procedures for Safer Patient Care and Anatomy and Physiology: OpenStax" as noted by the authors.
The book appears to be accurate. Although one of the learning outcomes is as follows: "Demonstrate an understanding of the Canadian healthcare delivery system.", I did not find anything that is ONLY specific to the Canadian healthcare delivery system other than some of the terminology, i.e. "porter" instead of "transporter" and a few french words. I found this to make the book more interesting for students rather than deter from it. These are patient case studies that are relevant in any country.
The content is up-to-date. Changes in medical science may occur, i.e. a different test, to treat a diagnosis that is included in one or more of the case studies, however, it would be easy and straightforward to implement these changes.
This book is written in lucid, accessible prose. The technical/medical terminology that is used is appropriate for medical and allied health professionals. Something that would improve this text would to provide a glossary of terms for the terms in blue font.
This book is consistent with current medical terminology
This text is easily divided into each of the 6 case studies. The case studies can be used singly according to the body system being addressed or studied.
Because this text is a collection of case studies, flow doesn't pertain, however the organization and structure of the case studies are excellent as they are clear and easy to read.
There are no distractions in this text that would distract or confuse the reader.
I did not identify any grammatical errors.
This text is not culturally insensitive or offensive in any way and uses patients and healthcare workers that are of a variety of races, ethnicities and backgrounds.
I believe that this text would not only be useful to students enrolled in healthcare professions involved in direct patient care but would also be useful to students in supporting healthcare disciplines such as health information technology and management, medical billing and coding, etc.
Table of Contents
- Introduction
Case Study #1: Chronic Obstructive Pulmonary Disease (COPD)
- Learning Objectives
- Patient: Erin Johns
- Emergency Room
Case Study #2: Pneumonia
- Day 0: Emergency Room
- Day 1: Emergency Room
- Day 1: Medical Ward
- Day 2: Medical Ward
- Day 3: Medical Ward
- Day 4: Medical Ward
Case Study #3: Unstable Angina (UA)
- Patient: Harj Singh
Case Study #4: Heart Failure (HF)
- Patient: Meryl Smith
- In the Supermarket
- Day 0: Medical Ward
Case Study #5: Motor Vehicle Collision (MVC)
- Patient: Aaron Knoll
- Crash Scene
- Operating Room
- Post Anaesthesia Care Unit (PACU)
- Surgical Ward
Case Study #6: Sepsis
- Patient: George Thomas
- Sleepy Hollow Care Facility
Case Study #7: Colon Cancer
- Patient: Fred Johnson
- Two Months Ago
- Pre-Surgery Admission
Case Study #8: Deep Vein Thrombosis (DVT)
- Patient: Jamie Douglas
Appendix: Overview About the Authors
Ancillary Material
About the book.
Health Case Studies is composed of eight separate health case studies. Each case study includes the patient narrative or story that models the best practice (at the time of publishing) in healthcare settings. Associated with each case is a set of specific learning objectives to support learning and facilitate educational strategies and evaluation.
The case studies can be used online in a learning management system, in a classroom discussion, in a printed course pack or as part of a textbook created by the instructor. This flexibility is intentional and allows the educator to choose how best to convey the concepts presented in each case to the learner.
Because these case studies were primarily developed for an electronic healthcare system, they are based predominantly in an acute healthcare setting. Educators can augment each case study to include primary healthcare settings, outpatient clinics, assisted living environments, and other contexts as relevant.
About the Contributors
Glynda Rees teaches at the British Columbia Institute of Technology (BCIT) in Vancouver, British Columbia. She completed her MSN at the University of British Columbia with a focus on education and health informatics, and her BSN at the University of Cape Town in South Africa. Glynda has many years of national and international clinical experience in critical care units in South Africa, the UK, and the USA. Her teaching background has focused on clinical education, problem-based learning, clinical techniques, and pharmacology.
Glynda‘s interests include the integration of health informatics in undergraduate education, open accessible education, and the impact of educational technologies on nursing students’ clinical judgment and decision making at the point of care to improve patient safety and quality of care.
Faculty member in the critical care nursing program at the British Columbia Institute of Technology (BCIT) since 2003, Rob has been a critical care nurse for over 25 years with 17 years practicing in a quaternary care intensive care unit. Rob is an experienced educator and supports student learning in the classroom, online, and in clinical areas. Rob’s Master of Education from Simon Fraser University is in educational technology and learning design. He is passionate about using technology to support learning for both faculty and students.
Part of Rob’s faculty position is dedicated to providing high fidelity simulation support for BCIT’s nursing specialties program along with championing innovative teaching and best practices for educational technology. He has championed the use of digital publishing and was the tech lead for Critical Care Nursing’s iPad Project which resulted in over 40 multi-touch interactive textbooks being created using Apple and other technologies.
Rob has successfully completed a number of specialist certifications in computer and network technologies. In 2015, he was awarded Apple Distinguished Educator for his innovation and passionate use of technology to support learning. In the past five years, he has presented and published abstracts on virtual simulation, high fidelity simulation, creating engaging classroom environments, and what the future holds for healthcare and education.
Janet Morrison is the Program Head of Occupational Health Nursing at the British Columbia Institute of Technology (BCIT) in Burnaby, British Columbia. She completed a PhD at Simon Fraser University, Faculty of Communication, Art and Technology, with a focus on health information technology. Her dissertation examined the effects of telehealth implementation in an occupational health nursing service. She has an MA in Adult Education from St. Francis Xavier University and an MA in Library and Information Studies from the University of British Columbia.
Janet’s research interests concern the intended and unintended impacts of health information technologies on healthcare students, faculty, and the healthcare workforce.
She is currently working with BCIT colleagues to study how an educational clinical information system can foster healthcare students’ perceptions of interprofessional roles.
Contribute to this Page
Log in using your username and password
- Search More Search for this keyword Advanced search
- Latest content
- Current issue
- Write for Us
- BMJ Journals
You are here
- Volume 21, Issue 1
- What is a case study?
- Article Text
- Article info
- Citation Tools
- Rapid Responses
- Article metrics
- Roberta Heale 1 ,
- Alison Twycross 2
- 1 School of Nursing , Laurentian University , Sudbury , Ontario , Canada
- 2 School of Health and Social Care , London South Bank University , London , UK
- Correspondence to Dr Roberta Heale, School of Nursing, Laurentian University, Sudbury, ON P3E2C6, Canada; rheale{at}laurentian.ca
https://doi.org/10.1136/eb-2017-102845
Statistics from Altmetric.com
Request permissions.
If you wish to reuse any or all of this article please use the link below which will take you to the Copyright Clearance Center’s RightsLink service. You will be able to get a quick price and instant permission to reuse the content in many different ways.
What is it?
Case study is a research methodology, typically seen in social and life sciences. There is no one definition of case study research. 1 However, very simply… ‘a case study can be defined as an intensive study about a person, a group of people or a unit, which is aimed to generalize over several units’. 1 A case study has also been described as an intensive, systematic investigation of a single individual, group, community or some other unit in which the researcher examines in-depth data relating to several variables. 2
Often there are several similar cases to consider such as educational or social service programmes that are delivered from a number of locations. Although similar, they are complex and have unique features. In these circumstances, the evaluation of several, similar cases will provide a better answer to a research question than if only one case is examined, hence the multiple-case study. Stake asserts that the cases are grouped and viewed as one entity, called the quintain . 6 ‘We study what is similar and different about the cases to understand the quintain better’. 6
The steps when using case study methodology are the same as for other types of research. 6 The first step is defining the single case or identifying a group of similar cases that can then be incorporated into a multiple-case study. A search to determine what is known about the case(s) is typically conducted. This may include a review of the literature, grey literature, media, reports and more, which serves to establish a basic understanding of the cases and informs the development of research questions. Data in case studies are often, but not exclusively, qualitative in nature. In multiple-case studies, analysis within cases and across cases is conducted. Themes arise from the analyses and assertions about the cases as a whole, or the quintain, emerge. 6
Benefits and limitations of case studies
If a researcher wants to study a specific phenomenon arising from a particular entity, then a single-case study is warranted and will allow for a in-depth understanding of the single phenomenon and, as discussed above, would involve collecting several different types of data. This is illustrated in example 1 below.
Using a multiple-case research study allows for a more in-depth understanding of the cases as a unit, through comparison of similarities and differences of the individual cases embedded within the quintain. Evidence arising from multiple-case studies is often stronger and more reliable than from single-case research. Multiple-case studies allow for more comprehensive exploration of research questions and theory development. 6
Despite the advantages of case studies, there are limitations. The sheer volume of data is difficult to organise and data analysis and integration strategies need to be carefully thought through. There is also sometimes a temptation to veer away from the research focus. 2 Reporting of findings from multiple-case research studies is also challenging at times, 1 particularly in relation to the word limits for some journal papers.
Examples of case studies
Example 1: nurses’ paediatric pain management practices.
One of the authors of this paper (AT) has used a case study approach to explore nurses’ paediatric pain management practices. This involved collecting several datasets:
Observational data to gain a picture about actual pain management practices.
Questionnaire data about nurses’ knowledge about paediatric pain management practices and how well they felt they managed pain in children.
Questionnaire data about how critical nurses perceived pain management tasks to be.
These datasets were analysed separately and then compared 7–9 and demonstrated that nurses’ level of theoretical did not impact on the quality of their pain management practices. 7 Nor did individual nurse’s perceptions of how critical a task was effect the likelihood of them carrying out this task in practice. 8 There was also a difference in self-reported and observed practices 9 ; actual (observed) practices did not confirm to best practice guidelines, whereas self-reported practices tended to.
Example 2: quality of care for complex patients at Nurse Practitioner-Led Clinics (NPLCs)
The other author of this paper (RH) has conducted a multiple-case study to determine the quality of care for patients with complex clinical presentations in NPLCs in Ontario, Canada. 10 Five NPLCs served as individual cases that, together, represented the quatrain. Three types of data were collected including:
Review of documentation related to the NPLC model (media, annual reports, research articles, grey literature and regulatory legislation).
Interviews with nurse practitioners (NPs) practising at the five NPLCs to determine their perceptions of the impact of the NPLC model on the quality of care provided to patients with multimorbidity.
Chart audits conducted at the five NPLCs to determine the extent to which evidence-based guidelines were followed for patients with diabetes and at least one other chronic condition.
The three sources of data collected from the five NPLCs were analysed and themes arose related to the quality of care for complex patients at NPLCs. The multiple-case study confirmed that nurse practitioners are the primary care providers at the NPLCs, and this positively impacts the quality of care for patients with multimorbidity. Healthcare policy, such as lack of an increase in salary for NPs for 10 years, has resulted in issues in recruitment and retention of NPs at NPLCs. This, along with insufficient resources in the communities where NPLCs are located and high patient vulnerability at NPLCs, have a negative impact on the quality of care. 10
These examples illustrate how collecting data about a single case or multiple cases helps us to better understand the phenomenon in question. Case study methodology serves to provide a framework for evaluation and analysis of complex issues. It shines a light on the holistic nature of nursing practice and offers a perspective that informs improved patient care.
- Gustafsson J
- Calanzaro M
- Sandelowski M
Competing interests None declared.
Provenance and peer review Commissioned; internally peer reviewed.
Read the full text or download the PDF:
- PRO Courses Guides New Tech Help Pro Expert Videos About wikiHow Pro Upgrade Sign In
- EDIT Edit this Article
- EXPLORE Tech Help Pro About Us Random Article Quizzes Request a New Article Community Dashboard This Or That Game Happiness Hub Popular Categories Arts and Entertainment Artwork Books Movies Computers and Electronics Computers Phone Skills Technology Hacks Health Men's Health Mental Health Women's Health Relationships Dating Love Relationship Issues Hobbies and Crafts Crafts Drawing Games Education & Communication Communication Skills Personal Development Studying Personal Care and Style Fashion Hair Care Personal Hygiene Youth Personal Care School Stuff Dating All Categories Arts and Entertainment Finance and Business Home and Garden Relationship Quizzes Cars & Other Vehicles Food and Entertaining Personal Care and Style Sports and Fitness Computers and Electronics Health Pets and Animals Travel Education & Communication Hobbies and Crafts Philosophy and Religion Work World Family Life Holidays and Traditions Relationships Youth
- Browse Articles
- Learn Something New
- Quizzes Hot
- Happiness Hub
- This Or That Game
- Train Your Brain
- Explore More
- Support wikiHow
- About wikiHow
- Log in / Sign up
- Education and Communications
- Medical Studies
How to Write a Medical Case Study Report
Last Updated: April 18, 2024 Approved
This article was medically reviewed by Mark Ziats, MD, PhD and by wikiHow staff writer, Jennifer Mueller, JD . Dr. Mark Ziats is an Internal Medicine Physician, Scientist, Entrepreneur, and the Medical Director of xBiotech. With over five years of experience, he specializes in biotechnology, genomics, and medical devices. He earned a Doctor of Medicine degree from Baylor College of Medicine, a Ph.D. in Genetics from the University of Cambridge, and a BS in Biochemistry and Chemistry from Clemson University. He also completed the INNoVATE Program in Biotechnology Entrepreneurship at The Johns Hopkins University - Carey Business School. Dr. Ziats is board certified by the American Board of Internal Medicine. There are 15 references cited in this article, which can be found at the bottom of the page. wikiHow marks an article as reader-approved once it receives enough positive feedback. In this case, 100% of readers who voted found the article helpful, earning it our reader-approved status. This article has been viewed 189,056 times.
You've encountered an interesting and unusual case on your rounds, and a colleague or supervising physician says, "Why don't you write up a case study report?" If you've never written one before, that might sound intimidating, but it's a great way to get started in medical writing. Case studies always follow a standard structure and format, so the writing is very formulaic once you get the hang of it. Read on for a step-by-step guide to writing your first case study report.
What is a case study report?

- Medical students or residents typically do the bulk of the writing of the report. If you're just starting your medical career, a case study report is a great way to get a publication under your belt. [2] X Research source

- If the patient is a minor or is incapable of giving informed consent, get consent from their parents or closest relative. [4] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source
- Your hospital likely has specific consent forms to use. Ask your supervising physician if you're not sure where to get one.
- Some journals also have their own consent form. Check your target journal's author or submission information to make sure. [5] X Research source
How is a case study report structured?

- Even though the introduction is the first part of a case study report, doctors typically write it last. You'll have a better idea of how to introduce your case study to readers after you've written it.
- Your abstract comes at the top, before the introduction, and provides a brief summary of the entire report. Unless your case study is published in an open-access journal, the abstract is the only part of the article many readers will see.

- Many journals offer templates and checklists you can use to make sure your case study includes everything necessary and is formatted properly—take advantage of these! Some journals, such as BMJ Case Reports , require all case studies submitted to use their templates.
Drafting Your Medical Case Study Report

- Patient description
- Chronological case history
- Physical exam results
- Results of any pathological tests, imaging, or other investigations
- Treatment plan
- Expected outcome of treatment
- Actual outcome of treatment

- Why the patient sought medical help (you can even use their own words)
- Important information that helped you settle on your diagnosis
- The results of your clinical examination, including diagnostic tests and their results, along with any helpful images
- A description of the treatment plan
- The outcome, including how and why treatment ended and how long the patient was under your care [11] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source

- You will need references to back up symptoms of the condition, common treatment, and the expected outcome of that common treatment.
- Use your research to paint a picture of the usual case of a patient with a similar condition—it'll help you show how unusual and different your patient's case is.
- Generally, aim for around 20 references—no fewer than 15, but no more than 25. [13] X Trustworthy Source PubMed Central Journal archive from the U.S. National Institutes of Health Go to source

- Close your discussion section with a summary of the lessons learned from the case and why it's significant to consider when treating similar cases in the future.
- Outline any open questions that remain. You might also provide suggestions for future research.

- In your conclusion, you might also give suggestions or recommendations to readers based on what you learned as a result of the case.
- Some journals don't want a separate conclusion section. If that's the case for one of your target journals, just move this paragraph to the end of your discussion section.
Polishing Your Report for Submission to Publishers

- Most titles are fewer than 10 words long and include the name of the disease or condition treated.
- You might also include the treatment used and whether the outcome was successful. When deciding what to include, think about the reason you wrote the case study in the first place and why you think it's important for other clinicians to read.

- Made a significant intellectual contribution to the case study report
- Was involved in the medical care of the patient reported
- Can explain and defend the data presented in the report
- Has approved the final manuscript before submission for publication

- Keep in mind that the abstract is not just going to be the first thing people read—it will often be the only thing people read. Make sure that if someone is going to walk away having only read the abstract, they'll still get the same message they would have if they read the whole thing.
- There are 2 basic types of abstract: narrative and structured. A narrative abstract is a single paragraph written in narrative prose. A structured abstract includes headings that correspond with the sections of the paper, then a brief summary of each section. Use the format preferred by your target journal.

- Look for keywords that are relevant to your field or sub-field and directly related to the content of your article, such as the name of the condition or specific treatments you used.
- Most journals allow 4-8 keywords but check the submission guidelines of your target journal to make sure.

- Blur out the patient's face as well as any tattoos, birthmarks, or unrelated scars that are visible in diagnostic images.

- It's common to thank the patient, but that's up to you. Even if you don't, include a statement indicating that you have the patient's written, informed consent to publish the information.
- Read the journal's submission guidelines for a definition of what that journal considers a conflict of interest. They're generally the same, but some might be stricter than others. [22] X Research source

- If you're not familiar with the citation style used by your target journal, check online for a guide. There might also be one available at your hospital or medical school library.
- Medical librarians can also help with citation style and references if you run into something tricky—don't just wing it! Correct citation style insures that readers can access the materials you cite.

- It's also a good idea to get a beta reader who isn't a medical professional. Their comments can help you figure out where you need to clarify your points.
- Read a lot of case studies published in your target journals—it will help you internalize the tone and style that journal is looking for.
Submitting Your Report to Publishers

- Look into the background and reputation of journals before you decide to submit to them. Only seek publication from reputable journals in which articles go through a peer-review process.
- Find out what publishing fees the journals charge. Keep in mind that open-access journals tend to charge higher publishing fees. [26] X Research source
- Read each journal's submission and editorial guidelines carefully. They'll tell you exactly how to format your case study, how long each section should be, and what citation style to use. [27] X Research source
- For electronic journals that only publish case reports, try BMJ Case Reports , Journal of Medical Case Reports , or Radiology Case Reports .

- If your manuscript isn't suitable for the journal you submitted to, the journal might offer to forward it to an associated journal where it would be a better fit.
- When your manuscript is provisionally accepted, the journal will send it to other doctors for evaluation under the peer-review process.
- Most medical journals don't accept simultaneous submissions, meaning you'll have to submit to your first choice, wait for their decision, then move to the next journal on the list if they don't bite.

- Along with your revised manuscript, include a letter with your response to each of the reviewer's comments. Where you made revisions, add page numbers to indicate where the revisions are that address that reviewer's comments.
- Sometimes, doctors involved in the peer review process will indicate that the journal should reject the manuscript. If that's the case, you'll get a letter explaining why your case study report won't be published and you're free to submit it elsewhere.

- Some journals require you to have your article professionally copy-edited at your own cost while others do this in-house. The editors will let you know what you're responsible for.

- With your acceptance letter, you'll get instructions on how to make payment and how much you owe. Take note of the deadline and make sure you pay it as soon as possible to avoid publication delays.
- Some journals will publish for free, with an "open-access option" that allows you to pay a fee only if you want open access to your article. [32] X Research source

- Through the publishing agreement, you assign your copyright in the article to the journal. This allows the journal to legally publish your work. That assignment can be exclusive or non-exclusive and may only last for a specific term. Read these details carefully!
- If you published an open-access article, you don't assign the copyright to the publisher. The publishing agreement merely gives the journal the right to publish the "Version of Record." [33] X Research source
How do I find a suitable case for a report?

- A rare disease, or unusual presentation of any disease
- An unusual combination of diseases or conditions
- A difficult or inconclusive diagnosis
- Unexpected developments or responses to treatment
- Personal impact
- Observations that shed new light on the patient's disease or condition

- There might be other members of your medical team that want to help with writing. If so, use one of these brainstorming sessions to divvy up writing responsibilities in a way that makes the most sense given your relative skills and experience.
- Senior doctors might also be able to name some journals that would potentially publish your case study. [36] X Research source
Expert Q&A
You Might Also Like

- ↑ https://www.elsevier.com/connect/authors-update/the-dos-and-donts-of-writing-and-publishing-case-reports
- ↑ https://www.bmj.com/content/350/bmj.h2693
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5686928/
- ↑ https://health.usf.edu/medicine/internalmedicine/im-impact/~/media/B3A3421F4C144FA090AE965C21791A3C.ashx
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2597880/
- ↑ https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6476221/
- ↑ https://www.springer.com/gp/authors-editors/authorandreviewertutorials/writing-a-journal-manuscript/title-abstract-and-keywords/10285522
- ↑ http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2597880/
- ↑ https://thelancet.com/pb/assets/raw/Lancet/authors/tl-info-for-authors.pdf
- ↑ https://jmedicalcasereports.biomedcentral.com/articles/10.1186/s13256-017-1351-y
- ↑ https://guides.himmelfarb.gwu.edu/casereports
- ↑ https://casereports.bmj.com/pages/authors/
- ↑ https://jmedicalcasereports.biomedcentral.com/articles/10.1186/1752-1947-7-239
- ↑ https://research.chm.msu.edu/students-residents/writing-a-case-report
- ↑ https://authorservices.taylorandfrancis.com/publishing-your-research/moving-through-production/copyright-for-journal-authors/#
About This Article

Medical Disclaimer
The content of this article is not intended to be a substitute for professional medical advice, examination, diagnosis, or treatment. You should always contact your doctor or other qualified healthcare professional before starting, changing, or stopping any kind of health treatment.
Read More...
To start a medical case study report, first choose a title that clearly reflects the contents of the report. You’ll also need to list any participating authors and develop a list of keywords, as well as an abstract summarizing the report. Your report will need to include an introduction summarizing the context of the report, as well as a detailed presentation of the case. Don’t forget to include a thorough citation list and acknowledgements of anyone else who participated in the study. For more tips from our Medical co-author, including how to get your case study report published, keep reading! Did this summary help you? Yes No
- Send fan mail to authors
Reader Success Stories
Sep 5, 2020
Did this article help you?

Asfia Banu Pasha
Apr 10, 2017
Jun 20, 2021
Mar 1, 2017

Featured Articles

Trending Articles

Watch Articles

- Terms of Use
- Privacy Policy
- Do Not Sell or Share My Info
- Not Selling Info
Don’t miss out! Sign up for
wikiHow’s newsletter
- McMaster University Health Sciences Library
Medicine - Undergraduate
- Case Scenarios
- Students from a Non-Science Background
- Anatomy & Physiology
- Clinical Skills
- Drugs, Natural Products & Nutrition
- Indigenous Health Collection
- Black Health Resources
- EBM & JAMA Evidence
- PICO: writing a searchable question
- Research Support - Book a Consult
- Landmark Papers
- Student Wellness

Case Studies & Scenarios

- Johns Hopkins Medicine- Case Studies
- Case Studies for Fellows (American Society of Hematology)
- LITFL Clinical Cases Dedicated to Emergency Medicine and Critical Care
- Emergency Medicine Cases Canadian medical education podcast
- NEJM Clinical Cases
- Springer Clinical Cases series A catalogue search
- << Previous: LMCC Prep
- Next: Anatomy & Physiology >>
- Last Updated: Aug 26, 2024 6:29 AM
- URL: https://hslmcmaster.libguides.com/medicine_UGM


Clinical Cases
Litfl clinical cases database.
The LITFL Clinical Case Collection includes over 250 Q&A style clinical cases to assist ‘ Just-in-Time Learning ‘ and ‘ Life-Long Learning ‘. Cases are categorized by specialty and can be interrogated by keyword from the Clinical Case searchable database.
Search by keywords; disease process; condition; eponym or clinical features…
| Topic | Title | Keywords |
|---|---|---|
| ECG | ||
| ECG | WCT, ECG, Broad complex, fascicular, RVOT | |
| Toxicology | valproate, valproic acid, hyperammonemia | |
| Toxicology | valproate, valproic acid, hyperammonemia | |
| Toxicology | ||
| Metabolic | priapsim, intracavernosal, cavernosal gas, Ischaemic priapism, stuttering priapism, urology | |
| Metabolic | RTA, strong ion difference, hypocalcaemia | |
| Bone and Joint | DRUJ, dislcoation | |
| ICE | wellens, ECG, cardiac, delay | |
| ICE | SJS, stevens-johnson syndrome, erythema multiforme, rash | |
| ICE | pneumothorax | |
| ICE | ||
| ICE | tibia, fracture, toddler, toddler's fracture | |
| ICE | ECG, EKG, hyperkalaemia, hyperkalemia | |
| ICE | dengue, returned traveler, traveller | |
| ICE | Lisfranc | |
| ICE | mountain, mount everest, alkalaemia, alkalemia | |
| ICE | pancreatitis, alcohol | |
| ICE | segond fracture | |
| ICE | Brugada | |
| ICE | STEMI, hyperacture, myocardial ischemia, anterior | |
| ICE | eryhthema nodosum, panniculitis | |
| ICE | BOS fracture, battle sign, mastoid ecchymosis, bruising | |
| ICE | Galleazi, fracture dislocation | |
| Toxicology | methylene blue, Methaemoglobinemia, methemoglobin | |
| Toxicology | clozapine | |
| Toxicology | Methamphetamine, body stuffing, body packer, body stuffer | |
| Toxicology | TCA, tricyclic, overdose, sodium channel blockade | |
| Toxicology | alprazolam, BZD, benzo, benzodiazepine, benzodiazepines, flumazenil | |
| Toxicology | lithium, neurotoxicity, acute toxicity | |
| Toxicology | baclofen, GABA, Gamma-Hydroxybutyrate, GHB | |
| Toxicology | Carbamazepine, toxidrome, carbamazepine cardiotoxicity, Tegretol, multiple-dose activated charcoal, MDAC | |
| Toxicology | Hepatotoxicity, Acetaminophen, Schiodt score, hepatic encephalopathy, N-acetylcysteine, NAC | |
| Toxicology | beta-blocker, B Blocker, | |
| Toxicology | Cannabinoid Hyperemesis Syndrome, cyclical vomiting, THC, delta-nine-tetrahydrocannabinol | |
| Toxicology | Colchicine | |
| Toxicology | Clonidine | |
| Toxicology | Bath salts | |
| Toxicology | Mephedrone | |
| Toxicology | Bromo-DragonFLY, M-ket, Kmax, Mexxy, Meow-Meow, Mephedrone, Methoxetamine, Naphyrone, NRG-1, Salvia, K2, Spice | |
| Toxicology | ixodes holocyclus, tick, paralysis, | |
| Toxicology | cyanide, carbon monoxide | |
| Toxicology | hypoglycemia | |
| Toxicology | Ciguatera, Scombroid, fugu, puffer fish | |
| Toxicology | ethylene glycol, HAGMA, high anion gap metabolic acidosis, osmolar gap, Fomepizole, alcohol, ethanol | |
| Toxicology | iron toxicity, Desferrioxamine chelation therapy | |
| Toxicology | chloroquine | |
| Toxicology | corrosive agent | |
| Toxicology | Antidote | |
| Toxicology | Oculogyric crisis, OGC, acute dystonia, Acute Dystonic Reaction, butyrophenone, Metoclopramide, haloperidol, prochlorperazine, Benztropine | |
| Toxicology | Tricyclic, Theophylline, Sulfonylureas, Propanolol, Opioids, Dextropropoxyphene, Chloroquine, Calcium channel blockers, Amphetamines, ectasy | |
| Toxicology | verapamil, calcium channel blocker, cardiotoxic, HIET, high-dose insulin euglycemic therapy, | |
| Toxicology | aroma, smell | |
| Toxinology | snake-bite, snake bite, Brown snake, Black, Death adder, Taipan, sea snake, tiger | |
| Toxicology | Anticholinergic syndrome, Malignant hyperthermia, Neuroleptic malignant syndrome, Serotonin toxicity | |
| Toxicology | Serotonin toxicity, Serotonin syndrome, toxidrome | |
| Toxicology | proconvulsive, venlafaxine, tramadol, amphetamines, Bupropion, Otis Campbell | |
| Toxicology | TCA, tricyclic, overdose, sodium channel blockade, Amitriptyline | |
| Toxicology | anticoagulation, warfarin | |
| Toxicology | Mickey Finn, pear, | |
| Toxicology | thyrotoxic storm, Thyroxine, T4 | |
| Toxinology | white-tailed spider, Lampona, L. cylindrata, L. murina | |
| Toxicology | Citalopram, SSRI, | |
| Toxicology | warfarin | |
| Toxicology | warfarin, accidental ingestion, toddler | |
| Toxicology | ||
| Toxinology | Marine, envenoming | |
| Toxinology | Marine, envenoming, penetrating, barb, steve irwin, | |
| Toxinology | Marine, envenoming, Blue-Ringed Octopus, BRO, Hapalochlaena | |
| Toxinology | Jellyfish, marine, Chironex fleckeri, Box Jellyfish | |
| Toxinology | Jellyfish, marine, Jack Barnes, Carukia barnesi, Irukandji Syndrome, Darwin | |
| Toxinology | Jellyfish, marine, Jack Barnes, Carukia barnesi, Irukandji Syndrome | |
| Toxicology | Strychnine, opisthotonus, risus sardonicus | |
| Toxicology | naloxone, Buprenorphine | |
| Toxinology | snake-bite, snake bite, SVDK | |
| Toxinology | Red back spider, redback, envenoming, RBS | |
| Toxinology | Red back spider, redback, envenoming, RBS | |
| Toxicology | ||
| Toxicology | Acetaminophen, N-acetylcysteine, NAC | |
| Pediatric |
| Henoch-Schonlein Purpura, HSP, Henoch-Schönlein |
| Pediatric |
| adrenal insufficiency, glucocorticoid deficiency, NAGMA, endocrine emergency |
| Pediatric |
| Penile Zipper Entrapment, foreskin, release, Zip |
| Pediatric |
| diarrohea, vomiting, hypokalemia, hypokalaemia, dehydration |
| Pediatric |
| infantile colic, TIM CRIES, crying baby |
| Pediatric |
| Pyloric stenosis, projectile vomit, hypertrophic pyloric stenosis, HPS, Rankin |
| Pediatric |
| respiratory distress, wheeze, foreign body, RMB, CXR, right main bronchus |
| Pediatric |
| airway obstruction, stridor, severe croup, harsh cough, heliox, intubation, sevoflurane |
| Pediatric |
| boot-shaped, TOF, coeur en sabot, Tetralogy of Fallot |
| Pediatric |
| Spherocytes, Shistocytes, Polychromasia, reticulocytosis, anemia, anaemia, hemolytic uremic syndrome, HUS |
| Pediatric |
| Reye syndrome, ammonia, metabolic encephalopathy, aspirin |
| Pediatric |
| Ketamine, procedural sedation, pediatric sedation |
| Pediatric |
| Foreign Body, ketamine, laryngospasm, Larson's point, laryngospasm notch |
| EYE |
| ophthalmology, eye trauma, Eyelid laceration, lacrimal punctum |
| EYE |
| ophthalmology, Retrobulbar hemorrhage, haemorrhage, RAPD, lateral canthotomy, DIP-A CONE-G, cantholysis |
| EYE |
| ophthalmology, corneal abrasion, eye trauma, eyelid eversion |
| EYE |
| ophthalmology, commotio retinae, eye trauma, traumatic eye injury |
| EYE |
| ophthalmology, Traumatic iritis, hyphaema, hyphema, |
| EYE |
| ophthalmology, lens dislocation, Anterior dislocation of an intraocular lens |
| EYE |
| ophthalmology, visual loss, loss of vision , blind |
| EYE |
| ophthalmology, Central retinal vein occlusion, CRVO, branch retinal vein occlusion, BRVO |
| EYE |
| ophthalmology, Central retinal artery occlusion, CRAO, cherry red spot, Branch retinal artery occlusion, BRAO |
| EYE |
| ophthalmology, miosis, partial ptosis, anhidrosis, enophthalmos, horner |
| EYE |
| ophthalmology, visual loss, Amaurosis fugax, TIA, transient ischemic attack |
| EYE |
| ophthalmology, Pre-septal cellulitis, preseptal cellulitis, peri-orbital cellulitis, Post-septal cellulitis, post septal cellulitis, orbital cellulitis |
| EYE |
| ophthalmology, AION, giant cell arteritis, GCA, Anterior ischemic optic neuropathy |
| EYE |
| ophthalmology, Herpes simplex keratitis, dendritic ulcer |
| EYE |
| ophthalmology, Conjunctival injection, conjunctivitis, keratoconjunctivitis, Adenovirus, trachoma, bacterial, viral, Parinaud oculoglandular conjunctivitis |
| EYE |
| ophthalmology, Chemical injury, cement, alkali, burn, chemical conjunctivitis, colliquative necrosis, liquefactive |
| EYE |
| ophthalmology, Ultraviolet keratitis, keratopathy, solar keratitis, photokeratitis, welder's flash, arc eye, bake eyes snow blindness. |
| EYE |
| ophthalmology, Parinaud, adie, holmes, tabes dorsalis, neurosyphylis, argyll Robertson, small irregular |
| EYE |
| ophthalmology, anterior Uveitis, HLA-B27, hypopyon |
| EYE |
| ophthalmology, POCUS, ONSD, |
| EYE |
| ophthalmology, Blowout fracture, infraorbital fracture |
| EYE |
| ophthalmology, endophthalmitis, sympathetic ophthalmia, penetrating eye trauma |
| EYE |
| ophthalmology, tobacco dust, Posterior vitreous detachment, vitreous debris, retinal tear, retinal break, Washer Machine Sign, Eales disease |
| EYE |
| ophthalmology, Herpes zoster ophthalmicus, dendriform keratitis, Hutchinson sign |
| EYE |
| ophthalmology, Siedel, FB, rust ring, Corneal foreign body, Seidel test |
| EYE |
| ophthalmology, Papilloedema, Papilledema, pseudopapilloedema |
| EYE |
| ophthalmology, optic disc, optic neuritis, Marcus-Gunn, papillitis, multiple sclerosis, funduscopy, optic atrophy, papilledema |
| EYE |
| ophthalmology, retinal break, POCUS, retinoschisis, Retinal detachment |
| EYE |
| ophthalmology, cupping, glaucoma, optic neuropathy, tonometry, intraocular pressure, open angle, closed angle, gonioplasty, Acute closed-angle glaucoma |
| EYE |
| ophthalmology, Subconjunctival hemorrhage |
| EYE |
| ophthalmology, Meibomitis, blepharitis, entropion, ectropion, canaliculitis, dacryocystitis |
| EYE |
| ophthalmology, blepharospasm, blink, blinking |
| EYE |
| Iritis, keratitis, acute angle-closure glaucoma, scleritis, orbital cellulitis, cavernous sinus thrombosis (CST) |
| EYE |
| ophthalmology, fixed, dilated, pupil, holmes-adie, glass eye |
| ECG |
| Wenckebach, AV block, SA, deliberate mistake, SA block |
| ECG |
| dual chamber AV sequential pacemaker |
| ECG |
| anterior AMI, De Winter T waves, LAD stenosis |
| ECG |
| LMCA Stenosis, ST elevation in aVR, Left Main Coronary Artery |
| ECG |
| LMCA, Left Main Coronary Artery Occlusion, ST elevation in aVR |
| ECG |
| VT, BCT, WCT, Brugada criteria, Verekie |
| ECG |
| severe hypokalaemia, spironalactone, rhabdomyolysis, ECG, u wave, diabetic ketoacidosis |
| ECG |
| pacing, pacemaker, post-op, Mobitz I, Wenckebach, AV block |
| ECG |
| bidirectional ventricular tachycardia, Catecholaminergic Polymorphic Ventricular Tachycardia, CPVT, digoxin toxicity |
| ECG |
| congenital, short QT syndrome, SQTS, AF, Atrial fibrillation |
| ECG |
| RVOT, broad complex tachycardia, BCT, Right Ventricular Outflow Tract Tachycardia, VF, Arrest, Arrhythmogenic Right Ventricular Cardiomyopathy, ARVC |
| ECG |
| NSTEMI, inverted U wave, |
| ECG |
| tricyclic antidepressant, TCA, Doxepin, QRS broadening, cardiotoxic |
| ECG |
| AIVR, Accelerated idioventricular rhythm, Isorhythmic AV dissociation, Sinus arrhythmia, idioventricular |
| ECG |
| LAD, LBBB, High left ventricular voltage, HLVV, WPW, Broad Complex Tachycardia |
| ECG |
| tachy-brady, AVNRT, flutter, polymorphic VT, VF, torsades de pointes, R on T, Cardioversion |
| ECG |
| LBBB, Wellens, ECG, proximal LAD, occlusion, rate-dependent, inferior ischaemia |
| ECG |
| SI QIII TIII, PE, PTE pulmonary embolism, PEA arrest, RBBB, LAD |
| Cardiology |
| HOCM, STE, aVR, LMCA, torsades des pointes. TDP |
| Cardiology |
| aortic arch, right sided, diverticulum of Kommerell |
| Cardiology |
| IABP, CABG, shock, circulatory collapse |
| Cardiology |
| electrical alternans, ECG, pulsus paradoxus |
| Cardiology |
| Intra-aortic Balloon Pump, Waveform, dicrotic notch |
| Cardiology |
| DeBakey, TAA, aortic dissection, CTA |
| Cardiology |
| Tetraology of Fallot, BT shunt, Blalock-Tausig, ToF |
| Cardiology |
| PVP, cement, embolus, Percutaneous Vertebroplasty |
| Cardiology |
| Pulmonary Embolism, PTE, PE, McConnell, thrombolysis, echo |
| Bone and Joint |
| Missed posterior shoulder dislocation |
| Paediatrics |
| rash, neck nodule, Kawasaki |
| Paediatrics |
| rash, fever, scarlet, strawberry, Group A Beta Haemolytic Streptococci (GABHS) |
| Tropical Travel |
| diphtheria, pseudomembrance, grey tonsils, pseudomembrane, tonsillitis, diphtheria, Corynebacterium diphtheriae, gram-positive bacillus |
| Urinalysis |
| purple, urine, indican, indican |
| Urinalysis |
| brown, urine, rhabdomyolysis |
| Urinalysis |
| green, urine, propofol, PRIS |
| Urinalysis |
| green, urine |
| Urinalysis |
| orange, urine |
| Bone and Joint |
| Nail, trauma, hematoma, subungual, haematoma, nail-bed |
| Bone and Joint |
| Extensor tendon, hand injury, extensor digiti minimi, |
| Bone and Joint |
| Thumb, fracture, base, phalanx, metacarpal, Edward Hallaran Bennett, bipartate |
| Paediatrics |
| Food allergy, enterocolitis, |
| Bone and Joint |
| FOOSH, wrist fracture, FOOSH - 'fall onto outstretched hand', Barton fracture, John Rhea Barton |
| Paediatrics |
| pulled elbow, nursemaid, hyperpronation |
| Cardiology |
| Phlegmasia, dolens |
| Cardiology |
| ICC, intercostal, intra-cardiac, iatrogenic |
| Bone and Joint |
| Compartment syndrome, Volkmann, fasciotomy |
| Bone and Joint |
| Ankle, compound, fracture, dislocation, Six Hour Golden Rule, saline, iodine |
| ENT |
| retropharngeal abscess, posterior pharynx, mediastinitis, Lemierre syndrome, Fusobacterium necrophorum |
| ENT |
| enlarged tonsils, pharyngitis, tonsillitis |
| Toxicology | Colgout, colchicine, label, fenofibrate | |
| Tropical Travel | Mary Mallon, Salmonella typhi, typhoid, typhoid mary | |
| Tropical Travel | Dengue Fever, single-stranded RNA virus, Aedes, mosquito, Dengue Shock Syndrome (DSS), Dengue Haemorrhagic Fever (DHF) | |
| Tropical Travel | AIDS, Human immunodeficiency virus, lentivirus, anti-retroviral, | |
| Tropical Travel | tuberculosis | |
| Tropical Travel | Falciparum, Vivax, Ovale, Malariae, Knowlesi, Plasmodium | |
| Tropical Travel | cholera, gram-negative comma-shaped bacillus, rice water stool, John Snow Pump, V. cholerae, vibrio | |
| Tropical Travel | Entamoeba histolytica, protozoan parasite, Amoebic dysentery, Flask Shaped amoebic trophozoite, Bloody stool, | |
| Tropical Travel | shigellosis, Shigella, Enterotoxin, dysentery, | |
| Tropical Travel | Tetanus, Tetanispasmin, Clostridium tetani, lock jaw, Opisthotonus, Autonomic dysfunction, toxoid | |
| Tropical Travel | Rabies Immunoglobulin | |
| Tropical Travel | Koplik, measles, rash, rubeola, Morbilivirus, | |
| Trauma | permissive hypotension, MBA, MVA, widened mediastinum, pleural effusion, ICC | |
| Trauma | knife, penetrating chest wound | |
| Trauma | knife, penetrating chest wound | |
| Trauma | TBSA %, Burns Wound Assessment, Total Body Surface Area | |
| Trauma | Arterial pressure index (API), DPI (Doppler Pressure Index), Arterial Brachial Index or Ankle Brachial Index (ABI) | |
| Trauma | crush injury, degloving, deglove, amputation | |
| Trauma | hip dislocation, Allis reduction, pelvic fracture | |
| Trauma | Pelvis fracture, stabilization, stabilisation, | |
| Trauma | pelvic stabilization, Pelvis fracture, stabilisation, Pre-peritoneal packing | |
| Trauma | massive transfusion protocol, Recombinant Factor VIIa, Thromboelastography (TEG) | |
| Trauma | Critical bleeding, hemorrhagic shock, haemorrhagic shock, lethal triad, acute coagulopathy of trauma | |
| Trauma | penetrating abdominal trauma | |
| Trauma | ||
| Trauma | penetrating chest trauma wound, stab, | |
| Trauma | Right Main Bronchus, RMB, Tracheostomy, Tooth, foreign Body | |
| Trauma | Lobar collapse, aspiration, blood clot | |
| Trauma | ||
| Trauma | Traumatic rupture of the diaphragm with strangulation of viscera | |
| Trauma | eschar, burns, full thickness, | |
| Trauma | supine hypotension syndrome | |
| Trauma | ||
| Trauma | iPhone | |
| Trauma | oleoma, lipogranuloma, | |
| Trauma | oral commissure, lingual artery hemorrhage, | |
| Trauma | polymer fume fever, dielectric heating, super-heating, thermal injury | |
| Trauma | DRE, Digital rectal exam examination trauma | |
| Trauma | Injury Severity score, ISS, golden hour, seatbelt sign | |
| Trauma | primary secondary survey | |
| Trauma | extradural hemorrhage, EDH, Monro-Kellie | |
| Trauma | ||
| Trauma | ||
| Trauma | ||
| Trauma | ||
| Trauma | ||
| Trauma | GU, trauma, penis, penile, urethra, bladder, rupture | |
| Pulmonary | swine flu, pneumomediastinum, CXR | |
| Pulmonary | Thrombocytopenia, antiphospholipid syndrome | |
| Pulmonary | Hermann Boerhaave, Boerhaave syndrome, esophagus rupture, oesophagus | |
| Pulmonary | ||
| Pulmonary | pneumococcal pneumonia, HIV, bronchoscope, anatomy, RMB | |
| Pulmonary | subcutaneous emphysema, FLAAARDS, | |
| Pulmonary | respiratory acidosis, hypercapnoea | |
| Pulmonary | hypersensivity pneumonitis, diffuse alveolar haemorrhage, alveolar infiltrates | |
| Pulmonary | Lung collapse, recruitment maneuver, bronchoscopy | |
| Pulmonary | Vocal cord dysfunction, VCD, paradoxical vocal cord motion, PVCM, posterior chinking | |
| Pulmonary | pneumococcus, Streptococcus pneumoniae, penicillin-resistant | |
| Pulmonary | DOPES, | |
| Pulmonary | asthma | |
| Pulmonary | dyssynchrony, mechanical ventilation, PEEP, Plateau pressure | |
| Pulmonary | pneumomediastinum, tracheostomy, trachy, complication | |
| Pulmonary | PERC rule, D-Dimer, Pulmonary Embolism Rule-out Criteria, HAD CLOTS, | |
| Pulmonary | AMS, acute mountain sickness, high altitude, High-altitude cerebral edema, HACE, HAPE, High-altitude pulmonary edema | |
| Pulmonary | ||
| Resus | Pulseless electrical activity, PEA | |
| Resus | intraosseous access, EZ-IO, | |
| Resus | ||
| Resus | Rocuronium, suxamethonium, succinylcholine, non-depolarising muscle relaxant, sugammadex, safe apnoea time | |
| Resus | FEAST, trial, research, pediatric, fluid resuscitation | |
| Resus | ||
| Resus | ||
| Resus | ||
| Resus | ICC, intercostal | |
| Resus | Mechanical ventilation | |
| Oncology | SVC obstruction | |
| Oncology | Tumour lysis syndrome, Tumor lysis syndrome | |
| Oncology | lung metastases braine mets testicular cancer BEP chemotherapy, Cannonball metastases | |
| Oncology | re-expansion pulmonary oedema edema | |
| Metabolic | abdominal aortic aneurysm, AAA, rupture, CT, rhabdomyolysis, creatine kinase | |
| Metabolic | hypokalemia, hypokalaemia, periodic paralysis, u wave | |
| Metabolic | CATMUDPILES, OGRE, NAGMA, HAGMA, USED CARP, hyperchloraemic metabolic acidosis | |
| Metabolic | anion gap, pyroglutamic acidemia, HAGMA, high-anion gap, high anion, 5-oxoprolinemia, γ-glutamyl cycle, staph aureus, sepsis | |
| Metabolic | HAGMA, high-anion gap, high anion, hypernatraemia, hypernatremia | |
| Metabolic | hypokalaemia, hypokalemia, potassium, systemic bromism, coke, pepsi, coca-cola | |
| Metabolic | CATMUDPILES, renal failure, HAGMA, LTKR | |
| Metabolic | ||
| Metabolic | acute hepatitis, arterial blood gas, fulminant hepatic failure, lactic acidosis, lactic acidosis with hypoglycaemia, metabolic acidosis, metabolic muddle | |
| Metabolic | hyperammonaemia, hyperammonemia | |
| Metabolic | Hyponatraemia, hypertonic saline, ultramarathon, runner, EAH, pontine myelinoysis | |
| Metabolic | Hyponatraemia, hypertonic saline, pontine myelinoysis, Osmolality, desmopressin, SIADH, syndrome of inappropriate anti-diuretic hormone secretion | |
| Gastrointestinal | Appendagitis, Epiploic, Abdominal pain, CT abdomen | |
| Gastrointestinal | CT abdomen, Small bowel obstruction, SBO | |
| Gastrointestinal | cathine, cathione, khat, hepatitis, cathionine | |
| Gastrointestinal | rectal foreign body, FB | |
| Gastrointestinal | abdominal compartment syndrome, intra-abdominal pressure, intra-abdominal hypertension, IAH, ACS | |
| Hematology | fibrinolytic, VTE, Wells, PERC | |
| Hematology | factor VIIa, rFVIIa, novoseven | |
| Hematology | Critical Bleeding, Massive Transfusion, Tranexamic Acid, TxA, MTP | |
| Hematology | Dyshemoglobinemia, Acute myeloid leukemia, AML | |
| Immunological | angiodema, angioedema, lip sweliing | |
| Immunological | frusemide, furosemide, lasix, sulfa, | |
| Immunological | wegener, GPA, granulomatosis, palpable purpura | |
| Obstetric | amniotic fluid embolism, DIC, obstetric complication, disseminated intravascular coagulation, schistocytes, | |
| Microbial | CSF, Meningococcal meningitis, | |
| Microbial | fulminant bacterial pneumonia, septic shock, Pneumococcus, Streptococcus pyogenes, urinary pneumococcal antigen, | |
| Microbial | Legionella, community acquired pneumonia | |
| Microbial | Staphylococcal toxic shock syndrome, Toxic-shock syndrome | |
| Microbial | ||
| Microbial | ||
| Microbial | Norovirus | |
| Toxicology | Coma, similie, metaphor, flashcard, toxidromes, anticholinergic, cholinergic, PHAILS, OTIS CAMPBELL, PACED, FAST, COOLS, CT SCAN | |
| Neurology | HIV, Mass effect, CNS lesion, Brain lesion | |
| Neurology | pancoast, argyll robertson, holmes-adie, coma, pinpoint, pin-point, horner syndrome | |
| Neurology | rule of 4, rules of four, brainstem, weber syndrome, wallenberg | |
| Neurology | rule of 4, rules of four, brainstem, Nothnagel syndrome, benedikt, claude, | |
| Neurology | ||
| Neurology | ||
| Neurology | ||
| Neurology | Unilateral internuclear ophthalmoplegia, medial longitudinal fasciculus, MLF, INO, one-and-a-half syndrome | |
| Neurology | GSW, gunshot wound, bullet, TBI, Codman ICP monitor, Trans-cranial doppler, Near-infrared spectroscopy, NIRS, cerebral microdialysis catheter | |
| Neurology | BPPV, Benign Paroxysmal Positional Vertigo, Dix-Hallpike test, semont, epley, dix hallpike, brandt-daroff | |
| Neurology | Anti-NMDA Receptor Encephalitis, teratoma | |
| To err is human | cognitive error, bias, entrapment | |
| To err is human | rule of thumb, heuristic, satisficing, cognitive bias, metacognition | |
| To err is human | | Anchoring Bias, confirmation, satisficing, clustering bias |
| Cardiology | ||
| Paediatric | pediatric |
Compendium of Clinical Cases
LITFL Top 100 Self Assessment Quizzes
- - Google Chrome
Intended for healthcare professionals
- My email alerts
- BMA member login
- Username * Password * Forgot your log in details? Need to activate BMA Member Log In Log in via OpenAthens Log in via your institution

Search form
- Advanced search
- Search responses
- Search blogs
- How to present patient...
How to present patient cases
- Related content
- Peer review
- Mary Ni Lochlainn , foundation year 2 doctor 1 ,
- Ibrahim Balogun , healthcare of older people/stroke medicine consultant 1
- 1 East Kent Foundation Trust, UK
A guide on how to structure a case presentation
This article contains...
-History of presenting problem
-Medical and surgical history
-Drugs, including allergies to drugs
-Family history
-Social history
-Review of systems
-Findings on examination, including vital signs and observations
-Differential diagnosis/impression
-Investigations
-Management
Presenting patient cases is a key part of everyday clinical practice. A well delivered presentation has the potential to facilitate patient care and improve efficiency on ward rounds, as well as a means of teaching and assessing clinical competence. 1
The purpose of a case presentation is to communicate your diagnostic reasoning to the listener, so that he or she has a clear picture of the patient’s condition and further management can be planned accordingly. 2 To give a high quality presentation you need to take a thorough history. Consultants make decisions about patient care based on information presented to them by junior members of the team, so the importance of accurately presenting your patient cannot be overemphasised.
As a medical student, you are likely to be asked to present in numerous settings. A formal case presentation may take place at a teaching session or even at a conference or scientific meeting. These presentations are usually thorough and have an accompanying PowerPoint presentation or poster. More often, case presentations take place on the wards or over the phone and tend to be brief, using only memory or short, handwritten notes as an aid.
Everyone has their own presenting style, and the context of the presentation will determine how much detail you need to put in. You should anticipate what information your senior colleagues will need to know about the patient’s history and the care he or she has received since admission, to enable them to make further management decisions. In this article, I use a fictitious case to …
Log in using your username and password
BMA Member Log In
If you have a subscription to The BMJ, log in:
- Need to activate
- Log in via institution
- Log in via OpenAthens
Log in through your institution
Subscribe from £184 *.
Subscribe and get access to all BMJ articles, and much more.
* For online subscription
Access this article for 1 day for: £50 / $60/ €56 ( excludes VAT )
You can download a PDF version for your personal record.
Buy this article
- Intranet Login
- Clinical Services
- Patient Care
- Department Videos
- Job Opportunities
- Diversity and Inclusion
- Administration
- Student Fellows
- Recent Graduates
- Affiliated Hospitals
- Pepper Talk
- Residency Matters
- Roth Report
- Application
- AP/Neuropathology
- Physician-Scientist Pathway
- Conferences
- Teaching Opportunities
- Salary & Benefits
- Living in Philly
- Breast Pathology
- Cellular Therapy
- Clinical Chemistry
- Clinical Informatics
- Clinical Microbiology
- Cytopathology
- GI/Hepatic Pathology
- Hematopathology
- Molecular Genetic Pathology
- Neuropathology
- Soft Tissue/Bone Pathology
- Surgical Pathology
- Transfusion Medicine
- Student Fellowship
- Graduate Studies
- Medical School
- Case Studies
- PennLab Summer Internship
- Cancer and Immunobiology
- Neuropathology and Neurodegeneration
- Diagnostic Innovation
- Info for Investigators
- Research Labs
- Centers and Institutes
- Clinical Cell and Vaccine Production Facility
- CRISPR/Cas9 Mouse Targeting Core
- Penn Cytomics and Cell Sorting Resource Laboratory
- Human Immunology
- Tumor Tissue/ Biospecimen Bank
- Publications
- Electron Microscopy
- Immunohistochemistry
- Medical Pathology
- Clinical Chemistry Core Lab
- Endocrinology
- Coagulation
- Immunology and HLA
- Microbiology
- Point-of-Care Testing
- Reference Testing
- Apheresis Unit
- Flow Cytometry
- Center for Personalized Diagnostics
- Clinical Cancer Cytogenetics Laboratory
- Molecular Pathology
- Rittenhouse Molecular Laboratory
- Test, Biospecimen, & Research Requests
- Clinical Assay Development
- Molecular Genetic Pathology Fellowship
- AP Consults
- Immunotherapy
- Meet Your Pathologist
- Making an Appointment
- Clinical Trials
- Patient Resources
CASE STUDIES
- All case studies
Filter by: Clear Filters
This page offers a collection of interesting cases from the Penn Department of Pathology and Laboratory Medicine that are available to download as PDFs. To view specific case studies by organ system or subspecialty, use the filter checkboxes in the left sidebar.
56-year-old woman with 3.5 cm large right nasal mass, resected after 2 nondiagnostic biopsies
33-year-old man with complex ethmoid sinus mass and imaging concerning for a sinonasal malignancy, 34-year-old man with aml with sudden onset of headache and fever, 36-year-old woman presenting with hemoptysis, 65-year-old man with 2.3 cm right lower thyroid nodule, 56-year-old female presenting with a 3-month history of abdominal pain, 55-year-old male presenting with back pain, 62-year-old man with a right posterior nasal mass, 65-year-old female with a mass involving the maxillary sinus, 74-year-old female with an extradural tumor compressing the right frontal lobe, 35-year-old man with chronic rhinosinusitis and nasal septal perforation, 54-year-old man with a 3.6 cm right neck mass, 21-year-old man with asthma, chronic sinusitis, polyps, headache and proptosis, 57-year-old woman with a renal mass, 63-year-old man with history of iv drug use, 72-year-old man with polypoid esophageal mass, 20-year-old woman with 3 cm mass in the tail of pancreas, 40-year-old man with increasing frequency of hypoglycemic spells, 52-year-old woman with transient symptomatic hyperthyroidism, stay connected.
- Therapeutic Pathology
- Anatomic Pathology
- Lab Medicine Advances
- Science Breakthroughs
Sign up for the Department Newsletter:
Thank you for subscribing!
Department of Pathology and Laboratory Medicine
Perelman School of Medicine at the University of Pennsylvania 3400 Spruce St. Philadelphia, PA 19104-4238
© 2024 Trustees of the University of Pennsylvania
- About This Site


Clinical Cases
A collection of interactive clinical case scenarios aligned with UK Medical Licensing Assessment (MLA) presentations . Each scenario allows you to work through history taking , investigations , diagnosis and management . You might also be interested in our bank of 1000+ OSCE Stations .

Painful Hand – OSCE Case

Dizziness and Nausea – OSCE Case

Paediatric Rash – OSCE Case
Pain on Inspiration – OSCE Case

Vaginal Discharge – OSCE Case
Cough, Chest Pain and Fever – OSCE Case

Breathlessness and Acute Rash – OSCE Case
Wheezy Child – OSCE Case

Episode of Facial Weakness – OSCE Case

Lethargic Child – OSCE Case

Headache and Neck Pain – OSCE Case
Acute abdominal pain – osce case.

Other pages
- Product Bundles 🎉
- Join the Team 🙌
- Institutional Licence 📚
- OSCE Station Creator Tool 🩺
- Create and Share Flashcards 🗂️
- OSCE Group Chat 💬
- Newsletter 📰
- Advertise With Us
Join the community
Masks Strongly Recommended but Not Required in Maryland, Starting Immediately
Due to the downward trend in respiratory viruses in Maryland, masking is no longer required but remains strongly recommended in Johns Hopkins Medicine clinical locations in Maryland. Read more .
- Vaccines
- Masking Guidelines
- Visitor Guidelines
Center for Bloodless Medicine and Surgery
Case studies.
In this section we report a variety of clinical cases in an effort to share our experience with healthcare providers and with patients who may benefit. The cases are categorized into a wide variety of different medical and surgical specialties listed under subheadings.
The main objective is to share our knowledge and experience for the educational value, and to describe the methods of clinical care that worked for our patients. These methods, however, may not be appropriate for all patients, as each patient and each condition may have unique and special issues to consider.
The cases presented here range from commonly seen routine, medical conditions to rare or uniquely challenging medical conditions and treatment modalities that may not be appropriate for smaller medical centers without special equipment or experience.
Case Study Subjects
- Cardiac Surgery
- Gastroenterology
- Hepatobiliary Surgery
- Neurosurgery
- Obstetrics and Gynecology
- Orthopedic Surgery
- Rescue Therapy
- Surgical Oncology
- Vascular Surgery
Additional Resources for Clinicians
This collection of research papers features information on Medicine and Surgery, Pediatrics, Diseases and Conditions and Bioethics and Law.
- Find your Librarian Connect with a librarian with expertise in your research subject area
- Toolkits Curated information resources grouped by discipline
- Research Guides Librarian-recommended resources, research tips, and how-to guides
- Faculty & Staff Guide Quick links for faculty and staff
- UW Libraries Search Online catalog for materials held by UW and Summit Libraries
- Books Health sciences print and electronic books, sorted by subject
- Databases Indexed collections of full-text articles, citations and other research materials
- Journals Searchable list of health sciences journal titles held at UW
- Videos Educational and procedural videos, sorted by subject
- Request Library Resources Request articles, books, and media for pickup or delivery
- Your Library Account Portal for renewing borrowed materials, viewing item request updates, and paying fines
- Course Reserves Library materials reserved for your classes
- Interlibrary Loan Receive scanned print articles delivered via email and borrow items not held at UW
- Off-Campus Access Instruction for connecting to UW Libraries resources while away from campus
- Study Rooms Reservable rooms for individual and small-group studying
- Li Lu Library Open library space located in the UW Health Sciences Education Building
- Meeting and Event Spaces Larger spaces available for fee-based reservations
- Learning Commons Lab Testing facility with drop-in computer access (closed Apr-Dec 2024)
- Accessibility at HSL We are committed to providing equal access to library collections, services, and facilities for all library users
- Collection Guidelines Guidelines for resource purchases by HSL
- FAQ Commonly asked questions from HSL users
- News Announcements, upcoming events, and new resources
- Staff Directory Current HSL, NNLM Region 5, and HEALWA staff
- UW Libraries Policies Policies governing use of library resources, space, and services
Evidence-based Complementary and Alternative Medicine (CAM)
- CAM Welcome
- Find Articles/Databases
Case Study Scenario
Search databases for evidence-based information on fenugreek used for diabetes..
- Citing Sources
Mrs. Hernandez, a 45 year-old Spanish speaking woman, has been recently diagnosed with Diabetes Type 2.
- She is admitted to UWMC with uncontrolled hypertension
- She takes fenugreek
- Prescribed medications include Lisinopril, Clonidine, and Metformin
Search the databases below for evidence-based information on fenugreek used for diabetes.
For more information on each database, see Find Articles/Databases.
Filter to Article Type
Case Study example:
- In the PubMed Search box, type: (fenugreek OR trigonella) AND diabetes
- Then filter from left sidebar to: English, Human
- For evidence-based articles, filter by Article Type to: Randomized controlled trial, meta-analysis, clinical trial
Filter to Subject
- In the PubMed Search box, type: Diabetes Mellitus, Type 2/th
- Then filter to: Complementary Medicine and/or Dietary Supplements under Subjects.
- This strategy will produce results about alternative treatments for diabetes.
- For evidence-based articles, filter under Article Type to: Randomized controlled trial, meta-analysis, clinical trial
Search PubMed Clinical Queries
Search By Clinical Study Category
- Type: (fenugreek OR trigonella) AND diabetes
- Select Therapy and Narrow (specific) search.
Find Systematic Reviews

Limit to Research Article
- Type in the Search box: (fenugreek OR trigonella) and diabetes
- Click on Research Article under Limit your Results.
Limit by Publication Type
- Type in Search box: (fenugreek OR trigonella) and diabetes
- In Limit your Results, select Clinical Trial or Systematic Review or Research under Publication Type.
Limit by Clinical Query
- Select any of the choices under Clinical Queries limits.
Limit by Journal Subset
- In Limit your Results, select Alternative/Complementary Therapies under Journal Subset.
Limit by Evidence-Based Practice
- Click on Evidence-Based Practice under Limit your Results.
- In the Search box, type: fenugreek AND diabetes
- In this case, there are no systematic reviews on this topic. You do retrieve, however, some citations to clinical trials.
- Type fenugreek in Search box and then click on Professional Monograph: Fenugreek.
- Note the diabetes evidence grade under Clinical Bottom Line/Effectiveness.
IBIDS (International Bibliographic Information on Dietary Supplements)
- Type in Search box: +fenugreek +diabetes
- Change drop-down menu to Peer-reviewed References instead of All IBIDS References .
- look under Treatment>Diet for use of fenugreek
- OR, click on Search Within Text link and follow directions to find fenugreek
- Type in Search box: fenugreek and diabetes and ('clinical trial'/exp or 'controlled study'/exp or 'controlled clinical trial' or 'randomized controlled trial')
- << Previous: Find Articles/Databases
- Next: eBooks >>
- Last Updated: Jul 9, 2024 10:24 AM
- URL: https://guides.lib.uw.edu/hsl/cam
- Introduction
- Conclusions
- Article Information
A, Distribution of reports of SSNHL by week of vaccination and time to onset after vaccination. The rate of SSNHL reports per 100 000 vaccine doses (blue line) is overlaid. B, Mean age of people reporting SSNHL to VAERS according to the time period reported who met the definition of probable SSNHL (n = 555). Note that the weekly time periods are identical in A and B. VAERS indicates Vaccine Adverse Events Reporting System.
A, Includes 555 cases reported to the VAERS database that met the definition of probable SSNHL during the period examined. B, Includes 21 patients in multi-institutional case series. The x-axis extends to only 15 days after vaccination because no new cases were observed after day 15. VAERS indicates Vaccine Adverse Events Reporting System.
eTable 1. Representative Examples of VAERS Incident Reports Meeting Criteria for Probable SSNHL Compared With Those Unlikely to Represent True SSNHL
eTable 2. Rate of SSNHL Reports in VAERS by Vaccine Manufacturer
eFigure 1. Scattergrams of Pretreatment and Posttreatment Hearing Results
eFigure 2. Audiogram Revealing Unilateral Sensorineural Hearing Loss Occurring 14 Days After Each of 2 COVID-19 Vaccine Doses in 1 Patient
eFigure 3. Number of People in the US With at Least 1 COVID-19 Vaccine Dose According to Age Group at 3 Points During the Initial COVID-19 Vaccination Rollout
- Studies Examine Risk of Hearing Loss After COVID-19 Vaccination JAMA News From the JAMA Network May 3, 2022 Anita Slomski
- Association Between COVID-19 Vaccine and Sudden Sensorineural Hearing Loss JAMA Otolaryngology–Head & Neck Surgery Original Investigation April 1, 2022 This cohort study of patients in Israel assesses whether the BNT162b2 messenger RNA COVID-19 vaccine is associated with sudden sensorineural hearing loss. Yoav Yanir, MD; Ilana Doweck, MD; Rana Shibli, MD, MPH; Ronza Najjar-Debbiny, MD; Walid Saliba, MD, MPH
- Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited—An Ongoing Conversation—Reply JAMA Otolaryngology–Head & Neck Surgery Comment & Response August 1, 2022 Eric J. Formeister, MD, MS; Daniel Q. Sun, MD
- Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited—Reply JAMA Otolaryngology–Head & Neck Surgery Comment & Response August 1, 2022 Yoav Yanir, MD, MPH; Ilana Doweck, MD; Walid Saliba, MD, MPH
- Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited JAMA Otolaryngology–Head & Neck Surgery Comment & Response August 1, 2022 Selena E. Briggs, MD, PhD, MBA; Michael J. Brenner, MD; Sujana S. Chandrasekhar, MD
- Error in Data JAMA Otolaryngology–Head & Neck Surgery Correction November 1, 2022
- Hearing Loss After COVID-19 Vaccination Does Not Outweigh Vaccine Benefit JAMA Otolaryngology–Head & Neck Surgery Invited Commentary April 1, 2022 Angela K. Ulrich, PhD, MPH; Maria E. Sundaram, PhD, MSPH; Michael T. Osterholm, PhD, MPH
See More About
Select your interests.
Customize your JAMA Network experience by selecting one or more topics from the list below.
- Academic Medicine
- Acid Base, Electrolytes, Fluids
- Allergy and Clinical Immunology
- American Indian or Alaska Natives
- Anesthesiology
- Anticoagulation
- Art and Images in Psychiatry
- Artificial Intelligence
- Assisted Reproduction
- Bleeding and Transfusion
- Caring for the Critically Ill Patient
- Challenges in Clinical Electrocardiography
- Climate and Health
- Climate Change
- Clinical Challenge
- Clinical Decision Support
- Clinical Implications of Basic Neuroscience
- Clinical Pharmacy and Pharmacology
- Complementary and Alternative Medicine
- Consensus Statements
- Coronavirus (COVID-19)
- Critical Care Medicine
- Cultural Competency
- Dental Medicine
- Dermatology
- Diabetes and Endocrinology
- Diagnostic Test Interpretation
- Drug Development
- Electronic Health Records
- Emergency Medicine
- End of Life, Hospice, Palliative Care
- Environmental Health
- Equity, Diversity, and Inclusion
- Facial Plastic Surgery
- Gastroenterology and Hepatology
- Genetics and Genomics
- Genomics and Precision Health
- Global Health
- Guide to Statistics and Methods
- Hair Disorders
- Health Care Delivery Models
- Health Care Economics, Insurance, Payment
- Health Care Quality
- Health Care Reform
- Health Care Safety
- Health Care Workforce
- Health Disparities
- Health Inequities
- Health Policy
- Health Systems Science
- History of Medicine
- Hypertension
- Images in Neurology
- Implementation Science
- Infectious Diseases
- Innovations in Health Care Delivery
- JAMA Infographic
- Law and Medicine
- Leading Change
- Less is More
- LGBTQIA Medicine
- Lifestyle Behaviors
- Medical Coding
- Medical Devices and Equipment
- Medical Education
- Medical Education and Training
- Medical Journals and Publishing
- Mobile Health and Telemedicine
- Narrative Medicine
- Neuroscience and Psychiatry
- Notable Notes
- Nutrition, Obesity, Exercise
- Obstetrics and Gynecology
- Occupational Health
- Ophthalmology
- Orthopedics
- Otolaryngology
- Pain Medicine
- Palliative Care
- Pathology and Laboratory Medicine
- Patient Care
- Patient Information
- Performance Improvement
- Performance Measures
- Perioperative Care and Consultation
- Pharmacoeconomics
- Pharmacoepidemiology
- Pharmacogenetics
- Pharmacy and Clinical Pharmacology
- Physical Medicine and Rehabilitation
- Physical Therapy
- Physician Leadership
- Population Health
- Primary Care
- Professional Well-being
- Professionalism
- Psychiatry and Behavioral Health
- Public Health
- Pulmonary Medicine
- Regulatory Agencies
- Reproductive Health
- Research, Methods, Statistics
- Resuscitation
- Rheumatology
- Risk Management
- Scientific Discovery and the Future of Medicine
- Shared Decision Making and Communication
- Sleep Medicine
- Sports Medicine
- Stem Cell Transplantation
- Substance Use and Addiction Medicine
- Surgical Innovation
- Surgical Pearls
- Teachable Moment
- Technology and Finance
- The Art of JAMA
- The Arts and Medicine
- The Rational Clinical Examination
- Tobacco and e-Cigarettes
- Translational Medicine
- Trauma and Injury
- Treatment Adherence
- Ultrasonography
- Users' Guide to the Medical Literature
- Vaccination
- Venous Thromboembolism
- Veterans Health
- Women's Health
- Workflow and Process
- Wound Care, Infection, Healing
Others Also Liked
- Download PDF
- X Facebook More LinkedIn
- CME & MOC
Formeister EJ , Wu MJ , Chari DA, et al. Assessment of Sudden Sensorineural Hearing Loss After COVID-19 Vaccination. JAMA Otolaryngol Head Neck Surg. 2022;148(4):307–315. doi:10.1001/jamaoto.2021.4414
Manage citations:
© 2024
- Permissions
Assessment of Sudden Sensorineural Hearing Loss After COVID-19 Vaccination
- 1 Department of Otolaryngology–Head and Neck Surgery, Johns Hopkins University School of Medicine, Baltimore, Maryland
- 2 Massachusetts Eye and Ear Infirmary, Harvard Medical School, Boston, Massachusetts
- 3 Department of Otolaryngology–Head and Neck Surgery, University of Massachusetts School of Medicine, Worcester, Massachusetts
- 4 Anne Arundel Ear, Nose, and Throat Surgery, Annapolis, Maryland
- Invited Commentary Hearing Loss After COVID-19 Vaccination Does Not Outweigh Vaccine Benefit Angela K. Ulrich, PhD, MPH; Maria E. Sundaram, PhD, MSPH; Michael T. Osterholm, PhD, MPH JAMA Otolaryngology–Head & Neck Surgery
- News From the JAMA Network Studies Examine Risk of Hearing Loss After COVID-19 Vaccination Anita Slomski JAMA
- Original Investigation Association Between COVID-19 Vaccine and Sudden Sensorineural Hearing Loss Yoav Yanir, MD; Ilana Doweck, MD; Rana Shibli, MD, MPH; Ronza Najjar-Debbiny, MD; Walid Saliba, MD, MPH JAMA Otolaryngology–Head & Neck Surgery
- Comment & Response Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited—An Ongoing Conversation—Reply Eric J. Formeister, MD, MS; Daniel Q. Sun, MD JAMA Otolaryngology–Head & Neck Surgery
- Comment & Response Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited—Reply Yoav Yanir, MD, MPH; Ilana Doweck, MD; Walid Saliba, MD, MPH JAMA Otolaryngology–Head & Neck Surgery
- Comment & Response Sudden Sensorineural Hearing Loss and COVID-19 Vaccination Revisited Selena E. Briggs, MD, PhD, MBA; Michael J. Brenner, MD; Sujana S. Chandrasekhar, MD JAMA Otolaryngology–Head & Neck Surgery
- Correction Error in Data JAMA Otolaryngology–Head & Neck Surgery
Question Is COVID-19 vaccination associated with sudden sensorineural hearing loss (SSNHL)?
Findings In this cross-sectional study and case series involving 555 cases of SSNHL among adults reported to the Centers for Disease Control and Prevention Vaccine Adverse Events Reporting System, no increase in the rate of hearing loss after COVID-19 vaccination was found compared with the incidence in the general population. Assessment of 21 adult patients who presented to tertiary care centers with SSNHL after COVID-19 vaccination did not reveal any apparent associations with respect to clinical or demographic factors.
Meaning These results suggest that there is no association between vaccination and the development of SSNHL among adults who received a COVID-19 vaccine.
Importance Emerging reports of sudden sensorineural hearing loss (SSNHL) after COVID-19 vaccination within the otolaryngological community and the public have raised concern about a possible association between COVID-19 vaccination and the development of SSNHL.
Objective To examine the potential association between COVID-19 vaccination and SSNHL.
Design, Setting, and Participants This cross-sectional study and case series involved an up-to-date population-based analysis of 555 incident reports of probable SSNHL in the Centers for Disease Control and Prevention Vaccine Adverse Events Reporting System (VAERS) over the first 7 months of the US vaccination campaign (December 14, 2020, through July 16, 2021). In addition, data from a multi-institutional retrospective case series of 21 patients who developed SSNHL after COVID-19 vaccination were analyzed. The study included all adults experiencing SSNHL within 3 weeks of COVID-19 vaccination who submitted reports to VAERS and consecutive adult patients presenting to 2 tertiary care centers and 1 community practice in the US who were diagnosed with SSNHL within 3 weeks of COVID-19 vaccination.
Exposures Receipt of a COVID-19 vaccine produced by any of the 3 vaccine manufacturers (Pfizer-BioNTech, Moderna, or Janssen/Johnson & Johnson) used in the US.
Main Outcomes and Measures Incidence of reports of SSNHL after COVID-19 vaccination recorded in VAERS and clinical characteristics of adult patients presenting with SSNHL after COVID-19 vaccination.
Results A total of 555 incident reports in VAERS (mean patient age, 54 years [range, 15-93 years]; 305 women [55.0%]; data on race and ethnicity not available in VAERS) met the definition of probable SSNHL (mean time to onset, 6 days [range, 0-21 days]) over the period investigated, representing an annualized incidence estimate of 0.6 to 28.0 cases of SSNHL per 100 000 people per year. The rate of incident reports of SSNHL was similar across all 3 vaccine manufacturers (0.16 cases per 100 000 doses for both Pfizer-BioNTech and Moderna vaccines, and 0.22 cases per 100 000 doses for Janssen/Johnson & Johnson vaccine). The case series included 21 patients (mean age, 61 years [range, 23-92 years]; 13 women [61.9%]) with SSNHL, with a mean time to onset of 6 days (range, 0-15 days). Patients were heterogeneous with respect to clinical and demographic characteristics. Preexisting autoimmune disease was present in 6 patients (28.6%). Of the 14 patients with posttreatment audiometric data, 8 (57.1%) experienced improvement after receiving treatment. One patient experienced SSNHL 14 days after receiving each dose of the Pfizer-BioNTech vaccine.
Conclusions and Relevance In this cross-sectional study, findings from an updated analysis of VAERS data and a case series of patients who experienced SSNHL after COVID-19 vaccination did not suggest an association between COVID-19 vaccination and an increased incidence of hearing loss compared with the expected incidence in the general population.
Anecdotal reports of sudden sensorineural hearing loss (SSNHL) occurring after COVID-19 vaccination have emerged in otolaryngologic professional societies and have important public health implications. Tinnitus, dizziness, and vertigo have also been reported within 2 weeks of vaccination in a recent single-institution case series. 1 Otolaryngologists encounter increasing challenges to promoting public health conduct recommended during the pandemic when they are counseling and evaluating patients who have developed SSNHL and reported a temporal association with COVID-19 vaccination.
Other large-scale vaccination campaigns, such as those for the measles-mumps-rubella and influenza vaccines, have previously been investigated after anecdotal reports of SSNHL emerged among vaccinated individuals. In each campaign, epidemiologic studies 2 , 3 did not show an association between vaccination and SSNHL. Although data from similar epidemiologic studies are not yet available for COVID-19 vaccination, a preliminary analysis 4 of incident reports from the Centers for Disease Control and Prevention (CDC) Vaccine Adverse Events Reporting System (VAERS) during the early phase of public COVID-19 vaccination did not identify an association between vaccination and SSNHL. However, as vaccination campaigns have expanded across the US and currently include vaccines from 3 manufacturers (Pfizer-BioNTech [BNT162b2], Moderna [mRNA-1273], and Janssen/Johnson & Johnson [Ad26.COV2.S]), questions remain regarding whether an association exists between COVID-19 vaccination and SSNHL. In addition, VAERS does not provide detailed patient-level clinical data that may be valuable in evaluating specific patient cofactors.
The purposes of the present study were to (1) extend the preliminary incidence estimate of SSNHL after COVID-19 vaccination 4 to the present phase of vaccination across 3 manufacturers and (2) examine whether emerging patterns in VAERS incident reports suggest an association between COVID-19 vaccination and SSNHL. In addition, we sought to augment this public database evaluation with an in-depth analysis of clinical characteristics among a multi-institutional series of patients who presented with confirmed SSNHL after COVID-19 vaccination.
This study was approved by the institutional review boards of Johns Hopkins University School of Medicine and the Massachusetts Eye and Ear Infirmary/Harvard Medical School. Because the VAERS records review obtained data from a publicly available deidentified database, this portion of the study was deemed exempt from review; similarly, the case series was deemed exempt because the patients’ files did not contain identifiable data.
The study was performed in 2 phases. In the first phase, VAERS was queried for reports of SSNHL after COVID-19 vaccination between December 14, 2020, and July 16, 2021. Cases deemed to represent probable SSNHL were compiled for analysis using previously dfescribed methods. 4 In brief, the search terms sudden hearing loss , deafness , deafness neurosensory , deafness unilateral , deafness bilateral , and hypoacusis were selected as adverse events (AEs) for data extraction. Because multiple symptoms could be selected for each incident report, deduplication was performed to ensure there was only 1 unique VAERS identification number per report. Narratives and laboratory data from all reports were reviewed to assess the likelihood of a report representing probable SSNHL. Inclusion criteria for probable SSNHL consisted of a temporal association with COVID-19 vaccination (defined as onset within 21 days after vaccination) and a high credibility of reporting. A report was deemed credible if it could demonstrate at least 1 of the following: (1) reference to an audiographic test result confirming hearing loss, (2) evaluation by an otolaryngologist, audiologist, or other physician resulting in a diagnosis of sudden hearing loss, or (3) evaluation by an otolaryngologist resulting in treatment with systemic steroid or intratympanic steroid medications, performance of magnetic resonance imaging, or any combination thereof. Incident reports were excluded if they did not reference evaluation by a physician or audiologist leading to a diagnosis of hearing loss, did not contain details within the report or laboratory results section to indicate that a diagnosis of sudden hearing loss was provided (eg, no mention of audiologic testing, no receipt of systemic or intratympanic steroid medications, or no magnetic resonance imaging scan), or indicated that hearing loss onset occurred more than 21 days after vaccination. In addition, reports that described the discovery of an alternative origin for hearing loss (eg, vestibular schwannoma or stroke) were excluded. Examples of narratives and their classifications are shown in eTable 1 in the Supplement .
The number of vaccine doses administered in the US during the study period was obtained from the CDC. 5 An incidence estimate of probable SSNHL on a per-person basis during the study period was obtained and annualized. To account for intrinsic uncertainties, such as the number of unique individuals receiving a vaccine relative to the number of doses administered, the true case numbers of SSNHL based on VAERS incident reports, and potential underreporting bias in VAERS, we conducted a sensitivity analysis that adjusted these assumptions to achieve a range estimate of the incidence of SSNHL. The maximum incidence estimate was produced based on the assumptions that (1) all reports submitted to VAERS represented true cases of SSNHL (eTable 1 in the Supplement ); (2) the number of reports submitted to VAERS was likely subject to a 50% underreporting bias based on previous studies of VAERS sensitivity for rare AEs, such as Guillain-Barré syndrome and anaphylaxis 6 ; and (3) each vaccinated individual received 2 doses, resulting in the smallest possible population size given the number of vaccine doses administered (ie, the highest possible incidence).
Because VAERS reports are unverified and lack detailed clinical data, 6 an in-depth record review of a multi-institutional consecutive series of all adult patients with audiometrically confirmed SSNHL after COVID-19 vaccination was performed in the second phase of the study. The study sites comprised 2 large academic neurotologic centers and 1 community otolaryngological practice. Cases were included if audiometrically confirmed SSNHL occurred within 3 weeks of vaccination and was contemporaneous with VAERS reports of SSNHL (ie, occurring between January 1 and June 30, 2021). Patients with a history of Ménière disease were excluded.
Reports of SSNHL were exported from VAERS into Excel software, version 16.57 (Microsoft Corporation). Simple descriptive statistics (means, ranges, and percentages) were calculated using this software for both the VAERS reports and the case series.
Between December 14, 2020, and July 16, 2021, 185 424 899 COVID-19 vaccine doses were administered in the US across the 3 manufacturers. 5 After deduplication, 2170 VAERS reports of hearing loss based on search criteria and occurring within 21 days of vaccination were extracted and compiled. In total, 555 of the 2170 reports met our definition of probable SSNHL. A total of 305 incidents (55.0%) occurred among women, and 250 incidents (45.0%) occurred among men, with a mean age of 54 years (range, 15-93 years) ( Table 1 ). Data on race and ethnicity were not available in VAERS. Overall, 305 incidents (55.0%) involved the Pfizer-BioNTech vaccine, 222 (40.0%) involved the Moderna vaccine, and 28 (5.0%) involved the Janssen/Johnson & Johnson vaccine.
A sensitivity analysis was then performed to estimate the incidence range on an annualized basis, revealing 0.6 to 28.0 cases of SSNHL per 100 000 people per year ( Table 2 ). In comparison, the annual incidence of idiopathic SSNHL was estimated to be 11 to 77 cases per 100 000 people per year, depending on age. 7 Because speculation has occurred regarding the novel lipid nanoparticle delivery vehicle and the messenger RNA (mRNA) technologies that underlie the Moderna and Pfizer-BioNTech vaccines, we next investigated whether vaccines produced by these 2 manufacturers accounted for a disproportionate number of reports of SSNHL. A total of 186.88 million doses of the Pfizer-BioNTech vaccine were administered, 136.48 million doses of the Moderna vaccine were administered, and 12.97 million doses of the Janssen/Johnson & Johnson vaccine were administered over the period examined. The VAERS reporting rate of probable SSNHL was similar across manufacturers, with 0.16 cases per 100 000 doses administered for both the Pfizer-BioNTech and Moderna vaccines, and 0.22 cases per 100 000 doses administered for the Janssen/Johnson & Johnson vaccine (eTable 2 in the Supplement ).
To further investigate whether reports of SSNHL were associated with COVID-19 vaccination, we examined the total number of reports of the condition submitted to VAERS over each weekly period from the beginning of the public vaccination campaign ( Figure 1 A). The number of submitted reports peaked in the last week of March 2021, which corresponded to the largest number of vaccine doses (16 177 521) administered during a 1-week period since the vaccination campaign began. 5 However, over each weekly period, the relative number of SSNHL reports decreased when accounting for the number of doses administered nationally, from 1.10 reports per 100 000 doses at the beginning of the campaign in December 2020 to 0.01 reports per 100 000 doses by June 2021.
Because the risk of idiopathic SSNHL is highly dependent on age, 7 we specifically examined the mean ages of patients who submitted reports of probable SSNHL, which remained relatively stable over the study period (eg, mean age, 45.9 years [range, 34.0-79.0 years] in December 2020 and 41.6 years [range, 19.0-54.0 years] in June 2021) ( Figure 1 B). We also estimated the age of the overall vaccinated population using publicly available data from the CDC 8 (eFigure 3 in the Supplement ). In the early phases of the vaccination campaign, no preponderance of older individuals (who may have been at higher risk of idiopathic SSNHL) receiving vaccine doses was apparent. In addition, in the later phases of the campaign, no preponderance of younger individuals (who may have been at lower risk of idiopathic SSNHL) was seen.
We then evaluated the possible temporal association between COVID-19 vaccination and the onset of idiopathic SSNHL as documented in VAERS incident reports ( Figure 2 A). The mean time to onset of SSNHL was 6 days (range, 0-21 days), with the highest incidence occurring at 0 days (70 reports), 1 day (104 reports), and 2 days (72 reports) after vaccination and a smaller second peak occurring at 7 days (38 reports) after vaccination.
To better understand the clinical profiles of patients who reported SSNHL after COVID-19 vaccination, we examined the detailed clinical characteristics of patients with confirmed hearing loss occurring after COVID-19 vaccination in a multi-institutional case series. A total of 21 patients were identified across study sites, with a mean age of 61 years (range, 23-92 years; 13 women [61.9%]). Demographic, clinical, and audiometric characteristics of patients are shown in Table 3 . Six patients (28.6%) had a history of autoimmune disease, including eczema, episcleritis, Hashimoto thyroiditis, multiple sclerosis, and rheumatoid arthritis. The mean time to onset of SSNHL was 6 days (range, 0-15 days) after vaccination, with the highest number of cases (6) occurring at 7 days after vaccination ( Figure 2 B). Overall, 18 of 21 patients (85.7%) received treatment; of those, 9 patients (50.0%) received intratympanic steroids, 5 (27.8%) received oral corticosteroids, and 4 (22.2%) received both. No adjuvant therapies were prescribed. Complete posttreatment audiometric data were available for 14 patients, 8 of whom (57.1%) experienced audiometric improvement ( Table 3 ; eFigure 1 in the Supplement ).
One patient without any history of Ménière disease or autoimmune inner ear disease experienced new-onset low-frequency (250-500 Hz) SSNHL at 14 days after the first vaccine dose; the condition improved with oral and intratympanic steroid treatment but worsened again at 14 days after the second vaccine dose (15-dB threshold increase in hearing loss at 500 Hz) (eFigure 2 in the Supplement ).
This comprehensive cross-sectional study of CDC VAERS reports of SSNHL after COVID-19 vaccination during the first 7 months of the national vaccination campaign included 185 million doses across all 3 manufacturers. Although VAERS reports contain raw data that are unverified, they present a national snapshot of potential AEs occurring after vaccination. Our analysis found that, based on VAERS reports, the estimated incidence of SSNHL after COVID-19 vaccination did not exceed the reported incidence of idiopathic SSNHL in the general population. 7 Furthermore, despite the novel delivery vehicle and immunologic mechanism of the mRNA-based vaccines manufactured by Pfizer-BioNTech and Moderna, we did not find an increased reporting rate of SSNHL associated with lipid nanoparticle mRNA vaccines compared with the adenoviral platform used in the Janssen/Johnson & Johnson vaccine.
We also hypothesized that if an association existed between COVID-19 vaccination and SSNHL, we would find an association between the number of reports of SSNHL submitted to VAERS and the number of vaccine doses administered. However, we found the rate of reports per 100 000 doses decreased across the vaccination period, despite large concomitant increases in the absolute number of vaccine doses administered per week ( Figure 1 A).
We also tested the hypothesis that the increased rate of reports of SSNHL in the initial vaccination phase could be associated with older individuals being vaccinated first 9 ; our analysis of the mean ages of people reporting SSNHL after vaccination to VAERS ( Figure 1 B) and the CDC COVID-19 tracking data on the number of individuals vaccinated in each age group over time (eFigure 3 in the Supplement ) did not support this hypothesis. Given that health care professionals were also included in the first phase of vaccination, one might assume that this group would be more attuned to AEs and more likely to report SSNHL; however, the relative number of health care professionals who initially experienced SSNHL was impossible to ascertain based on VAERS data. Taken together, these data suggest that an association between COVID-19 vaccination and SSNHL during the first 7 months of vaccination was unlikely at the population level.
Because VAERS incident reports lack clinical detail, conclusions regarding specific risk factors associated with SSNHL after COVID-19 vaccination cannot be reached. Narrative information within VAERS is self-reported and highly variable, ranging from no information on medical history to detailed information on both medical history and medication use. Thus, we assessed the clinical characteristics of patients with confirmed SSNHL at 3 large otolaryngological practices. The demographic and clinical characteristics of patients examined in our multi-institutional case series ( Table 3 ) did not clearly identify any specific cofactors among those experiencing SSNHL after vaccination, and patient characteristics appeared similar to the highly heterogeneous profiles observed among those with idiopathic SSNHL and those included in case series conducted at other institutions. 1 A previous study suggested that autoimmune disease may increase the risk of idiopathic SSNHL, 10 and we observed that autoimmune disease was present in 28.6% of the 21 patients in the case series reporting SSNHL after COVID-19 vaccination. Autoimmune disease as a risk factor for SSNHL with or without vaccination remains speculative, and further research is needed.
Both the mRNA payload and the lipid nanoparticle delivery vehicle have been suggested to be potential mechanisms of autoimmunogenicity. 11 Notably, the patient in the case series who reported having normal hearing before vaccination (no prevaccination audiometric data were available) and no history of autoimmune disease ( Table 3 ) was found to have low-frequency unilateral SSNHL at 14 days after the first vaccine dose. The patient received treatment with a course of oral steroid medication and experienced partial recovery of hearing; however, the patient subsequently reported new hearing deficit at 14 days after the second vaccine dose and was found to have a 15-dB threshold increase in hearing loss at 500 Hz (eFigure 2 in the Supplement ). Although not meeting the American Academy of Otolaryngology–Head and Neck Surgery criteria for SSNHL, 12 the observed audiometric changes were nonetheless concerning. Sudden sensorineural hearing loss after each COVID-19 vaccine dose was also reported among 3 patients in a recent case series, although 2 of those 3 patients had autoimmune inner ear disease, Ménière disease, or both. 1 Thus, our findings suggested that although no association between COVID-19 vaccination and SSNHL was found at the population level, an association among some individuals cannot be excluded without further research.
We also considered the timing of SSNHL after COVID-19 vaccination because this timing may have offered insight into the mechanistic basis of any potential biological association. For instance, Wichova et al 1 hypothesized that otologic symptoms, such as dizziness or SSNHL occurring 10 to 14 days after vaccination, could coincide with the production of immunoglobulin G at 10 to 14 days after vaccine administration. In both the national VAERS reports and our multi-institutional case series, we found that the mean time to onset of SSNHL was 6 days, with the highest incidence at 0 to 2 days and 7 days after vaccination ( Figure 2 A and B). These temporal patterns were consistent with the timing of onset for other COVID-19 vaccine–associated AEs, such as myocarditis (2-4 days) 13 - 15 and vaccine-induced immune thrombotic thrombocytopenia (7-10 days). 16 In a large epidemiologic study, Baxter et al 3 reported that the mean time to onset of reported SSNHL after influenza vaccination was also 2 days.
Observed peaks in reports of SSNHL at 1 and 7 days after vaccination in both VAERS and our case series could be partly accounted for by recall bias, which has been well documented in studies of passive vaccine AE reporting. 17 , 18 For example, an analysis of AEs associated with the hepatitis B vaccine, in which patient self-reports were cross-referenced with specific vaccination records, found substantial recall bias that produced an inaccurate association between vaccination and the development of multiple sclerosis. 19 The VAERS data may have been especially sensitive to recall bias because a substantial number of reports were submitted in a delayed manner, sometimes weeks to months after the onset of SSNHL. In particular, it is possible that patients, or health care professionals reporting on their behalf, may have estimated “about 1 day” or “about 1 week” when asked about the timing to onset of hearing loss because these are intuitive intervals for estimation. Bias in the perception of vaccine-associated AEs has substantial implications for an individual’s decision to receive a vaccine, as Betsch et al 20 reported in a study of a simulated online social network. Participants in that study were more likely to overestimate true vaccine-associated AE rates if presented with narratives from others that suggested a higher risk of experiencing a vaccine-associated AE, and they were subsequently less likely to receive a vaccine. 20 Notably, narrative information included in reports of AEs was more meaningful in influencing participants’ decisions to receive a vaccine than were statistical summaries. 20
Similar to recommendations provided by other reports of AE clusters, including cerebral venous sinus thrombosis 21 and myocarditis, 13 - 15 after COVID-19 vaccination, long-term epidemiologic and vaccine safety studies supported by mechanistic research are needed to more definitively address any potential association between COVID-19 vaccination and SSNHL. Reports of recovery of SARS-CoV-2 RNA in the middle ear of individuals who died of COVID-19 22 and recent findings of the ability of SARS-CoV-2 to directly infect human vestibular hair and Schwann cells 23 provide plausible biological mechanisms for COVID-19–associated hearing loss and may open avenues of investigation into immune mechanisms in the inner ear.
This study has several limitations. One limitation of the case series is its lack of a comparison group (eg, a group of patients who did not receive a COVID-19 vaccine but experienced SSNHL within the same period examined). Nonetheless, the detailed patient data in this series may serve as a supplement to the national patterns identified through analysis of SSNHL reports in the VAERS database.
Although an important tool for systematic vaccine safety studies, 24 the VAERS incident reports used in the present study are not yet verified by the CDC and therefore need to be interpreted with caution. We specifically focused on SSNHL, which is a well-defined clinical condition with a known population-level incidence, in contrast to other otolaryngological conditions, such as tinnitus or Ménière disease. To account for inherent uncertainties associated with raw report data, we developed a standardized case definition for probable SSNHL to identify the most credible incident reports. Few data exist to guide selection of the risk interval for SSNHL after vaccination. The 3-week interval used in the present study was designed to be longer than the primary interval used in previous studies 3 to balance considerations of temporal association with the risk of overexclusion.
It was also not possible to apply American Academy of Otolaryngology–Head and Neck Surgery criteria for SSNHL (loss of 30 dB over 3 consecutive frequencies) 12 to VAERS reports given the lack of numerical audiometric testing results contained within those reports. Using a sensitivity analysis, the maximum incidence estimate was produced based on the assumptions that (1) all submitted reports represented true SSNHL, which was unlikely (eTable 1 in the Supplement ), and (2) reports were subject to an additional 50% underreporting bias based on previous studies of VAERS sensitivity to detect rare AEs, such as Guillain-Barré syndrome and anaphylaxis. 6 Therefore, our calculated maximum incidence is likely an overestimate of the true incidence of SSNHL, especially given that our 3-week time to onset interval was substantially longer than the interval of 0 to 72 hours endorsed by the American Academy of Otolaryngology–Head and Neck Surgery. 12 In the absence of incident report verification and large-scale vaccine safety studies using verified reports, the estimation strategies used in this study nonetheless provide a snapshot and a potential tool that can be used by otolaryngologists challenged by this difficult clinical issue and its important public health implications.
This cross-sectional study and case series used an up-to-date analysis of VAERS case reports during the first 7 months of the US COVID-19 vaccination campaign across 3 vaccine manufacturers along with retrospective data from a series of patients with confirmed SSNHL, finding no population-level association between COVID-19 vaccination and SSNHL. Assessment of verified cases of SSNHL revealed heterogeneity in patient demographic characteristics, risk factors, and audiologic patterns. Further prospective investigation is needed to identify any potential associations between COVID-19 vaccination and SSNHL in some individuals. It is important that clinicians report all suspected COVID-19 vaccine–associated AEs rigorously and accurately to VAERS to allow verification and future performance of systematic vaccine safety studies.
Accepted for Publication: January 3, 2022.
Published Online: February 24, 2022. doi:10.1001/jamaoto.2021.4414
Corresponding Author: Eric J. Formeister, MD, MS, Department of Otolaryngology–Head and Neck Surgery, Johns Hopkins University School of Medicine, 601 N Caroline St, 6th Floor, Baltimore, MD 21287 ( [email protected] ).
Author Contributions: Drs Formeister and Sun had full access to all of the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Drs Galaiya, Kozin, and Sun contributed equally.
Concept and design: Formeister, Wu, Chari, Rauch, Remenschneider, Quesnel, Stewart, Galaiya, Kozin, Sun.
Acquisition, analysis, or interpretation of data: Formeister, Wu, Chari, Meek, Rauch, Remenschneider, Quesnel, de Venecia, Lee, Chien, Stewart, Kozin, Sun.
Drafting of the manuscript: Formeister, Wu, Chari, Stewart, Kozin, Sun.
Critical revision of the manuscript for important intellectual content: All authors.
Statistical analysis: Formeister, Wu, Sun.
Obtained funding: Sun.
Administrative, technical, or material support: Wu, Chari, Meek, Remenschneider, de Venecia, Galaiya, Sun.
Supervision: Chari, Remenschneider, Quesnel, Chien, Stewart, Galaiya, Kozin, Sun.
Conflict of Interest Disclosures: Dr Lee reported receiving personal fees from 3NT Medical and income and personal fees from Frequency Therapeutics outside the submitted work. Dr Quesnel reported receiving grants from Frequency Therapeutics and Grace Medical and personal fees from Frequency Therapeutics, and owning a patent for a protective drape to mitigate aerosol spread during otologic surgery (licensed to Grace Medical) outside the submitted work. No other disclosures were reported.
- Register for email alerts with links to free full-text articles
- Access PDFs of free articles
- Manage your interests
- Save searches and receive search alerts

843-469-1001
250 Mathis Ferry Road, Suite 101 Mt. Pleasant, SC 29464

- Lab Testing Offered
- Health Blogs
- Patient Portal
- Testimonials
- Career Page
- Aromatherapy
- Bemer Therapy
- Breast Thermography
- Chinese Medicine
- Corporate Wellness
- Detoxification
- Herbal Medicine
- Holistic Medicine
- Holistic Nutrition
- Hormone Pellets
- Hormone Replacement Therapy
- IV Vitamin Therapy
- Ozone IV MAH
- Pain Management Treatment
- Prolotherapy
- Weight Management
- Brain and Emotional Health
- Heart Health
- Men & Women’s Health
- Weight Loss
843-388-1612
Merge Medical Center
- Men & Women’s Health
Conditions Treated
- Brain & Emotional Health
- Men & Women’s Health

How Functional Medicine Addresses Hormonal Imbalances in Women
- Health Blog
By P. Lovegrove Medically Reviewed by Lindsay Langley, BSN, RN, CHT
Posted wednesday, august 28th, 2024.
Hormonal imbalances can wreak havoc on a woman’s body, causing everything from mood swings and fatigue to weight gain and irregular periods. Traditional medicine often focuses on treating symptoms, but functional medicine aims to address the root cause. In this article, we’ll dive deep into how functional medicine can help women with hormonal imbalances, offering a holistic approach to health and wellness.
What is Functional Medicine?
Before we get into the specifics of how functional medicine tackles hormonal imbalances, let’s break down what functional medicine is all about.
A Holistic Approach
Functional medicine is all about looking at the body as a whole. Instead of just treating symptoms, it aims to understand the underlying causes of health issues. It’s like being a detective—functional medicine practitioners gather clues from your lifestyle, environment, genetics, and biochemistry to figure out what’s going on.
Personalized Treatment
No two people are the same, and functional medicine recognizes that. Treatments are tailored to the individual, taking into account unique factors like diet, stress levels, sleep patterns, and more. This personalized approach ensures that you get the care that’s right for you.
Understanding Hormonal Imbalances
Hormones play a crucial role in regulating many of the body’s processes, from metabolism and mood to reproductive health. When these hormones are out of balance, it can lead to a variety of issues.
Common Symptoms
Hormonal imbalances can manifest in many ways. Here are some common symptoms that women might experience:
- Mood swings and irritability
- Fatigue and low energy
- Weight gain or difficulty losing weight
- Irregular periods or menstrual issues
- Hot flashes and night sweats
- Decreased libido
- Sleep disturbances
Causes of Hormonal Imbalances
Several factors can contribute to hormonal imbalances, including:
- Stress: Chronic stress can lead to elevated cortisol levels, which can interfere with other hormones.
- Diet: Poor nutrition, especially diets high in sugar and processed foods can disrupt hormone production.
- Lack of Exercise: A sedentary lifestyle can contribute to hormonal imbalances.
- Environmental Toxins: Exposure to chemicals and pollutants can affect hormone levels.
- Sleep Issues: Poor sleep can disrupt the balance of hormones like melatonin and cortisol.
- Genetics: Some women may be more predisposed to hormonal imbalances due to their genetic makeup.
How Functional Medicine Approaches Hormonal Imbalances
Functional medicine addresses hormonal imbalances by looking at the bigger picture. Here’s how it works:
Comprehensive Testing
Functional medicine practitioners start with comprehensive testing to get a clear picture of what’s happening inside your body. This might include blood tests, saliva tests, and even stool tests to check for gut health issues. These tests help identify specific hormonal imbalances and other underlying issues.
Personalized Treatment Plans
Once the testing is done, a personalized treatment plan is created. This plan might include:
- Dietary Changes: Nutrition plays a huge role in hormone health. A functional medicine practitioner will likely recommend a diet rich in whole foods, healthy fats, and plenty of vegetables. They might also suggest eliminating foods that can cause inflammation, like sugar and processed foods.
- Supplements: Certain supplements can help balance hormones. Common recommendations might include omega-3 fatty acids, vitamin D, magnesium, and adaptogenic herbs like ashwagandha.
- Stress Management: Since stress is a major factor in hormonal imbalances, finding ways to manage stress is crucial. This might involve techniques like mindfulness, meditation, yoga, or even just taking time for self-care.
- Exercise: Regular physical activity can help regulate hormones. A functional medicine practitioner might suggest a mix of cardio, strength training, and activities that promote relaxation, like yoga or tai chi.
- Sleep Hygiene: Improving sleep can have a big impact on hormone health. This might involve creating a bedtime routine, reducing screen time before bed, and making your sleep environment as comfortable as possible.
The Role of Nutrition in Hormonal Health
Let’s take a closer look at how diet and nutrition play a key role in managing hormonal imbalances.
The Importance of Whole Foods
A diet rich in whole foods can help stabilize blood sugar levels and support hormone production. Here are some particularly beneficial foods:
- Leafy Greens: Spinach, kale, and other greens are packed with nutrients that support overall health.
- Healthy Fats: Avocados, nuts, seeds, and olive oil provide essential fatty acids that are crucial for hormone production.
- Lean Proteins: Chicken, fish, and plant-based proteins like beans and lentils help maintain muscle mass and keep you feeling full.
- Fiber-Rich Foods: Fiber helps regulate digestion and can aid in the removal of excess hormones from the body.
Foods to Avoid
Certain foods can disrupt hormone balance and should be limited or avoided:
- Sugar and Refined Carbs: These can cause spikes in blood sugar and insulin, leading to hormonal imbalances.
- Processed Foods: Often high in unhealthy fats and chemicals, processed foods can interfere with hormone production.
- Caffeine and Alcohol: Both can affect cortisol levels and disrupt sleep, impacting overall hormone health.
Supplements for Hormonal Balance
While a healthy diet is crucial, supplements can also play a key role in supporting hormonal health. Here are some commonly recommended supplements in functional medicine:
Omega-3 Fatty Acids
Omega-3s are anti-inflammatory and can help balance hormones. They are found in fish oil, flaxseeds, and walnuts.
Vitamin D is important for hormone production and overall health. Many people are deficient in this vitamin, especially those who live in areas with limited sunlight.
Magnesium helps regulate cortisol and supports overall hormonal balance. It’s found in foods like nuts, seeds, and leafy greens, but many people benefit from a supplement.
Adaptogenic Herbs
Adaptogens like ashwagandha, Rhodiola, and maca can help the body adapt to stress and support adrenal health, which in turn can help balance hormones.
Stress Management Techniques
Managing stress is a key component of balancing hormones. Here are some effective stress management techniques:
Mindfulness and Meditation
Practicing mindfulness and meditation can help reduce stress and lower cortisol levels. Even a few minutes a day can make a big difference.
Yoga and Tai Chi
These gentle forms of exercise not only promote physical health but also help reduce stress and improve mental clarity.
Taking time for self-care is crucial. This might include activities like reading, taking a bath, or spending time in nature.
Exercise and Hormonal Health
Regular exercise is essential for maintaining hormonal balance. Here are some tips for incorporating exercise into your routine:
A combination of cardio, strength training, and flexibility exercises is ideal. Cardio helps burn calories and improve heart health, strength training builds muscle, and flexibility exercises like yoga promote relaxation and stress reduction.
Consistency is Key
Consistency is more important than intensity. Aim for at least 30 minutes of exercise most days of the week.
Listen to Your Body
It’s important to listen to your body and not overdo it. Over-exercising can lead to hormonal imbalances, so find a balance that works for you.
Sleep and Hormonal Balance
Good sleep is crucial for hormone health. Here are some tips for improving sleep:
Create a Bedtime Routine
Creating a calming bedtime routine can signal to your body that it’s time to wind down. This might include activities like reading, taking a warm bath, or practicing relaxation techniques.
Reduce Screen Time
Reducing screen time before bed can help improve sleep quality. The blue light from screens can interfere with the production of melatonin, a hormone that regulates sleep.
Make Your Sleep Environment Comfortable
A comfortable sleep environment can make a big difference. This might include investing in a good mattress and pillows, keeping the room cool and dark, and reducing noise.
Case Studies: Functional Medicine Success Stories
Let’s look at real-life examples of how functional medicine has helped women with hormonal imbalances.
Case Study 1: Sarah’s Journey to Balance
Sarah was in her mid-30s and struggling with fatigue, weight gain, and irregular periods. Traditional treatments weren’t helping, so she turned to functional medicine. After comprehensive testing, it was found that Sarah had high cortisol levels and insulin resistance. Her personalized treatment plan included dietary changes, stress management techniques, and targeted supplements. Within six months, Sarah’s energy levels improved, she lost weight, and her periods became regular.
Case Study 2: Emily’s Road to Recovery
Emily, a 45-year-old mother of two, was experiencing hot flashes, mood swings, and trouble sleeping. A functional medicine approach revealed that Emily’s estrogen levels were low, and she was also dealing with chronic stress. Her treatment plan included hormone-supporting foods, mindfulness practices, and herbal supplements. Over time, Emily’s symptoms lessened, and she regained a sense of well-being.
Functional medicine offers a comprehensive, personalized approach to addressing hormonal imbalances in women. By looking at the whole picture and addressing the root causes, functional medicine can help women achieve better health and well-being. Whether through dietary changes, stress management, supplements, or lifestyle adjustments, functional medicine provides the tools and strategies to restore hormonal balance and improve quality of life.
Remember, it’s important to work with a qualified functional medicine practitioner to develop a plan tailored to your unique needs. With the right support, you can take control of your health and feel your best.
Categories:
Functional medicine, about the author, p. lovegrove, recent news, connect with us, please follow us on facebook to stay informed about our promotions and events., subscribe to our newsletter, get dr. lovegrove's newsletter and biggest health secrets.
Site powered by Zeniark. All Rights Reserved 2024.
- Education Home
- Medical Education Technology Support
- Graduate Medical Education
- Medical Scientist Training Program
- Public Health Sciences Program
- Continuing Medical Education
- Clinical Performance Education Center
- Center for Excellence in Education
- Research Home
- Biochemistry & Molecular Genetics
- Biomedical Engineering
- Cell Biology
- Genome Sciences
- Microbiology, Immunology, & Cancer Biology (MIC)
- Molecular Physiology & Biological Physics
- Neuroscience
- Pharmacology
- Public Health Sciences
- Office for Research
- Clinical Research
- Clinical Trials Office
- Funding Opportunities
- Grants & Contracts
- Research Faculty Directory
- Cancer Center
- Cardiovascular Research Center
- Carter Immunology Center
- Center for Behavioral Health & Technology
- Center for Brain Immunology & Glia
- Center for Diabetes Technology
- Center for Immunity, Inflammation & Regenerative Medicine
- Center for Membrane & Cell Physiology
- Center for Research in Reproduction
- Myles H. Thaler Center for AIDS & Human Retrovirus Research
- Child Health Research Center (Pediatrics)
- Division of Perceptual Studies
- Research News: The Making of Medicine
- Core Facilities
- Virginia Research Resources Consortium
- Center for Advanced Vision Science
- Charles O. Strickler Transplant Center
- Keck Center for Cellular Imaging
- Institute of Law, Psychiatry & Public Policy
- Translational Health Research Institute of Virginia
- Clinical Home
- Anesthesiology
- Dermatology
- Emergency Medicine
- Family Medicine
- Neurosurgery
- Obstetrics & Gynecology
- Ophthalmology
- Orthopaedic Surgery
- Otolaryngology
- Physical Medicine & Rehabilitation
- Plastic Surgery, Maxillofacial, & Oral Health
- Psychiatry & Neurobehavioral Sciences
- Radiation Oncology
- Radiology & Medical Imaging
- UVA Health: Patient Care
- Diversity Home
- Diversity Overview
- Student Resources
- GME Trainee Resources
- Faculty Resources
- Community Resources
- Medical Student Case Studies
Each student is required to prepare a teaching case to present to his/her colleagues and the course director. Past examples provided below.
General Diagnostic Case Studies
- Arteriovenous Fistula Secondary to Trauma
- Chylothorax, Cellulitis, Gas Gangrene
- Ectopic ACTH-Secreting Tumor
- Epiphrenic Diverticula
- Incidental Mediastinal Hilar Lymphadenopathy
- Liver Metastasis
- Neurocystercircosis
- Pneumoperitoneum
- Pneumothorax
- Posterior Urethralcutaneous Fistula
- Renal Cysts
- Rocky Mountain Spotted Fever
- Septic Emboli to the Brain
- Submassive Bilateral PE
- Tailgut Duplication Cyst
- Tension Pneumocephalus
- Tracheo-Esophageal Fistula
- Vestibular Schwannoma
Radiology Pathology Correlation Case Studies
- Adenoid Cystic Carcinoma
- Cholangiocarcinoma
- Cryptococcal PNA
- Disseminated Histoplasmosis
- Lung Adenocarcinoma
- Marginal Zone B-cell Lymphoma
- Metastatic Urothelial Carcinoma
- Mixed Clear Cell Papillary RCC
- Ovarian Carcinoma
- Pancreatic Adenocarcinoma
- Squamous Cell Carcinoma of the Lung
- Squamous Cell Carcinoma
- Uterine Leiomyosarcoma
- Cervical Adenosquamous Carcinoma
- Clear Cell Renal Cell Carcinoma
- Esophageal Adenocarcinoma
- Metastic Esophageal Gastrointestinal Stomal Tumor
- Ocular Melanoma with Mets to Liver
- Pancreatic Metatasis
- Papillary Thyroid Carcinoma
- Papillary Thyroid Carcinoma #2
- Renal Transplant Rejection
- Small Cell Lung Cancer
- Urothelial Carcinoma Lung Met
- Yolk Sac Tumor
- Burkitt’s Lymphoma
- Ewing Sarcoma
- Hepatocellular Carcinoma
- Hepatocellular Carcinoma #2
- High-grade Serous Ovarian Carcinoma
- Metastatic Duodenal Adenocarcinoma
- Metastatic Melanoma
- Ovarian Cancer
- Papillary Thyroid Carcinoma #3
- Papillary Thyroid Carcinoma with Nodal Involvement
- Well Differentiated Hepatocellular Carcinoma
- How to Apply
- Alexander (Sasha) Sytov, MD
- Alexander Zhang, MD
- Anna Osment, MD
- Connor Sleeth, MD
- Eric Fromke, MD
- Hannah Clode, MD
- Jenna Pollock, MD
- Jineet Patel, MD
- Joshua Hunsaker, MD
- Joshua Ravicz, MD
- Julia Kariher, MD
- Kaelin Cockrell, MD
- Maclean Cook, MD
- Marissa Boettcher, MD
- Nabeel Mirza, MD
- Nathan Leaphart, MD
- Qi (Phoebe) Dong, MD
- Samantha Epstein, MD
- Tyler Dalton, MD
- Vatsal Lal, MD
- Warren Campbell IV, MD, PhD
- Wayne Dell, MD
- Zachary Miller, MD, PhD
- Salary and Benefits
- Resident Lifestyle
- Early Specialization in Interventional Radiology (ESIR)
- Diagnostic Radiology/Nuclear Medicine (DR/NM) Pathway
- Diagnostic Radiology Research Track
- Current IR Residents
- Where Are They Now?
- Letter from the Chief Residents
- Former Residents
- Your UVA Interview Day
- Living in Charlottesville
- Participating Residents
- Program News and Updates
- Make a Gift
- Current Representatives
- Ladyologists – Supporting Each Other
- 4th-year Medical Students Scholarship
- Radiology Electives
- Radiology Interest Group
- Abdominal Imaging
- Breast Imaging
- Cardiothoracic Imaging
- Diagnostic Neuroradiology
- Musculoskeletal Imaging
- Nuclear Radiology
- Pediatric Radiology
- Angiography and Interventional Radiology Observership
- Breast Imaging International Visiting Scholars
- Why Choose Structured Education?
- Program Directors
- Verification of Graduate Medical Education Training and Faculty Appointment
- Online Training Resources
- Open access
- Published: 26 August 2024
Association between social determinants of health and survival among the US cancer survivors population
- Hongbo Huang 1 ,
- Tingting Wei 1 ,
- Ying Huang 1 ,
- Aijie Zhang 2 ,
- Heng Zhang 1 ,
- Ze Zhang 1 ,
- Yijing Xu 1 ,
- Haonan Pan 3 ,
- Lingquan Kong 1 ,
- Yunhai Li 1 &
BMC Medicine volume 22 , Article number: 343 ( 2024 ) Cite this article
170 Accesses
Metrics details
Racial and ethnic disparities in mortality persist among US cancer survivors, with social determinants of health (SDoH) may have a significant impact on these disparities.
A population-based cohort study of a nationally representative sample of adult cancer survivors, who participated in the US National Health and Nutrition Examination Survey from 1999 to 2018 was included. Sociodemographic characteristics and SDoH were self-reported using standardized questionnaires in each survey cycle. The SDoH was examined by race and estimated for associations with primary outcomes, which included all-cause and cancer-specific mortality. Multiple mediation analysis was performed to assess the contribution of each unfavorable SDoH to racial disparities to all-cause and cancer-specific mortality.
Among 5163 cancer survivors (2724 [57.7%] females and 3580 [69.3%] non-Hispanic White individuals), only 881 (24.9%) did not report an unfavorable SDoH. During the follow-up period of up to 249 months (median 81 months), 1964 deaths were recorded (cancer, 624; cardiovascular, 529; other causes, 811). Disparities in all-cause and cancer-specific mortality were observed between non-Hispanic Black and White cancer survivors. Unemployment, lower economic status, education less than high school, government or no private insurance, renting a home or other arrangements, and social isolation were significantly and independently associated with worse overall survival. Unemployment, lower economic status, and social isolation were significantly associated with cancer-specific mortality. Compared to patients without an unfavorable SDoH, the risk of all-cause mortality was gradually increased in those with a cumulative number of unfavorable SDoHs (1 unfavorable SDoH: hazard ratio [HR] = 1.54, 95% CI 1.25–1.89; 2 unfavorable SDoHs: HR = 1.81, 95% CI 1.46–2.24; 3 unfavorable SDoHs: HR = 2.42, 95% CI 1.97–2.97; 4 unfavorable SDoHs: HR = 3.22, 95% CI 2.48–4.19; 5 unfavorable SDoHs: HR = 3.99, 95% CI 2.99–5.33; 6 unfavorable SDoHs: HR = 6.34 95% CI 4.51–8.90). A similar trend existed for cancer-specific mortality.
Conclusions
In this cohort study of a nationally representative sample of US cancer survivors, a greater number of unfavorable SDoH was associated with increased risks of mortality from all causes and cancer. Unfavorable SDoH levels were critical risk factors for all-cause and cancer-specific mortality, as well as the underlying cause of racial all-cause mortality disparities among US cancer survivors.
Peer Review reports
An increase in the cancer survivor population poses a significant health care and economic burden worldwide, and cancer is the second leading cause of death in the United States (US). Specifically, there will be approximately 2,001,140 new cancer cases and an estimated 611,720 deaths from cancer in the US in 2024 [ 1 ]. Although cancer mortality has declined overall by 33% since 1991, improved survival outcomes have not benefitted equally for all cancer populations [ 1 , 2 ]. Substantial racial and ethnic disparities in all-cause and cancer-related mortality rates persist in US cancer survivors [ 1 , 2 , 3 , 4 ]. For example, Black individuals have lower relative cancer survival rates than White individuals for almost every cancer type [ 1 , 5 ]. Interestingly, the most striking gaps in survival involve cancers that are most amenable to prevention and early detection, such as cervical cancer [ 5 ]. Recently, the racial and ethnic disparities in cancer mortality have slowly narrowed; however, these disparities in cancer health have become increasingly understood in the context of social determinants of health (SDoH) [ 2 , 5 , 6 ], which are responsible for an extremely important factor associated with cancer risk and treatment [ 7 ]. The World Health Organization (WHO) defined SDoH as non-medical factors that affect health outcomes, including the conditions in which people are born, grow, live, work, and age, and a wider set of forces and systems shaping daily life conditions [ 8 ]. The SDoH included factors related to economic stability, education, health care access, residential environment, and social context and support [ 9 , 10 , 11 ], associated with the health outcomes of cancer survivors [ 12 , 13 ]. Addressing social disparities in cancer health is essential in the quest to improve survival outcomes among cancer survivors, which reflects a commitment to health equity to achieve optimal health for everyone.
Previous studies have tended to examine the contribution of individual variables involving unfavorable SDoHs in the separate associations with mortality or morbidity, most of which focused on the direct and indirect influence of socioeconomic factors on the disparity in survival [ 9 , 14 , 15 , 16 , 17 , 18 , 19 , 20 , 21 , 22 , 23 ]. Among the general population, a large multicohort study and meta-analysis with more than 1.7 million individuals from 7 WHO member countries reported that low socioeconomic status was associated with a 46% (95% CI, 39–53%) and 43% (95% CI, 34–52%) greater risk of all-cause and cancer mortality, respectively, compared to high socioeconomic status [ 15 ]. A low level of education, poverty, and a lack of health insurance coverage explain in part the continuous widening in mortality inequities across some adult sociodemographic groups in the US [ 14 , 17 , 20 , 24 ]. Additionally, a recent analysis demonstrated that the cumulative SDoH count was associated with an increased premature mortality risk [ 25 ]. However, limited evidence has been reported on the effect of SDoH in cancer survivors. Although previous cohort studies have shown that disadvantaged SDoH are associated with poor mental and physical health [ 26 ], resulting in a delay in medical and surgical treatment [ 27 ], and an increased risk of all-cause and cancer-related mortality among patients with cancers (such as breast and pancreatic cancer) [ 12 , 13 ]. To the best of our knowledge, few studies have examined the impact of the comprehensive and accumulating burden of SDoH on all-cause and cause-specific mortality, using methods published previously [ 13 , 25 ]. There is no study that has reported the relative contributions of these SDoH on racial disparities in the all-cause and cancer-specific mortality rates among the US cancer survivors at the population level.
The objective of the present study was to evaluate the relationships of multiple SDoH with all-cause, cancer-specific, and non-cancer mortality, and to investigate how SDoH mediates racial differences in all-cause and cancer-specific mortality among cancer survivors. We hypothesized that disparities exist in the cumulative number of unfavorable SDoH across racial and ethnic groups and that a higher number of these unfavorable SDoH is associated with higher mortality rates.
Study population
In this retrospective study, 10 cycles of cross-sectional data were collected from the National Health and Nutrition Examination Survey (NHANES) database, which used a complex, multistage, and probability sampling design to recruit participants representative of the civilian non-institutionalized US population [ 28 ]. Each participant was invited to attend an in-person or in-home interview to complete the questionnaire. The present study examined and analyzed existing data involving sociodemographic characteristics and several SDoH co-variables among cancer survivors of 20 years or older with information linked to the National Death Index through 31 December 2019 for 10 survey cycles of NHANES from 1999–2000 to 2017–2018. All the NHANES protocols were approved by the Research Ethics Review Board of the National Center for Health Statistics (NCHS), and written informed consent was provided by all participants at the time of recruitment.
Sociodemographic characteristics
In each 2-year survey, age, gender, and racial or ethnic groups (non-Hispanic White [NHW], Hispanic, non-Hispanic Black [NHB], and other [American Indian/Alaska Native/Pacific Islander, Asian, and multiracial]) were obtained from standardized questionnaires of in-home interviews by self or parent/guardian report from provided categories.
SDoH assessment
We included several variables that reflected SDoH information from standardized questionnaires, which were defined according to the Healthy People 2030 [ 11 ] and World Health Organization [ 29 ] by the following factors: economic stability; education access and quality; health access and quality; neighborhood and built environment; and social and community context. In the present study, we finally chose eight SDoH variables (employment status, family poverty income ratio, food security, education level, regular health care access, type of health insurance, home ownership, and marital status) in each NHANES cycles from 1999 to 2018, according to previously published studies [ 25 , 30 ]. Social support was excluded because it was only visible in surveys conducted between 1999 and 2008. More detailed description information on SDoH was provided in the supplement (Additional file 1: Table S1) [ 31 , 32 , 33 ], and the definition for unfavorable SDoH was based on the conventional cutoff points [ 10 , 11 , 23 , 34 , 35 ]. Furthermore, the associations between several single SDoH measures and all-cause mortality were investigated using various categorizations with adjustment for age, gender, race, and ethnicity regardless of survey weights (Additional file 1: Table S2). Each SDoH was divided into two levels based on the conventional cut-off points [ 11 , 23 , 34 , 35 ]. Unfavorable SDoH was significantly associated with a lower survival rate. During the in-person interview, participants were asked to respond to several questions about these SDoH. Economic stability was operationalized using self-reported measures of the family poverty income ratio (PIR, less than 2.4 [unfavorable SDoH] and more than 2.4 [favorable SDoH]), employment status (employed, student, or retired [favorable SDoH] and unemployed [unfavorable SDoH]) and household food security category, which was dichotomized as fully food security (no affirmative response) or marginal, low, or very low security (1–10 affirmative responses) based on the responses to the US Food Security Survey Module questions (Bickel et al. [ 36 ]). Education access and quality measurement used the highest grade or level of schooling completed or the highest degree received, dichotomized as less than high school (unfavorable SDoH) and high school graduate or higher (favorable SDoH). Health care access and quality were assessed by self-reported questionnaire about routine places for health care (at least one regular health care facility [favorable SDoH] and none or hospital emergency room [unfavorable SDoH]) and health insurance type (private [favorable SDoH] and none or government [unfavorable SDoH]). The residential environment was assessed by home ownership (owned or being bought [favorable SDoH] and rental or other arrangement [unfavorable SDoH]). Social community context was assessed by self-reported marital status (defined as married or living with a partner [favorable SDoH] and not married nor living with a partner [unfavorable SDoH]).
The cumulative number of unfavorable SDoH variables with a range from 0 (no unfavorable SDoH) to 6 or more (≥ 6 unfavorable SDoH) was calculated to explore the cumulative effect of unfavorable SDoH on all-cause and cancer-specific mortality. Because only a small proportion of participants reported having 6, 7, or 8 unfavorable SDoH variables simultaneously, thus we created a category of six or more, indicating the combination of 6, 7, or 8 unfavorable SDoH variables.
Definition of cancer survivors
Information on cancer diagnosis was collected from survey questionnaires during the in-person interview using the computer-assisted personal interview system, including cancer type(s), with up to three cancer diagnoses recorded and the age at first diagnosis for each cancer. Participants were asked, “Have you ever been told by a doctor or other health professional that you had cancer or a malignancy of any kind?” If individuals who answered “yes” were defined as cancer survivors and were asked further, “What kind of cancer was it?” and “How old were you when this cancer was first diagnosed?”.
Ascertainment of mortality
The NCHS provided mortality data that were linked to the National Death Index, with follow-up until 31 December 2019 [ 37 ]. Cause-of-death coding for all US deaths occurring after 1998 followed the 10th revision of the International Statistical Classification of Diseases, Injuries, and Causes of Death (ICD-10) guidelines. Cancer-related mortality was classified as death due to malignant neoplasms (ICD-10, codes C00-C97). The follow-up duration was defined as the interval elapsing from the date of the baseline interview to the date of death or the follow-up cut-off (31 December 2019) for those participants who did not have a death event in the whole study. We investigated the association between SDoH and all-cause, cancer-related, and non-cancer mortality (mortality instead of cancer, ICD-10 codes instead of C00-97). All-cause and cancer-specific mortality were the main outcomes of this study. The all-cause mortality was measured from the date of the baseline interview to the date of death from any cause or the follow-up cut-off. The cancer-specific mortality was calculated from the date of the baseline interview to the date of death from cancer.
Statistical analysis
All statistical analyses were conducted with the use of R (version 4.3.1) following the NHANES analysis guidance. The survey interview weights were used for analysis as appropriate to obtain nationally representative estimates. We calculated weighted sample sizes to be nationally representative and population-weighted percentages according to race and ethnicity. The chi-square test was used to determine the differences in participants’ sociodemographic characteristics and SDoH variables across four classifications of racial and ethnic groups. The pairwise correlation among the eight dichotomous SDoH was evaluated using the Spearman method. The weighted proportions of cancer survivors in each number of unfavorable SDoH category were estimated by gender, race, and ethnicity. Kaplan–Meier survival curves were examined to determine the all-cause cumulative mortality and cancer-specific cumulative mortality rates among cancer survivors stratified by SDoH. Furthermore, Kaplan–Meier analysis was used to plot the cumulative hazard for all-cause and cancer-specific mortality in entire and gender subgroups and race and ethnicity subgroups using age as the timescale [ 38 ].
Multivariable Cox proportional hazards regression models with the use of imputation-adjusted survey weights were applied to estimate the mortality risks (hazard ratio [HR]) and 95% confidence interval (CI) for the associations between cumulative SDoH variables and race with all-cause, cancer-specific, and non-cancer mortality. Final stage multivariable Cox models were adjusted for age, gender, race, and ethnicity, and additionally included the other SDoHs to identify independent, indirect associations. We plotted the HRs of the cumulative SDoH variables to visualize whether the relationship with all-cause and cancer-specific mortality was linear or non-linear. Sensitivity analyses were performed by excluding participants of deaths that occurred within the first 2-year follow-up to lessen the probability of reverse causation [ 39 ]. All statistical tests were 2-sided and P < 0.05 was considered statistically significant. Data analyses were performed from 1 June to 1 August 2023.
Because of racial disparity in all-cause and cancer-specific mortality between NHW and NHB among cancer survivors in the US, therefore mediation analysis was performed to explore whether SDoH factors contributed to White-Black disparity in mortality or not. We estimated the relative effect (corresponding direct or indirect effect divided by the total effect) of each SDoH variable to explain the racial and ethnic difference in mortality using R package mma [ 31 , 32 , 33 ]. More detailed information was contained in the Supplementary material (Additional file 1: Methods S1).
NHANES (1999–2018) data from 5163 individuals were enrolled in the final analysis (Additional file 1: Fig. S1). A total of 101,316 persons ≥ 1 year of age who participated in the in-person or in-home interview and 96,153 were excluded, as follows: (1) 46 235 participants < 20 years of age; (2) 49,915 whom were not diagnosed with cancer, and (3) 3 individuals who did not have unique identifiers to allow linkage to the National Death Index. Of the 5163 cancer survivors (weighted population, 32,623 176; 57.7% female) in this study cohort, 3580 (69.3%) were NHW, 631 (12.2%) were Hispanic, 718 (13.9%) were NHB, and 234 (4.5%) individuals of were classified as race and ethnicity, including American Indian/native Alaskan, Pacific Islander, Asian, and multiracial (Table 1 ). Compared to NHW, Hispanic, NHB and other race and ethnic cancer survivors were more likely to have unfavorable SDoH factors, including not being married nor living with a partner, education less than high school, a PIR < 2.4, renting a home or other arrangement, unemployment, government or none health insurance, and marginal, low, or very low security. However, a lower proportion of NHB participants had no place routine place when sick or in need of advice about healthcare compared with cancer survivors from all other racial and ethnic subgroups. Approximately 24.9% of cancer survivors did not have a cumulative number of unfavorable SDoH. The higher proportion of NHW cancer survivors with 0 and 1 cumulative unfavorable SDoH was observed compared to patients from all other race and ethnic subgroups. In addition, a higher proportion of Hispanic cancer survivors with 3, 4, 5, and 6 or more unfavorable SDoH was observed compared to patients from all other race and ethnic subgroups. NHB and Hispanic individuals had a higher prevalence of multiple unfavorable SDoH (cumulative of 3 or more) compared to NHW cancer survivors.
Then, we analyzed the relationship between the eight SDoH variables. The results showed that all eight SDoH variables were significantly correlated with each other (Additional file 1: Fig. S2). Furthermore, the proportion of male participants decreased stepwise from 34.8% (0 unfavorable SDoH) to 2.2% (6 or more number of unfavorable SDoH), whereas the proportion of female participants increased from 22.6% (0 unfavorable SDoH) to 24.8% (1 unfavorable SDoH), and then gradually decreased to 4.7% (6 or more number of unfavorable SDoH; Additional file 1: Fig. S3). Breast and prostate cancer were the most common malignant neoplasm type in males and females, respectively (Additional file 1: Table S3).
During the median follow-up of 81 months (ranged 0–249 months) in the 10 NHANES cycles linked mortality file cohort, a total of 1964 deaths occurred (all-cause), including 624 cancer patients who died from cancer (cancer-related mortality), 529 who died from cardiovascular disease, and 811 who died from other cause. Compared to participants who were NHW, NHB adults with cancer had a significantly higher overall mortality rate (HR, 1.57; 95% CI, 1.34–1.89) and cancer-specific mortality (HR, 2.03; 95% CI, 1.60–2.59; Fig. 1 ). Cancer survivors with each unfavorable SDoH variable, except access to regular health care, was significantly associated with higher all-cause, cancer-specific, and non-cancer mortality in the multivariable model adjusted for age (MV model 1), and adjusted for age, gender, race and ethnicity (MV model 2; Table 2 ). After adjustment for age, gender, race and ethnicity and other SDoHs, including unemployment status (HR, 1.83; 95% CI, 1.58–2.12; P < 0.001), family income-to-poverty less than 2.4 (HR, 1.51; 95% CI, 1.32–1.72; P < 0.001), education less than high school attached (HR, 1.23; 95% CI, 1.05–1.44; P = 0.012), government or none of health insurance (HR, 1.19; 95% CI, 1.05–1.36; P = 0.007), renting a home or other housing arrangement environment (HR, 1.39; 95% CI, 1.20–1.62; P < 0.001), and not being married nor living with a partner (HR, 1.22; 95% CI, 1.08–1.38; P < 0.001) were significantly associated with an increased risk for all-cause mortality, which was similar to non-cancer mortality (Table 2 ). Furthermore, unemployed individuals (HR, 2.13; 95% CI, 1.62–2.79; P < 0.001), family income-to-poverty less than 2.4 (HR, 1.35; 95% CI, 1.09–1.66; P = 0.006), and not being married nor living with a partner (HR, 1.22; 95% CI, 1.08–1.38; P < 0.001) were significantly associated with an increased cancer-specific mortality risk compared to those with favorable SDoH (Table 2 ). Specifically, individuals of being unemployed status were associated with almost more than 1.9- and 2.2-fold higher all-cause mortality and cancer-specific mortality rates, respectively.
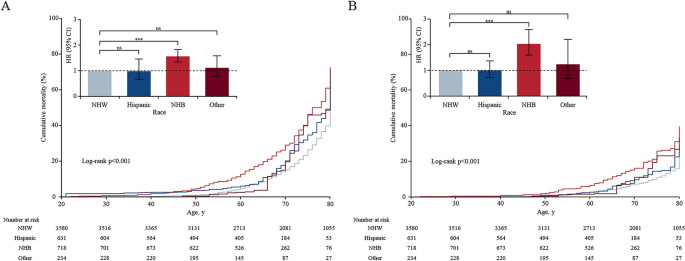
All-cause mortality ( A ), cancer-specific mortality ( B ), and hazard ratios in US adults diagnosed with cancers aged 20 years or older by race and ethnicity. Note: Kaplan–Meier curves showed cumulative mortality probability race and ethnicity using age as the timescale. The number at risk was unweighted observed frequencies. Cumulative mortality rates were estimated with the use of survey weights. The bar chart showed HRs of all-cause and cancer-specific mortality associated with race and ethnicity, adjusted for age, and gender. Error bars were 95% CIs. NHW indicated non-Hispanic White; NHB indicated non-Hispanic Black; HR indicated hazard ratio; ns was the abbreviation of no significance; *** meant p < 0.001, ** meant p < 0.01, and * meant p < 0.05
Cancer survivors with a greater cumulative number of SDoHs were significantly associated with an increased risk of death from all-cause and cancer (Additional file 1: Fig. S4; P < 0.001). In the multivariable of MV model 1 (adjusted for age, gender, race, and ethnic), the HRs for all-cause and cancer-specific mortality were 1.54 (95% CI, 1.25–1.89) and 1.52 (95% CI, 1.04–2.22) for cancer survivors with 1 unfavorable SDoH, 1.81 (95% CI, 1.46–2.24) and 1.70 (95% CI, 1.20–2.24) for those with 2 unfavorable SDoHs, 2.42 (95% CI, 1.97–2.97) and 2.22 (95% CI, 1.51–3.26) for those with 3 unfavorable SDoHs, 3.22 (95% CI, 2.48–4.19) and 2.44 (95% CI, 1.60–3.72) for those with 4 unfavorable SDoHs, 3.99 (95% CI, 2.99–5.33) and 3.60 (95% CI, 2.25–5.75) for those with 5 unfavorable SDoHs, and 6.34 (95% CI, 4.51–8.90) and 5.00 (95% CI, 3.00–8.31) for those with 6 or more unfavorable SDoHs, respectively, compared with of whom without unfavorable SDoH (Fig. 2 ). Kaplan–Meier curves were used to estimate the cumulative probability of all-cause and cancer-specific mortality using age as the timescale. The all-cause and cancer-specific mortality rates were significant across the several groups with a cumulative number of unfavorable SDoHs (Fig. 2 , P < 0.001). Pairwise comparison using log-rank showed that the all-cause mortality rate was similar and not significantly different among cancer survivors with 0, 1, 2, and 3 cumulative number of unfavorable SDoH across the entire age cohort (Additional file 1: Table S4). There was no significant difference in cancer-specific mortality among cancer survivors with 0, 1, 2, 3, and 4 cumulative number of unfavorable SDoH (Additional file 1: Table S5). Based on the linear dose–response analysis fitted curves (unfavorable SDoH ranged from 0 to 8), every cumulative unfavorable SDoH increase was significantly associated with 64% increased risks of death from all-cause (HR per 1-number increase, 1.64 [95% CI, 1.50–1.78]), and 53% of cancer (HR per 1-number increase, 1.53 [95% CI, 1.45–1.60]) (Additional file 1: Fig. S5 and Table 2 ; P < 0.001 for linear trend).
All-cause mortality ( A ), cancer-specific mortality ( B ), and hazard ratios in US adults diagnosed with cancer aged 20 years or older according to the cumulative number of unfavorable SDoH. Note: Kaplan–Meier curves showed cumulative mortality probability by age and a cumulative number of unfavorable SDoH using age as the timescale. The number at risk is unweighted observed frequencies. Cumulative mortality rates were estimated with the use of survey weights. Bar chart showed hazard ratios of all-cause mortality and cancer-specific mortality associated with a number of unfavorable SDoH, adjusted for age, gender, and race and ethnicity; error bars were 95% CIs. A Compared to those with 0 unfavorable SDoH, all-cause mortality of hazard ratios (95% CI) for cancer survivors with 1, 2, 3, 4, 5, or ≥ 6 unfavorable SDoH were 1.54 (1.25–1.89), 1.81 (1.46–2.24), 2.42 (1.97–2.97), 3.22 (2.48–4.19), 3.99 (2.99–5.33), and 6.34 (4.51–8.90), respectively. B Compared to those with 0 unfavorable SDoH, cancer-specific mortality of hazard ratios (95% CI) for cancer survivors with 1, 2, 3, 4, 5, or ≥ 6 unfavorable SDoH were 1.52 (1.04–2.22), 1.70 (1.20–2.24), 2.22 (1.51–3.26), 2.44 (1.60–3.72), 3.60 (2.25–5.75), and 5.00 (3.00–8.31), respectively. ns was the abbreviation of no significance; *** meant p < 0.001, ** meant p < 0.01, and * meant p < 0.05
Age-adjusted/ age-gender-adjusted all-cause mortality and cancer-specific mortality risk were significantly higher in NHB cancer survivors when compared with NHW. Further adjustment for all SDoH factors, black-white disparity in cancer-specific mortality was still observed (HR 1.45, 95% CI 1.07–1.96), and the all-cause mortality did not show a statistically significant difference (HR, 1.08; 95% CI 0.89–1.30; Table 3 ). In the mediation analysis, the socioeconomic factor of unemployment (17.5% for all-cause mortality; 15.3% for cancer-specific mortality) can mostly explain the racial disparity in all-cause and cancer-specific mortality, and unemployment was associated with a nearly 90% and 120% greater all-cause and cancer-specific mortality, respectively. A family income-to-poverty ratio less than 2.4 (15.7%), an education less than high school (8.1%), government health insurance (6.9%), renting a home or other housing arrangement (15.4%), and not being married nor living with a partner (13.4%) indicated effective relative contribution to the disparity of all-cause mortality between NHB and NHW cancer survivors. An additional factor (not being married nor living with a partner [10.2%]) contributed significantly to the racial difference in cancer-specific mortality (Table 3 ).
In the subgroup analysis, NHW cancer survivors who were unemployed, a lower level of PIR, an education less than high school, government or none of health insurance, renting a home or other housing arrangement, and not being married nor living with a partner were significantly more likely to die of all-cause mortality compared to NHW cancer survivors without unfavorable SDoH. Unemployment and not being married nor living with a partner were significantly associated with a higher risk of cancer-specific mortality (Additional file 1: Table S6). Being unemployed and having no access to a regular health care facility or emergency room was significantly associated with all-cause mortality in NHB cancer survivors. Only unemployed status was associated with cancer-specific mortality (Additional file 1: Table S6). In the stratified analysis by gender (female and male), almost all unfavorable SDoH were significantly associated with greater all-cause and cancer-specific mortality for female and male subgroups after adjusting for age, except for cancer-specific mortality for unfavorable home ownership (Additional file 1: Table S7). In all sensitivity analyses excluding mortalities that happened during the first 2-year follow-up since the baseline interview, all results remained similar in association with unfavorable SDoH with all-cause, cancer-specific, and non-cancer mortality (Additional file 1: Table S8).
In this US nationally representative cohort study of cancer survivors, we found that NHB and Hispanic adult cancer survivors self-reported a higher proportion of multiple unfavorable SDoHs compared to NHW adults diagnosed with cancer. Compared to NHW cancer survivors, NHB cancer survivors had significantly higher all-cause and cancer-specific mortality after adjusting for age and gender. In addition, after further adjusting for all SDoH, there was no longer a difference between NHB and NHW cancer survivors in all-cause mortality, but a significant difference in cancer-specific mortality was still observed. These findings suggest that racial differences in all-cause mortality between NHW and NHB cancer survivors were largely attributable to the explained by differences in SDoH, while cancer-specific mortality disparities were partly explained by differences in SDoH. Furthermore, unfavorable SDoH were associated with a higher risk of all-cause and cancer-specific mortality for cancer survivors. During the 20 years of follow-up, an increasing number of unfavorable SDoHs in the same individual was associated with an increased risk of dying from all causes, cancer, and noncancer causes, even after adjusting for demographic factors, such as age, gender, and race. Of note, there were significantly linear dose–response relationships between the cumulative number of unfavorable SDoHs and all-cause and cancer-specific mortality among cancer survivors, and cancer survivors having six or more unfavorable SDoH increased the HR for mortality of 6.34 and 5.00 compared to those having no unfavorable SDoH, respectively.
NHB cancer survivors were more likely than NHW patients to have unfavorable levels of all SDoH. Compared to NHW cancer survivors, NHB and Hispanic cancer survivors were 3.0 times and 3.9 times more likely to experience six or more unfavorable SDoHs, respectively, which may partly explain the racial disparity in mortality. Most predominantly, NHB cancer survivors were 1.6 times more likely than NHW cancer survivors to have family PIR less than 2.4, which was associated with almost 50% and 25% greater all-cause mortality and cancer-specific mortality, respectively. Most recently, Connolly et al. [ 30 ] conducted a study involving a cohort of 3590 participants from NHANES between 1999 and 2014, and demonstrated that the SDoH level was more favorable for NHW compared to NHB adolescents. Our finding was consistent with another previous study that reported a lower level of PIR, lower level of education attachment, lack of health insurance coverage, dietary insecurity, and limited health access were more common in NHB compared to NHW, which was a key mediator in explaining race disparity in all-cause and cause-specific mortality, especially cardiovascular disease and neoplasms [ 17 , 40 ].
The persistent disparities in survival by race and ethnicity among cancer patients have been well-documented [ 2 , 3 , 4 , 6 , 41 ], and these disparities between NHB and NHW cancer survivors were particularly stark [ 42 ]. Indeed, the overall cancer mortality in 2022 for male and female together was 12% (166.8 vs. 149.3 per 100,000 persons, respectively) higher in NHB compared to NHW cancer survivors [ 6 ]. However, racial differences were not the only factor that contributed to observed mortality disparity and the underlying causes attributed to these disparities have not been well established [ 43 ]. Various factors have been suggested as contributors to these racial and ethnic disparities in survival outcomes among cancer survivors, including differences in tumor characteristics [ 44 , 45 ], neighborhood socioeconomic deprivation [ 42 ], and accessibility to health care. In the current study, disparities in the all-cause mortality HR for NHB cancer survivors compared to NHW cancer survivors decreased from 1.59 (95% CI, 1.36–1.86) to 1.09 (95% CI, 0.91–1.31) after adjusting for all SDoHs, which mostly mediated the racial disparity in all-cause mortality. With respect to cancer-specific mortality, the HR for NHB cancer survivors compared to NHW cancer survivors decreased from 2.04 (95% CI, 1.60–2.62) to 1.45 (95% CI, 1.07–1.96) after adjusting for all SDoHs, which has a partly mediator role in the racial difference. We found that cancer survivors with employed, student or retired status (17.5% relative contribution), and PIR more than 2.4 (15.7% relative contribution) explained the greatest percentage of disparities in all-cause mortality. Furthermore, we also showed that employed, student, or retired status (15.3% relative contribution) and being married or living with a partner (10.2% relative contribution) explained the largest portions of disparities in cancer-specific mortality. Taken together, the traditional socioeconomic factors consisting of household income, level of education completed, and unemployment status were important explanatory factors, that mediated around 45% and 25% of all-cause and cancer-specific mortality in survival inequities between NHB and NHW cancer survivors, respectively, which was consistent with the findings of Bundy et al. (nearly 50% mediated the differential in all-cause premature mortality) [ 25 ]. The SDoH, through an impact on occupational opportunities and income levels, have a substantial influence on insurance coverage, which was one of the main factors determining access to and delivery of health care services in the US as well as associated disparities in survival [ 40 ]. Conversely, these traditional economic factors have a greater effect on the racial/ethnic disparities in the general population compared to cancer patients. Specifically, Luo et al. [ 20 ] suggested that income mediated 62% of the association in mortality between NHB and NHW, which was consistent with the dominant contributors to family income (40%) and education (19%) to the gap between NHB and NHW adult populations [ 17 ]. Interestingly, NHW cancer survivors were approximately 25% more likely to be married or living with a partner compared to NHB cancer survivors. Being married or living with a partner was associated with the cancer-related survival benefits, possibly due to increased social support and higher psychological well-being and instrumental support, helping navigate the health care system [ 46 , 47 ]. According to Fuzzel et al. [ 48 ], barriers to health care accessibility and insurance coverage have a significant impact on rates of cancer screening, as well as the burden and attributions of the disease. These findings suggested SDoH factors, as an important mediator, drive racial health disparities, as well as all-cause and cancer-specific mortality, highlighting the necessity of the level of SDoH contexts for all people, especially those who are more vulnerable to unfavorable SDoH.
The cumulative adverse SDoHs were associated with poor all-cause survival and cause-specific survival rates among the cancer-free population have been previously reported, e.g., among patients with cardiovascular disease. Sameroff et al. [ 49 ] reported that cumulative unfavorable social risk factors, such as food insecurity combined with social isolation and loneliness, have a higher relevance to poor health outcomes than single social risk factors. Jilani et al. [ 50 ] suggested that greater SDoH adversity was linked to a higher burden of cardiovascular risk factors and poor health outcomes, such as stroke, myocardial infarction, coronary heart disease, heart failure, and mortality. Similarly, Zhang et al. [ 16 ] combined family income level, occupation, education level, and health insurance to measure socioeconomic status, and reported that participants who met low socioeconomic status had higher risks of all-cause mortality (HR, 2.13 and 95% CI, 1.90–2.38 in the US NHANES; HR, 1.96 and 95% CI, 1.87–2.06 in the UK Biobank), cardiovascular disease mortality (HR, 2.25; 95% CI, 2.00–2.53), and incident cardiovascular disease (HR, 1.65; 95% CI, 1.52–1.79) in UK Biobank, compared to high socioeconomic status. Our results were consistent with the findings of a study in which each additional SDoH conferred additional cancer-related mortality, compared to cancer survivors without any SDoH (1 SDoH [HR, 1.39; 95% CI, 1.11–1.75], 2 SDoHs [HR, 1.61; 95% CI, 1.26–2.07], and ≥ 3 SDoHs [HR, 2.09; 95% CI, 1.58–2.75]) [ 13 ]. In contrast, Weires et al. [ 51 ] observed that women with a higher socioeconomic status showed increased mortality due to breast cancer in Sweden. This finding may be due to the structure of the Swedish family cancer database (Swedes born after 1931 and their biological parents), as well as analytical restrictions on individuals 30–60 years of age in 1960, which may exclude low-socioeconomic adults with severe health problems. Previous studies have shown that these unfavorable SDoH have a tendency to cluster in individuals [ 13 , 23 ]. For example, individuals in the general US population who self-reported food insecurity were more likely to be combined with a low level of education attachment, not being married, a low level of family income, and a bad lifestyle. This finding was consistent with our observation that these unfavorable SDoH were not isolated but interrelated, and each unfavorable SDoH included in our study has been found to independently increase the risk of mortality. Compared to most previous studies based on a single SDoH, we found that there was a simple linear dose–response relationship reflecting the cumulative effect of multiple unfavorable SDoHs on all-cause and cancer-specific mortality. Collectively, these SDoH appear to synergistically increase the risk of all-cause and cancer-specific mortality among cancer survivors. However, the cumulative risk derived from a sum of the number of unfavorable SDoH assumed that all SDoH had equal and independent effects on survival outcomes, which might not be precise. We suggest that future research may need to use more complex models, such as interaction models, to more accurately capture the complex interactions of unfavorable SDoH.
Strengths and limitations
The major strength of this study was the use of large sample size data from the NHANES, which provides an opportunity to comprehensively evaluate the complex relations of SDoH with all-cause and cancer-specific mortality among cancer survivors. In addition, we focused on multiple SDoH factors and estimated the effect of accumulating unfavorable SDoH burden on mortality. We also performed mediation analysis to show the contribution of SDoH to disparities in all-cause and cancer-specific mortality. There were some limitations in the present study. First, we conducted the analyses based on the follow-up of time-to-event, however, all data on SDoH variables were only assessed at the baseline interview, which may not reflect factors that changed during the follow-up period. Therefore, our study was not able to quantify the effect of changes in eight SDoH on the mortality of cancer survivors over time. It is essential to conduct several repeated interviews about the level of SDoH during the follow-up period to reveal the influence of SDoH factors on survival among cancer survivors. Second, the assessment of SDoH was limited by the availability of variables in the NHANES database. Some SDoH such as neighborhood environment, social support, and exposure to racism, were not widely available, which may also contribute to the all-cause and cancer-specific mortality. Third, the follow-up duration was relatively short (median, 81 months) and an important bias among these cancer survivors such that socially disadvantaged who died during the study period might have had severe disease at baseline.
In conclusion, in this cohort study of a nationally representative sample of US cancer survivors between 1999 and 2018, there were significant differences in SDoH and mortality rates across self-reported racial and ethnic groups. Unfavorable SDoH were more common among NHB cancer survivors than NHW cancer survivors, were strongly associated with an increased risk of all-cause and cancer-specific mortality, and largely explained the difference between NHB and NHW cancer survivors in all-cause mortality, as well as partially explained these racial disparities in cancer-specific mortality. In addition, the cancer participants with a greater cumulative number of unfavorable SDoHs also appeared to be associated with higher risks of death from all-causes, and cause-specific (cancer and non-cancer). Taken together with previous findings, the unfavorable SDoH levels were the major risk factors for all-cause and cancer-specific mortality and were the underlying causes in all-cause racial health disparities among US cancer survivors. The entire government, civil society, local communities, businesses, and international agencies must pay more attention to the upstream SDoH, such as economic resources, employment, education quality, and racial discrimination [ 52 ]. We believe that these findings shed highlight on the cumulative burden of SDoHs on all-cause and cancer-specific mortality among cancer survivors, providing insight for ongoing and future initiatives aimed at mitigating mortality rates within vulnerable populations, including racial/ethnic minorities and individuals with an unfavorable level of SDoH status. Addressing social disparities in cancer health is a very important part of improving survival outcomes for cancer survivors, reflecting a commitment to health equity—aimed at achieving the optimal health for everyone.
Availability of data and materials
The US NHANES are publicly available database and all data can be accessed from https://wwwn.cdc.gov/nchs/nhanes/ . The statistical code and data required to reproduce the results presented in this article can be requested from Hongbo Huang ([email protected]) or Fan Li ([email protected]).
Abbreviations
- Social determinants of health
World Health Organization
National Center for Health Statistics
International Statistical Classification of Diseases, Injuries, and Causes of Death
Non-Hispanic White
Non-Hispanic Black
Hazard ratio
National Health and Nutrition Examination Survey
United States
Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024;74(1):12–49.
Article PubMed Google Scholar
Alcaraz KI, Wiedt TL, Daniels EC, Yabroff KR, Guerra CE, Wender RC. Understanding and addressing social determinants to advance cancer health equity in the United States: A blueprint for practice, research, and policy. CA Cancer J Clin. 2020;70(1):31–46.
Miller KD, Nogueira L, Mariotto AB, Rowland JH, Yabroff KR, Alfano CM, Jemal A, Kramer JL, Siegel RL. Cancer treatment and survivorship statistics, 2019. CA Cancer J Clin. 2019;69(5):363–85.
Kurani SS, McCoy RG, Lampman MA, Doubeni CA, Finney Rutten LJ, Inselman JW, Giblon RE, Bunkers KS, Stroebel RJ, Rushlow D, et al. Association of Neighborhood Measures of Social Determinants of Health With Breast, Cervical, and Colorectal Cancer Screening Rates in the US Midwest. JAMA Netw Open. 2020;3(3).
Article PubMed PubMed Central Google Scholar
Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019;69(1):7–34.
Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. 2023;73(1):17–48.
Yu Z, Yang X, Guo Y, Bian J, Wu Y. Assessing the Documentation of Social Determinants of Health for Lung Cancer Patients in Clinical Narratives. Front Public Health. 2022;10.
World Health Organization. Social determination of health. [ https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1 ].
Braveman P, Gottlieb L. The social determinants of health: it’s time to consider the causes of the causes. Public Health Rep. 2014;129 Suppl 2(Suppl 2):19–31.
A conceptual framework for action on the social determinants of health. [ https://iris.who.int/handle/10665/44489 ].
U.S Department of Health and Human Services, Office of Disease Prevention and Health Promotion. Healthy People 2030: Social Determinants of Health. [ https://health.gov/healthypeople/priority-areas/social-determinants-health ].
Fabregas JC, Riley KE, Brant JM, George TJ, Orav EJ, Lam MB. Association of social determinants of health with late diagnosis and survival of patients with pancreatic cancer. J Gastrointest Oncol. 2022;13(3):1204–14.
Pinheiro LC, Reshetnyak E, Akinyemiju T, Phillips E, Safford MM. Social determinants of health and cancer mortality in the Reasons for Geographic and Racial Differences in Stroke (REGARDS) cohort study. Cancer. 2022;128(1):122–30.
Chetty R, Stepner M, Abraham S, Lin S, Scuderi B, Turner N, Bergeron A, Cutler D. The Association Between Income and Life Expectancy in the United States, 2001–2014. JAMA. 2016;315(16):1750–66.
Article PubMed PubMed Central CAS Google Scholar
Stringhini S, Carmeli C, Jokela M, Avendaño M, Muennig P, Guida F, Ricceri F, d’Errico A, Barros H, Bochud M, et al. Socioeconomic status and the 25 × 25 risk factors as determinants of premature mortality: a multicohort study and meta-analysis of 1·7 million men and women. Lancet. 2017;389(10075):1229–37.
Zhang YB, Chen C, Pan XF, Guo J, Li Y, Franco OH, Liu G, Pan A. Associations of healthy lifestyle and socioeconomic status with mortality and incident cardiovascular disease: two prospective cohort studies. BMJ. 2021;373:n604.
Beydoun MA, Beydoun HA, Mode N, Dore GA, Canas JA, Eid SM, Zonderman AB. Racial disparities in adult all-cause and cause-specific mortality among us adults: mediating and moderating factors. BMC Public Health. 2016;16(1):1113.
Braveman PA, Egerter SA, Cubbin C, Marchi KS. An approach to studying social disparities in health and health care. Am J Public Health. 2004;94(12):2139–48.
Meneton P, Kesse-Guyot E, Méjean C, Fezeu L, Galan P, Hercberg S, Ménard J. Unemployment is associated with high cardiovascular event rate and increased all-cause mortality in middle-aged socially privileged individuals. Int Arch Occup Environ Health. 2015;88(6):707–16.
Luo J, Hendryx M, Wang F. Mortality disparities between Black and White Americans mediated by income and health behaviors. SSM Popul Health. 2022;17:101019.
Petrovic D, de Mestral C, Bochud M, Bartley M, Kivimäki M, Vineis P, Mackenbach J, Stringhini S. The contribution of health behaviors to socioeconomic inequalities in health: A systematic review. Prev Med. 2018;113:15–31.
Sun Y, Liu B, Rong S, Du Y, Xu G, Snetselaar LG, Wallace RB, Bao W. Food Insecurity Is Associated With Cardiovascular and All-Cause Mortality Among Adults in the United States. J Am Heart Assoc. 2020;9(19):e014629.
Walker RJ, Chawla A, Garacci E, Williams JS, Mendez C, Ozieh MN, Egede LE. Assessing the relationship between food insecurity and mortality among U.S. adults. Ann Epidemiol. 2019;32:43–8.
Pappas G, Queen S, Hadden W, Fisher G. The increasing disparity in mortality between socioeconomic groups in the United States, 1960 and 1986. N Engl J Med. 1993;329(2):103–9.
Article PubMed CAS Google Scholar
Bundy JD, Mills KT, He H, LaVeist TA, Ferdinand KC, Chen J, He J. Social determinants of health and premature death among adults in the USA from 1999 to 2018: a national cohort study. Lancet Public Health. 2023;8(6):e422–31.
Su LJ, O’Connor SN, Chiang TC. Association Between Household Income and Self-Perceived Health Status and Poor Mental and Physical Health Among Cancer Survivors. Front Public Health. 2021;9:752868.
Neroda P, Hsieh MC, Wu XC, Cartmell KB, Mayo R, Wu J, Hicks C, Zhang L. Racial Disparity and Social Determinants in Receiving Timely Surgery Among Stage I-IIIA Non-small Cell Lung Cancer Patients in a U.S. Southern State. Front Public Health. 2021;9:662876.
Centers for Disease Control and Prevention. National Center for Health Statistics. National Health and Nutrition Examination Survey. [ https://wwwn.cdc.gov/nchs/nhanes/ ].
Marmot M, Friel S, Bell R, Houweling TA, Taylor S. Closing the gap in a generation: health equity through action on the social determinants of health. Lancet. 2008;372(9650):1661–9.
Connolly SD, Lloyd-Jones DM, Ning H, Marino BS, Pool LR, Perak AM. Social Determinants of Cardiovascular Health in US Adolescents: National Health and Nutrition Examination Surveys 1999 to 2014. J Am Heart Assoc. 2022;11(22):e026797.
Yu Q, Li B. mma: an R package for mediation analysis with multiple mediators. J Open Res Software. 2017. https://doi.org/10.5334/jors.160 .
Yu Q, Scribner RA, Leonardi C, Zhang L, Park C, Chen L, Simonsen NR. Exploring racial disparity in obesity: A mediation analysis considering geo-coded environmental factors. Spat Spatiotemporal Epidemiol. 2017;21:13–23.
Yu Q, Wu X, Li B, Scribner RA. Multiple mediation analysis with survival outcomes: With an application to explore racial disparity in breast cancer survival. Stat Med. 2019;38(3):398–412.
Gómez CA, Kleinman DV, Pronk N, Wrenn Gordon GL, Ochiai E, Blakey C, Johnson A, Brewer KH. Addressing Health Equity and Social Determinants of Health Through Healthy People 2030. J Public Health Manag Pract. 2021;27(Suppl 6):S249-s257.
He J, Zhu Z, Bundy JD, Dorans KS, Chen J, Hamm LL. Trends in Cardiovascular Risk Factors in US Adults by Race and Ethnicity and Socioeconomic Status, 1999–2018. JAMA. 2021;326(13):1286–98.
Bickel G, Nord M, Price C, Hamilton W, Cook J. Guide to measuring household food security : revised March, 2000. Alexandria, VA: U.S. Department of Agriculture, Food and Nutrition Service, Alexandria VA. March; 2000.
Google Scholar
National Center for Health Statistics. 2019 Public-Use Linked Mortality Files. [ https://www.cdc.gov/nchs/data-linkage/mortality-public.htm ].
Korn EL, Graubard BI, Midthune D. Time-to-event analysis of longitudinal follow-up of a survey: choice of the time-scale. Am J Epidemiol. 1997;145(1):72–80.
Sattar N, Preiss D. Reverse Causality in Cardiovascular Epidemiological Research: More Common Than Imagined? Circulation. 2017;135(24):2369–72.
Islami F, Guerra CE, Minihan A, Yabroff KR, Fedewa SA, Sloan K, Wiedt TL, Thomson B, Siegel RL, Nargis N, et al. American Cancer Society’s report on the status of cancer disparities in the United States, 2021. CA Cancer J Clin. 2022;72(2):112–43.
Aizer AA, Wilhite TJ, Chen MH, Graham PL, Choueiri TK, Hoffman KE, Martin NE, Trinh QD, Hu JC, Nguyen PL. Lack of reduction in racial disparities in cancer-specific mortality over a 20-year period. Cancer. 2014;120(10):1532–9.
Ellis L, Canchola AJ, Spiegel D, Ladabaum U, Haile R, Gomez SL. Racial and Ethnic Disparities in Cancer Survival: The Contribution of Tumor, Sociodemographic, Institutional, and Neighborhood Characteristics. J Clin Oncol. 2018;36(1):25–33.
Institute of Medicine Committee on U, Eliminating R, Ethnic Disparities in Health C. In: Unequal Treatment: Confronting Racial and Ethnic Disparities in Health Care. edn. Edited by Smedley BD, Stith AY, Nelson AR. Washington (DC): National Academies Press (US). Copyright 2002 by the National Academy of Sciences. All rights reserved. 2003.
Özdemir BC, Dotto GP. Racial Differences in Cancer Susceptibility and Survival: More Than the Color of the Skin? Trends Cancer. 2017;3(3):181–97.
Virnig BA, Baxter NN, Habermann EB, Feldman RD, Bradley CJ. A matter of race: early-versus late-stage cancer diagnosis. Health Aff (Millwood). 2009;28(1):160–8.
Gomez SL, Hurley S, Canchola AJ, Keegan TH, Cheng I, Murphy JD, Clarke CA, Glaser SL, Martínez ME. Effects of marital status and economic resources on survival after cancer: A population-based study. Cancer. 2016;122(10):1618–25.
Pinquart M, Duberstein PR. Associations of social networks with cancer mortality: a meta-analysis. Crit Rev Oncol Hematol. 2010;75(2):122–37.
Fuzzell LN, Perkins RB, Christy SM, Lake PW, Vadaparampil ST. Cervical cancer screening in the United States: Challenges and potential solutions for underscreened groups. Prev Med. 2021;144:106400.
Sameroff AJ, Rosenblum KL. Psychosocial constraints on the development of resilience. Ann N Y Acad Sci. 2006;1094:116–24.
Jilani MH, Javed Z, Yahya T, Valero-Elizondo J, Khan SU, Kash B, Blankstein R, Virani SS, Blaha MJ, Dubey P, et al. Social Determinants of Health and Cardiovascular Disease: Current State and Future Directions Towards Healthcare Equity. Curr Atheroscler Rep. 2021;23(9):55.
Weires M, Bermejo JL, Sundquist K, Sundquist J, Hemminki K. Socio-economic status and overall and cause-specific mortality in Sweden. BMC Public Health. 2008;8:340.
Braveman P, Egerter S, Williams DR. The social determinants of health: coming of age. Annu Rev Public Health. 2011;32:381–98.
Download references
Acknowledgements
We thank Dr Jing Yi (School of Public Health, Chongqing Medical University, Chongqing, 400016, China) for suggestions on the used in mediation analysis and International Science Editing ( http://www.internationalscienceediting.com ) for editing this manuscript.
This study was supported by grant 82202913 (Dr Yunhai Li) and 82372996 (Dr Fan Li) from the National Natural Science Foundation of China.
Author information
Authors and affiliations.
Department of Breast and Thyroid Surgery, The First Affiliated Hospital of Chongqing Medical University, No.1 Youyi Road, Yuzhong District, Chongqing, 400016, China
Hongbo Huang, Tingting Wei, Ying Huang, Heng Zhang, Ze Zhang, Yijing Xu, Lingquan Kong, Yunhai Li & Fan Li
Health Management Center of University-Town Hospital Affiliated to Chongqing Medical University, Chongqing, 401331, China
Aijie Zhang
School of Public Health, Chongqing Medical University, Chongqing, 400016, China
You can also search for this author in PubMed Google Scholar
Contributions
HBH, YHL and FL designed the study. HBH, YHL, TTW, and YH conducted the statistical analyses. HBH, TTW, YH, AJZ, ZZ, HZ, YJX, HNP, YHL, and FL drafted the original manuscript. HBH, YHL, LQK and FL review the manuscript. All authors approved the final version of manuscript.
Corresponding authors
Correspondence to Yunhai Li or Fan Li .
Ethics declarations
Ethics approval and consent to participate.
The US NHANES database has been approved by the National Center for Health Statistics Ethics Review Board ( https://www.cdc.gov/nchs/nhanes/irba98.htm ), and written informed consent was provided by all participants.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
12916_2024_3563_moesm1_esm.docx.
Additional files 1: Method S1. Details about mediation analysis. Table S1. SDoH based on the Office of Disease Prevention and Health Promotion’s Healthy People 2030 and World Health Organization in NHANES 1999–2018. Table S2. Candidate social determinants of healthand associations with all-cause mortality in the US cancer survivors, NHANES 1999–2018. Table S3. Number of cancer survivor by cancer type and gender, NHANES 1999–2018. Table S4. P value for overall survival outcomes pairwise comparisons using log-rank test with Bonferroni adjustment. Table S5. P value for cancer-specific survival outcomes pairwise comparisons using log-rank test with Bonferroni adjustment. Table S6. Associations of social determinants of healthwith all-cause and cancer-specific mortality in US cancer survivors by race/ethnicity, NHANES 1999–2018. Table S7. Associations of social determinants of healthwith all-cause and cancer-specific mortality in US cancer survivors by gender, NHANES 1999–2018. Table S8. Sensitivity analyses of association social determinants of healthand all-cause, cancer-specific and non-cancer mortality in weighted and fully adjusted multivariable analysis among cancer survivors, NHANES 1999–2018. Fig. S1 Flowchart of participants selection for current analysis from NHANES 1999–2018. Fig. S2 The pairwise correlation between social determinants of healthusing Spearman method. Fig. S3 The proportion for each cumulative number of social determinants of healthby gender. Fig. S4 All-cause mortalityand cancer-specific mortalityfor cancer survivors aged 20 years or older in US between 1999 and 2018 stratified by cumulative number of unfavorable social determinants of health. Fig. S5 Linear dose–response association between cumulative number of unfavorable social determinants of healthand all-cause of death, and cancer deathamong US cancer survivors.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/ .
Reprints and permissions
About this article
Cite this article.
Huang, H., Wei, T., Huang, Y. et al. Association between social determinants of health and survival among the US cancer survivors population. BMC Med 22 , 343 (2024). https://doi.org/10.1186/s12916-024-03563-0
Download citation
Received : 09 March 2024
Accepted : 14 August 2024
Published : 26 August 2024
DOI : https://doi.org/10.1186/s12916-024-03563-0
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Health inequities
- Racial disparities
- Cancer survivors
BMC Medicine
ISSN: 1741-7015
- General enquiries: [email protected]

Glucose monitoring wearables for type 1 diabetes sufferers are a fad for athletes, and others are following. Let’s weigh the benefits—and dangers
B lood sugar monitors (aka continuous glucose monitors or CGMs) have been ubiquitous for people with diabetes. But now they are a fad for elite athletes and even some weekend warriors without the disease. Endurance athletes in particular have been quick to adopt this new technology to improve training and racing performance. They monitor their glucose levels with a small sensor attached to the arm or belly.
They are now widely used for training and are ubiquitous in triathlon competitions. But in 2021, cycling’s international governing body, the UCI, banned their use during competition. This ban includes, at a minimum, glucose and lactate measurement. Meanwhile, athletes in other sports are experimenting.
The fad has also crossed over to healthy people without diabetes. Fitness enthusiasts have been using smart wearables for years to track a variety of parameters such as sleep, heart rate, stress levels, and step count over the day. According to the companies that are pitching the gadget to workout enthusiasts, CGMs add another dimension, a way to improve metabolic health by optimizing blood glucose levels. Their pitch is that, even in the absence of diabetes, minimizing blood glucose spikes could prevent diabetes or other diseases.
Do CGMs work? Are they safe?
If you suffer from diabetes it’s often difficult to know the cause. Both Type 1 and Type 2 diabetes are linked in part to genetics, although the family connections are murkier in Type 1. In people with diabetes, after eating, their blood sugar, also known as blood glucose, can shoot up. That could cause organ damage if not contained.
In type 1 diabetes the immune system attacks the beta cell that produces insulin in your pancreas. The attack causes permanent damage and leaves your pancreas unable to produce insulin. Unless sufferers have access to insulin pumps and wearable devices that automatically deliver insulin, they need to inject themselves with insulin a couple of times every day.
For many, that requires pricking their finger to evaluate blood glucose levels to avoid injecting insulin when not needed. “I used to prick my fingers a couple of times a day and it was very painful,“ said Siddharth Kankaria, a science communicator who lives with type 1 diabetes.
There are multiple problems with that kind of test. Sufferers like Kankaria could only measure glucose levels up to four times a day. Plus, most monitors impose a ‘one-size-fits-all” standard. However, research has shown that two healthy people can have different responses to the same food; for example, one person’s blood sugar might spike and dip more after eating carbohydrates than another person’s.
Because of these complications, Kankaria has recently started wearing a CGM patch that connects to his phone, so he can track his glucose levels throughout the day.
Are there benefits for individuals with type 1 diabetes?
People with type 1 diabetes need to make dozens of decisions every day to keep their blood glucose in check: what to eat and when, the carbohydrate content of their meals, when to take insulin, whether and how to exercise, and so on. CGMs can reduce the cognitive load for some of these decisions.
“With a CGM, I can figure out how my body is reacting to different kinds of food, environments, and stimuli,” said Kankaria.
Advocates say they are also life-saving devices. If people with diabetes take insulin when their blood glucose level is already low, it can fall below what is healthy for them. These episodes of hypoglycemia (or low blood glucose) can cause drowsiness, anxiety, increased heart rate, and even death in extreme cases. People with diabetes can have multiple episodes per week.
Over time, repeat episodes impair awareness of early symptoms, increasing the risk of severe episodes. A PLOS One study on South Indian patients noted that 44 of 73 patients were unaware of their hypoglycemia .
In the absence of early symptoms, there is some evidence that CGMs can prevent low blood sugar episodes. Conversely, not taking sufficient insulin or eating carb-heavy foods can cause hyperglycemia. While blood sugar levels quickly revert to normal in healthy individuals, they may stay outside the healthy range for a prolonged time in people with type 1 diabetes.
“I use the CGM to check if I have hypoglycemia,” said Kankaria. “I can see it going down in real-time and intervene before the blood glucose level is too low.”
Some experts are skeptical of the utility of CGMs
That’s debatable. The use of glucose monitors for those with the disease remains controversial in some medical circles. Hyperglycemia (or high blood glucose) is a symptom of diabetes, not its cause. CGMs also do not measure glucose levels in the blood but in the interstitial fluid surrounding cells. Changes in blood glucose levels reflect changes in interstitial glucose levels after 5-20 minutes. In other words, if a CGM shows higher or lower than normal glucose levels, that’s likely old news. This lag is more pronounced when blood glucose levels change rapidly, such as immediately after large meals or during exercise.
CGM readings can be inaccurate for a variety of other reasons. For example, sleeping on the arm where the sensor is can cause pressure-induced errors, producing false lows. Conversely, drugs like acetaminophen (Tylenol) that cause chemical interference in the interstitial fluid can produce false highs.
These monitors often tell what we already know or do not need to know, some experts say. Digestion breaks down carbohydrates in the food into glucose and other sugars, causing a spike in blood sugar levels in all of us. In response, the body produces insulin which usually brings the glucose levels down to pre-meal levels within two hours. In other words,
Glucose levels straying outside what is considered the normal range for a healthy individual is physiologically typical. In any case, when it occurs it’s for brief periods. In a study published in The Journal of Clinical Endocrinology & Metabolism , researchers found that the blood glucose levels for healthy individuals were normal 96% of the time .
In some people with type 1 diabetes, the anxiety of hypoglycemic episodes can cause excessive focus on correcting glucose levels. This can sometimes lead to orthorexia nervosa, an unhealthy fixation with healthy eating. Almas Fatma, a Mumbai-based diabetologist, added that non-diabetic individuals can also develop orthorexia nervosa if they let glucose readings to obsessively monitor their diet. In one case, a patient avoided all food that would cause even minor spikes in their CGM reading. It grew to a point where this individual ate only salads. “Over time, the restrictive diet led to multiple nutritional deficiencies and a significant loss of muscle mass,” said Fatma.
Use in sports?
Their use by those without diabetes crosses a line for many medical practitioners.
“There is a lack of clinical evidence to support the use of CGM in healthy individuals, Fatima warned. Companies selling CGMs to healthy people are pathologizing clinically insignificant fluctuations. With constant access to blood glucose levels, “even healthy people can get fixated on minor fluctuations and readings and it can have a negative impact on their mental health and quality of life”, she said.
Another example. While weight management is a common fitness goal, obsessing over glucose spikes can ironically impede it. If a user takes corrective steps every time CGM says their glucose is approaching lower or higher than the normal range, they might end up eating more or less than they need to.
Many other health officials agree that the benefits do not clearly outweigh the potential downside. “There is no strong evidence the gadgets help people without the condition,” UK National Health Service diabetes advisor Prof Partha Kar recently said .
But an increasing number of elite athletes and their coaches swear by them. “Having insight into the body’s individual response to carbohydrate and protein intake while racing will help you minimize unneeded fueling and excessive exposure to sugars,” said Suzanne Atkinson , an emergency medicine physician and elite-level triathlon coach.
Supporters maintain that CGMs can tell athletes what carbs they should eat to optimally refuel glycogen reserves. Additionally, pro athletes need to maintain their weight in a narrow range. A persistently low glucose level could hint at insufficient calorie intake before it’s evident as weight loss.
However, just like those with type 1 diabetes, elite athletes who use CGMs are best monitored by physicians, who can contextualize their data. In the absence of this important distinction, CGM use among non-diabetic fitness enthusiasts could cause mental and physical harm that supersedes any potential benefits . I n sum, people with type 1 diabetes, episodes of hypoglycemia or hyperglycemia can be a matter of life and death and, understandably, cause significant mental distress. For those individuals, CGM provides one more information resource.
But for non-diabetic users, the measurements from a CGM, more so if they are tracking other vitals, can cause an information overload. Some healthy people using CGMs worry unnecessarily about minor glucose spikes, even if it’s after eating a healthy meal. The use of CGM among healthy people can cause harm if a user tries to obsessively keep the glucose levels in the “healthy” range.
Sachin Rawat is a freelance science and tech writer based in Bangalore. He holds a master’s degree in biotechnology. Follow him on Twitter at @sachinxr .

GLP Podcasts & Podcast Videos More...

GLP podcast: Hot dogs probably don’t cause dementia; Subsidize plant burgers? Drinking fathers might damage fetal brain development

GLP podcast: Treating brain diseases with parasites; FDA rejects ecstasy treatment for PTSD; $6 billion—the cost of baby powder lawsuits
Videos more....

Video: Organic, non-gmo, gluten-free, keto? How food labels mislead us about health benefits
Bees & pollinators more....

Mosquito massacre: Can we safely tackle malaria with a CRISPR gene drive?

Are we facing an ‘Insect Apocalypse’ caused by ‘intensive, industrial’ farming and agricultural chemicals? The media say yes; Science says ‘no’

Dissecting claims about Monsanto suing farmers for accidentally planting patented seeds
Infographics more....

Infographic: Global regulatory and health research agencies on whether glyphosate causes cancer
Gmo faqs more....

Why is there controversy over GMO foods but not GMO drugs?

How are GMOs labeled around the world?

How does genetic engineering differ from conventional breeding?
| GLP Profiles |

Alex Jones: Right-wing conspiracy theorist stokes fear of GMOs, pesticides to sell ‘health supplements’

IARC (International Agency for Research on Cancer): Glyphosate cancer determination challenged by world consensus
Most popular.

Newsletter Subscription
- Weekly Newsletter (Wed)
- Daily Digest (Mon, Tue, Thu, Fri)
- Weekly Top Six (Sun)
- Featured Articles Only
- Human Articles Only
- Agriculture Articles Only
- All Types of Content
Get news on human & agricultural genetics and biotechnology delivered to your inbox.

IMAGES
COMMENTS
A 44-year-old woman presented with cough, dyspnea, and chest pain. On examination, she had tachycardia and hypotension. Evaluation revealed SARS-CoV-2 RNA in a nasopharyngeal swab, as well as eleva...
This case was presented at the Harvard Medical School postgraduate course, "Primary Care Internal Medicine 2018," directed by John D. Goodson, M.D. No potential conflict of interest relevant ...
An 81-year-old man presented with fever, cough, and shortness of breath. Within a few hours after presentation, chest pain and respiratory distress developed. A chest radiograph showed bilateral pa...
A case report is a detailed report of the symptoms, signs, diagnosis, treatment, and follow-up of an individual patient. Case reports usually describe an unusual or novel occurrence and as such, remain one of the cornerstones of medical progress and provide many new ideas in medicine. Some reports contain an extensive review of the relevant ...
First steps. Begin by sitting down with your medical team to discuss the interesting aspects of the case and the learning points to highlight. Ideally, a registrar or middle grade will mentor you and give you guidance. Another junior doctor or medical student may also be keen to be involved. Allocate jobs to split the workload, set a deadline ...
Editorial. Introduction. Case reports and case series or case study research are descriptive studies to present patients in their natural clinical setting. Case reports, which generally consist of three or fewer patients, are prepared to illustrate features in the practice of medicine and potentially create new research questions that may contribute to the acquisition of additional knowledge ...
Some of the case studies, for example the heart failure study, show clinical data before and after drug treatments so the students can get a feel for mechanism in physiological action. ... This text contains 8 medical case studies that reflect best practices at the time of publication. The text identifies 5 overarching learning objectives ...
A 53 year old man presents to clinic with swelling of his hands and a uric acid of 12. 15. A 58-year-old woman presents to clinic with difficulty walking. 16. A 49-year-old woman is seen with an abnormal Nerve Conduction Study. 17. A 55-year-old woman is seen because of her right knee is "giving out". 18.
Brought to you by Merck & Co, Inc., Rahway, NJ, USA (known as MSD outside the US and Canada) — dedicated to using leading-edge science to save and improve lives around the world. Learn more about the MSD Manuals and our commitment to Global Medical Knowledge. About. Disclaimer. Permissions.
Case study is a research methodology, typically seen in social and life sciences. There is no one definition of case study research.1 However, very simply… 'a case study can be defined as an intensive study about a person, a group of people or a unit, which is aimed to generalize over several units'.1 A case study has also been described as an intensive, systematic investigation of a ...
A case study report is an academic publication describing an unusual or unique case. Academic medical journals publish case study reports to inform and educate other medical practitioners. Case study reports might also prompt additional scholarly research on the medical condition or treatment plan discussed in the report.
AccessMedicine Case Files Collection This link opens in a new window AccessMedicine's Case Files Collection from McGraw-Hill Education offers the best-selling Case Files content in an interactive format. Updated regularly, this comprehensive case collection helps students learn and apply basic science and clinical medicine concepts in the context of realistic patient cases.
The LITFL Clinical Case Collection includes over 250 Q&A style clinical cases to assist ' Just-in-Time Learning ' and ' Life-Long Learning '. Cases are categorized by specialty and can be interrogated by keyword from the Clinical Case searchable database. Search by keywords; disease process; condition; eponym or clinical features….
A 68-year-old man was admitted to the hospital with fever, shortness of breath, and acute kidney injury. Testing of a nasopharyngeal swab for SARS-CoV-2 RNA was positive. Respiratory failure and hy...
Presenting patient cases is a key part of everyday clinical practice. A well delivered presentation has the potential to facilitate patient care and improve efficiency on ward rounds, as well as a means of teaching and assessing clinical competence.1 The purpose of a case presentation is to communicate your diagnostic reasoning to the listener, so that he or she has a clear picture of the ...
CASE STUDIES. This page offers a collection of interesting cases from the Penn Department of Pathology and Laboratory Medicine that are available to download as PDFs. To view specific case studies by organ system or subspecialty, use the filter checkboxes in the left sidebar.
Each scenario allows you to work through history taking, investigations, diagnosis and management. You might also be interested in our bank of 1000+ OSCE Stations. A collection of interactive medical and surgical OSCE cases (clinical case scenarios) to put your history, examination, investigation, diagnostic and management skills to the test.
Case Studies. In this section we report a variety of clinical cases in an effort to share our experience with healthcare providers and with patients who may benefit. The cases are categorized into a wide variety of different medical and surgical specialties listed under subheadings. The main objective is to share our knowledge and experience ...
Case Study example: In the PubMed Search box, type: Diabetes Mellitus, Type 2/th Then filter to: Complementary Medicine and/or Dietary Supplements under Subjects.
Observed peaks in reports of SSNHL at 1 and 7 days after vaccination in both VAERS and our case series could be partly accounted for by recall bias, which has been well documented in studies of passive vaccine AE reporting. 17,18 For example, an analysis of AEs associated with the hepatitis B vaccine, in which patient self-reports were cross ...
Dr. SooJeong Youn: This case highlights the importance of attending to the intricate, multilevel, systemic factors that affect the mental health experience and clinical presentation of patients ...
Case Studies: Functional Medicine Success Stories. Let's look at real-life examples of how functional medicine has helped women with hormonal imbalances. Case Study 1: Sarah's Journey to Balance. Sarah was in her mid-30s and struggling with fatigue, weight gain, and irregular periods. Traditional treatments weren't helping, so she turned ...
Also, the required sample size of a crossover trial is smaller than that of an RCT, which better solves the problem of limited number of subjects and improves the evidence-based medicine evidence for the treatment of CHF with TCM comprehensive intervention . These will make up the shortcomings of the previous studies and make potential results ...
Radiology Pathology Correlation Case Studies. 2017-2018. 2018-2019. 2019-2020. Early Specialization in Interventional Radiology (ESIR) Diagnostic Radiology/Nuclear Medicine (DR/NM) Pathway. Angiography and Interventional Radiology Observership. Verification of Graduate Medical Education Training and Faculty Appointment. Each student is required ...
Background Racial and ethnic disparities in mortality persist among US cancer survivors, with social determinants of health (SDoH) may have a significant impact on these disparities. Methods A population-based cohort study of a nationally representative sample of adult cancer survivors, who participated in the US National Health and Nutrition Examination Survey from 1999 to 2018 was included ...
On examination, the patient appeared to be in respiratory distress. The temperature was 35.9°C, the pulse 98 beats per minute, the blood pressure 129/58 mm Hg, the respiratory rate 24 breaths per ...
In any case, when it occurs it's for brief periods. In a study published in The Journal of Clinical Endocrinology & Metabolism, researchers found that the blood glucose levels for healthy ...
A Man with Confusion, Fatigue, and Garbled Speech. A 63-year-old man with a history of primary membranous nephropathy was evaluated because of fever, confusion, headache, garbled speech, fatigue ...
Virality, the sum of user interactions with online content, serves not only as an effectiveness metric, but also as a signal of message quality and acceptance (Alhabash et al., 2015).It is a crucial factor in the spread of influencers' misinformation on social media, as the virality of social media content increases the spread of information (Visentin et al., 2021), shapes perceptions of ...