Recent Approaches and Advancements in Natural Dyes
- First Online: 24 January 2024

Cite this chapter

- Silpinwita Das 3 &
- Sankar Roy Maulik ORCID: orcid.org/0000-0002-3065-641X 3
Part of the book series: Sustainable Textiles: Production, Processing, Manufacturing & Chemistry ((STPPMC))
283 Accesses
1 Citations
Natural dyes have played significant roles in shaping and rewriting history worldwide. Natural colorants, mainly from plant and animal origins, have been used for dyeing various things. They bear a crucial historical testimony that widens the horizon of looking at the glorious past and promising future. Recently, the detrimental environmental pollution caused by synthetic dyes has created room to reconsider natural dyes as alternative sources. The newly discovered natural dye sources and nontoxic and sustainable processes for their application have broadened the spectrum of natural dyes that can be employed in traditional and newer interventions. Today, advanced, optimized, and unconventional techniques, such as ultrasonic and microwave-assisted extraction, have been applied contrary to conventional extraction to obtain the maximum color yield with minimum environmental pollution. Statistically designed programs are now commonly exploited for optimization and process modeling. In recent years, relatively novel and scarcely investigated microbial dyes were being utilized to achieve high production yields, bright colors, and unique functional properties. These natural dye sources can also positively affect the textile industry and the environment by reducing water and contamination, energy consumption, etc. Besides textiles, eco-leather is now becoming an option for environmentally friendly, nontoxic leather production by overcoming the limitation of utilizing natural dyes. These dyes set the trend for the textile industry from being fashionable to functional for their innocuous, antiallergenic, and biodegradable qualities. More recently, certain artificial color additives have demonstrated that they can lead to negative health issues following their consumption. On the other hand, natural colors have been proven safe in these areas. Therefore, research into newer sources of pigments and their occurrence is coming to the front to find natural alternatives for synthetic dyes. Natural dyes in food, beverages, pharmaceuticals, etc. enhance their functional and aesthetic impressions. Moreover, the intelligent food packaging industry is trying to adopt natural dyes to monitor the freshness of foodstuffs, although color stability in different circumstances of these dyes still needs to be worked on. In the past few decades, natural dyes have been considered the feasible alternatives to the toxic, expensive, and rare inorganic sensitizers in solar cells. Natural sensitizers containing plant pigments responsible for chemical reactions absorb sunlight’s photons and convert them into electrical energy.
This review provides comprehensive breakthroughs in using newer ways of applying natural dyes, with technological developments in the functional finishing of textiles, food coloration, food packaging, cosmetics, leather, dye-sensitized solar cells, microencapsulation, etc.
This is a preview of subscription content, log in via an institution to check access.
Access this chapter
Subscribe and save.
- Get 10 units per month
- Download Article/Chapter or eBook
- 1 Unit = 1 Article or 1 Chapter
- Cancel anytime
- Available as PDF
- Read on any device
- Instant download
- Own it forever
- Available as EPUB and PDF
- Durable hardcover edition
- Dispatched in 3 to 5 business days
- Free shipping worldwide - see info
Tax calculation will be finalised at checkout
Purchases are for personal use only
Institutional subscriptions
Abdel-Latif, M., Abuiriban, M., Dahoudi, N., Al-Kahlout, A., Taya, S., El-Agez, T., & El-Ghamri, H. (2015). Dye-sensitized solar cells using fifteen natural dyes as sensitisers of nanocrystalline TiO2. Science, Technology and Development, 34 (3), 135–139.
Article Google Scholar
Adedokun, O., Titilope, K., & Awodugba, A. O. (2016). Review on natural dye-sensitized solar cells (DSSCs). International Journal of Engineering Technologies, 2 (2), 34–41.
Google Scholar
Aggarwal, S. (2021). Indian dye yielding plants: Efforts and opportunities. Natural Resources Forum, 45 (1), 63–86. Blackwell Publishing Ltd.
Ahmad, M. S., Pandey, A. K., & Abd Rahim, N. (2017). Advancements in the development of TiO2 photoanodes and its fabrication methods for dye-sensitized solar cell (DSSC) applications. A review. Renewable and Sustainable Energy Reviews, 77 , 89–108.
Alam, S. S., Ghosh, J., & Das, D. (2022). The coloration of cotton fabric with natural dye extracted from turmeric powder. Journal of Textile Engineering and Fashion Technology, 8 (4), 134–138.
Ali, N. F. (2018). Green strategy for the production of antimicrobial textiles. In Fashion and textiles: Breakthroughs in research and practice (pp. 442–461). IGI Global.
Chapter Google Scholar
Alizadeh-Sani, M., Mohammadian, E., Rhim, J. W., & Jafari, S. (2020). pH-sensitive (halochromic) smart packaging films based on natural food colourants for the monitoring of food quality and safety. Trends in Food Science and Technology, 105 , 93–144.
Article CAS Google Scholar
Allam, K., & Kumar, G. (2011). Colourants the cosmetics for the pharmaceutical dosage forms. International Journal of Pharmacy and Pharmaceutical Sciences, 3 (3), 13–21.
Amaya, E., & Nickell, D. (2015). 11 – Using feed to enhance the colour quality of fish and crustaceans. In I. D. A. Davis (Ed.), Feed and feeding practices in aquaculture (pp. 269–298). Woodhead Publishing.
Amogne, N., Ayele, D., & Tsigie, Y. (2020). Recent advances in anthocyanin dyes extracted from plants for dye. Materials for Renewable and Sustainable Energy, 9 (23), 1–16. https://doi.org/10.1007/s40243-020-00183-5
Amorim, L., Mouro, C., Riool, M., & Gouveia, I. (2022). Antimicrobial food packaging based on prodigiosin-incorporated double-layered bacterial cellulose and chitosan composites. Polymers, 14 (2). https://doi.org/10.3390/polym14020315
Askar, K., Alsawad, Z., & Khalaf, M. (2015). Evaluation of the pH and thermal stabilities of rosella anthocyanin extracts under solar light. Beni-Suef University Journal of Basic and Applied Sciences, 4 (3), 262–268.
Azman, N., Khairul, W., & Sarbon, N. (2022). A comprehensive review on biocompatible film sensor containing natural extract: Active/intelligent food packaging. Food Control, 141 , 109189. https://doi.org/10.1016/j.foodcont.2022.109189
Baig, G. A. (2011). Dyeing of polyester fabrics with indigo. Research Journal of Textile and Apparel, 15 (4), 19–25.
Bentes, A., de Souza, A. L., Amaya-Farfan, H., Lopes, J., & Faria, A. (2015). Influence of the composition of unripe genipap ( Genipa americana L.) fruit on the formation of blue pigment. Journal of Food Science and Technology, 52 (6), 3919–3924.
CAS Google Scholar
Bhargava, N., Sharanagat, V., Mor, R., & Kumar, K. (2020). Active and intelligent biodegradable packaging films using food and food waste-derived bioactive compounds: A review. Trends in Food Science & Technology, 105 , 385–401.
Bin-Jumah, M., Alwakeel, S. S., Moga, M., Buvnariu, L., Bigiu, N., & Zia-Ul-Haq, M. (2021). Application of carotenoids in cosmetics. Carotenoids: Structure and Function in the Human Body , 747–756.
Biswal, P., Mishar, M., Bhadouriya, A., & Yadav, V. (2015). An updated review on colorants as pharmaceutical excipients. International Journal of Pharmaceutical, Chemical and Biological Sciences, 5 (4), 1004–1017.
Boranbayeva, T., Karadeniz, F., & Yılmaz, E. (2014). Effect of storage on anthocyanin degradation in black mulberry juice and concentrates. Food and Bioprocess Technology, 7 , 1894–1902.
Borges, M., Tejera, R., Díaz, L., Esparza, P., & Ibáñez, E. (2012). Natural dyes extraction from cochineal (Dactylopius coccus). New extraction methods. Food Chemistry, 132 (4), 1855–1860.
Brauch, J., Zapata-Porras, S., Buchweitz, M., Aschoff, J., & Carle, R. (2016). Jagua blue derived from Genipa americana L. fruit: A natural alternative to commonly used blue food colorants? Food Research International, 89 (1), 391–398.
Brudzyńska, P., Sionkowska, A., & Grisel, M. (2021). Plant-derived colorants for food, cosmetic and textile industries: A review. Materials, 14 (13), 3484.
Bueno, A., Hoffmann, T., Souza, C., Carvalho, L., Bertoli, S., Barcellos, I., & Gonçalves, M. (2022). Optimal process conditions for recycled polyester dyeing using natural annatto dye. Journal of Cleaner Production, 370 .
Bujak, T., Zagórska-Dziok, M., Ziemlewska, A., Nizioł-Łukaszewska, Z., Lal, K., Wasilewski, T., & Hordyjewicz-Baran, Z. (2022). Flower extracts as multifunctional dyes in the cosmetics industry. Molecules, 27 (3), 922.
Chandru, P., & Praveena, R. (2021). Organic hybrid pigments from Crotalaria globosa for cotton dyeing – A sustainable approach. Materials Today: Proceedings, 47 (9), 1773–1776.
Chaudhari, N., Chaudhari, N., Chaudhari, H., Premchandani, L., Dhankani, A., & Pawar, S. (2018). A review on herbal lipstick from different natural colouring pigment. Indian Journal of Drugs, 6 (3), 174–179.
Chen, H.-Z., Zhang, M., Bhandari, B., & Yang, C.-H. (2020a). Novel pH-sensitive films containing curcumin and anthocyanins to monitor fish freshness. Food Hydrocolloids, 100 . https://doi.org/10.1016/j.foodhyd.2019.105438
Chen, S., Wu, M., Lu, P., Gao, L., Yan, S., & Wang, S. (2020b). Development of pH indicator and antimicrobial cellulose nanofibre packaging film based on purple sweet potato anthocyanin and oregano essential oil. International Journal of Biological Macromolecules, 149 , 271–280.
Chequer, F. D., De Oliveira, G. R., Ferraz, E. A., Cardoso, J. C., Zanoni, M. B., & De Oliveira, D. P. (2013). Textile dyes: Dyeing process and environmental impact. Eco-friendly Textile Dyeing and Finishing, 6 (6), 151–176.
Coultate, T., & Blackburn, R. S. (2018). Food colorants: Their past, present and future. Coloration Technology, 134 (3), 165–186.
Cui, H., Xie, W., Hua, Z., Cao, L., Xiong, Z., Tang, Y., & Yuan, Z. (2022). Recent advancements in natural plant colorants used for hair dye applications: A review. Molecules, 27 (22), 8062.
Cuong, D. X., Hoan, N. X., Dong, D. H., Thuy, L. T. M., Van Thanh, N., Ha, H. T., et al. (2019). Tannins: Extraction from plants. Tannins-Structural Properties, Biological Properties, and Current Knowledge , 57–76.
Das, S. K., Ganguli, S., Kabir, H., Khandaker, J., & Ahmed, F. (2020). Performance of natural dyes in dye-sensitized solar cells as photosensitizer. Transactions on Electrical and Electronic Materials, 21 , 105–116.
Dave, H., Ledwani, L., & Nema, S. (2016). Surface modification by atmospheric pressure air plasma treatment to improve dyeing with natural dyes is an environment-friendly approach for leather processing. Plasma Chemistry and Plasma Processing, 36 , 599–613.
Devadiga, D., & Ahipa, T. (2020). Betanin: A red-violet pigment – Chemistry and applications. In A. K. Samanta, N. S. Awwad, & H. Algarni (Eds.), Chemistry and technology of natural and synthetic dyes and pigments . https://doi.org/10.5772/intechopen.88939
Elshemy, N. (2011). Unconventional natural dyeing using microwave heating with cochineal as natural dyes. Research Journal of Textile and Apparel, 15 (4), 26–36.
Frimpong, G., Adotey, J., Ofori-Kwakye, K., Kipo, S., & Dwomo-Fokuo, Y. (2014). Investigating the suitability of Hibiscus sabdariffa calyx extract as a colouring agent for paediatric syrups. Journal of Applied Pharmaceutical Science, 4 (12). https://doi.org/10.7324/JAPS.2014.41201
Gokilamani, N., Muthukumarasamy, N., Thambidurai, M., Ranjitha, A., & Velauthapillai, D. (2013). Utilization of natural anthocyanin pigments as photosensitizers for dye-sensitized solar cells. Journal of Sol-Gel Science & Technology, 66 , 212–219.
Gómez-Ortíz, N., Vázquez-Maldonado, I., Pérez-Espadas, A., Mena-Rejón, G., Azamar-Barrios, J., & Oskam, G. (2010). Dye-sensitized solar cells with natural dyes extracted from achiote seeds. Solar Energy Materials and Solar Cells, 94 (1), 40–44. https://doi.org/10.1016/j.solmat.2009.05.013
Gulrajani, M. (1999). Natural dyes. Part I: Present status of natural dye. Colourage, 46 (7), 19–28.
Gulrajani, M. (2001). Present status of natural dyes. Indian Journal of Fibre & Textile Research, 26 (1–2), 191–201.
Gulrajani, M., & Gupta, D. (1992). Natural dyes and their application to textiles . IIT New Delhi.
Haji, A., Ghaheh, F., & Indrie, L. (2022). Pomegranate fallen leaves as a source of natural dye for the mordant-free dyeing of wool. Coloration Technology . https://doi.org/10.1111/cote.12651
Halonen, N., Pálvölgyi, P., Bassani, A., Fiorentini, C., Nair, R., Spigno, G., & Kordas, K. (2020). Bio-based smart materials for food packaging and sensors – A review. Frontiers in Materials . https://doi.org/10.3389/fmats.2020.00082
Hao, S., Wu, J., Huang, Y., & Lin, J. (2006). Natural dyes as photosensitizers for dye-sensitized solar cell. In X. Mathew (Ed.), Solar energy (pp. 209–214). Elsevier.
Haq, Z. U., Riaz, M., Saad, M., Riaz, B., Zia-Ul-Haq, M., & Saad, B. (2016). Biosynthesis and stability of anthocyanins. Springer International Publishing, 1 (1), 71–86.
Hasby, N., Pamungkas, G., & Akbar, S. A. (2020). Dye-sensitized solar cells properties from natural dye as light-reaping materials extracted from Gayo arabica coffee husks. Rasayan Journal of Chemistry, 13 (1), 38–43.
Hosseinnezhad, M., Moradian, S., & Gharanjig, K. (2015). Fruit extract dyes as photosensitizers in solar cells. Current Science, 109 (5), 953–956.
Hosseinnezhad, M., Gharanjig, K., Rouhani, S., Imani, H., & Razani, N. (2022). Environmentally dyeing using dried walnut husk as bio-mordant: Investigation of creating new red and yellow shades on wool. Journal of Natural Fibers, 19 (15), 10953–10963.
Jabar, J., Owokotomo, I., & Ogunsade, A. (2022). Sustainable dyeing of cotton fabric with mangiferin: Roles of microwave-rays and bio-mordants on fabric colorimetric and fastness properties. Sustainable Chemistry and Pharmacy, 29 , 100822.
Jan, B., Parveen, R., Zahiruddin, S., Khan, M., Mohapatra, S., & Ahmad, S. (2021). Nutritional constituents of mulberry and their potential applications in food and pharmaceuticals: A review. Saudi Journal of Biological Sciences, 28 (7), 3909–3921.
Kabir, F., Bhuiyan, M. M. H., Manir, M. S., Rahaman, M. S., Khan, M. A., & Ikegami, T. (2019). Development of dye-sensitized solar cell based on the combination of natural dyes extracted from Malabar spinach and red spinach. Results in Physics, 14 , 102474.
Kassinger, R. (2003). Dyes: From sea snails to synthetics . Twenty-First Century Books.
Kramar, A., & Kostic, M. (2022). Bacterial secondary metabolites as biopigments for textile dyeing. Text, 2 (2), 252–264.
Křížová, H. (2015). Natural dyes: Their past, present, future and sustainability. In Recent developments in fibrous material science (pp. 59–71). Kosmas Publishing.
Latos-Brozio, M., & Masek, A. (2020). The application of natural food colorants as indicator substances in intelligent biodegradable packaging materials. Food and Chemical Toxicology, 135 .
Lee, S., Shin, E., Kim, W., & Park, S. (2012). Dyeing process for improving properties of black color using natural dyes and mordant. Journal of the American Leather Chemists Association, 107 (2), 33–39.
Li, A., Xiao, R., He, S., An, X., He, Y., Wang, C., et al. (2019). Research advances of purple sweet potato anthocyanins: Extraction, identification, stability, bioactivity, application, and biotransformation. Molecules, 24 (21). https://doi.org/10.3390/molecules24213816
Li, X., Liu, D., Pu, Y., & Zhong, Y. (2023). Recent advances of intelligent packaging aided by artificial intelligence for monitoring food freshness. Food, 12 (15), 2976. https://doi.org/10.3390/foods12152976
Liu, J., Li, W., Kang, X., Zhao, F., He, M., She, Y., & Zhou, Y. (2021). Profiling by HPLC-DAD-MSD reveals a 2500-year history of the use of natural dyes in Northwest China. Dyes and Pigments, 187 . https://doi.org/10.1016/j.dyepig.2021.109143
Mahmoud, R., & Adelzaher, N. (2022). Investigation of thermal and structure properties of silk dyed with a natural dye. Letters in Applied NanoBioScience, 12 (2).
Martins, V., Santos, L., Romani, V., & Fernandes, S. (2022). Bio-based sensing: Role of natural dyes in food freshness indicators. In A. Shukla (Ed.), Bio- and nano-sensing technologies for food processing and packaging (pp. 37–62). The Royal Society of Chemistry.
Meshram, S., Limaye, R., Ghodke, S., Nigam, S., Sonawane, S., & Chikate, R. (2011). Continuous flow photocatalytic reactor using ZnO–bentonite nanocomposite for degradation of phenol. Chemical Engineering Journal, 172 (2–3), 1008–1015.
Mirnezhad, S., Sadeghi-Kiakhani, M., & Hashemi, E. (2022). Wool dyeing using Ziziphus bark extract as a natural dye: Studies on the dyeing, antibacterial and antioxidant characteristics. Environmental Science and Pollution Research . https://doi.org/10.21203/rs.3.rs-1894001/v1
Mridha, A., Mukhopadhyay, A., Das, D., Sarkar, B., & Roy Maulik, S. (2021). Sustainable dyeing of silk with natural dyes using natural sources of water. Journal of The Institution of Engineers (India): Series E, 102 (1), 61–74.
Mussak, R. A., & Bechtold, T. (2009). Natural colorants in textile dyeing. In Handbook of natural colorants (pp. 315–335).
Narayan, M. R. (2012). Dye sensitized solar cells based on natural photosensitizers. Renewable & Sustainable Energy Reviews, 16 (1), 208–215.
Natarajan, T., & Jayadevan, G. R. (2022). COVID-19 pandemic and the consumption behaviour of branded functional beverages in India: A conceptual framework. Nutrition & Food Science, 52 (3), 423–444.
Neri-Numa, I., Angolini, C., Bicas, J., GoisRuiz, A., & Pastore, G. (2018). Iridoid blue-based pigments of Genipa americana L. (Rubiaceae) extract: Influence of pH and temperature on color stability and antioxidant capacity during in vitro simulated digestion. Food Chemistry, 263 , 300–306.
Neri-Numa, I., Pessôa, M., Arruda, H., Pereira, G., Paulino, B., Angolini, R. C., & Pastore, G. (2020). Genipap (Genipa americana L.) fruit extract as a source of antioxidant and antiproliferative iridoids. Food Research International, 134 . https://doi.org/10.1016/j.foodres.2020.109252
Nguyen, T., & Nguyen, H. (2022). Green cosmetic from natural colorant of anthocyanin extracted from Ipomoea batatas L. Materials Science Forum , 41–50.
Nuryanti, S., Puspitasari, D., & Supriadi. (2019). Rosella (Hibiscus sabdariffa) flowers as alternative indicators of blue and red litmus. Oriental Journal of Chemistry, 35 (1), 476–480.
Ondruschka, B., & Asghari, J. (2006). Microwave-assisted extraction–a state-of-the-art overview of varieties. Chimia, 60 (6), 321–321.
Otaviano, B., Sannomiya, M., Lima, F., Tangerina, M., Tamayose, C., Ferreira, M., et al. (2022). Pomegranate peel extract and zinc oxide as a source of natural dye and functional material for textile fibers aiming for photoprotective properties. Materials Chemistry and Physics, 293 .
Pancapalaga, W., Ishartati, E., & Ambarwati, T. (2022). The Colorfastness and quality of eco-printed leather with different types of mordant in natural dyes from mangrove extract (Rhizophora mucronata). Tropical Animal Science Journal, 45 (3).
Pizzicato, B., Pacifico, S., Cayuela, D., Mijas, G., & Riba-Moliner, M. (2023). Advancements in sustainable natural dyes for textile applications: A review. Molecules, 28 (16), 5954.
Prabavathy, N., Shalini, S., Balasundaraprabhu, R., Prasanna, S., Prasanna, S., & Muthukumarasamy, N. (2017). Enhancement in the photostability of natural dyes for dye-sensitized solar cell (DSSC) applications: A review. International Journal of Energy Research, 41 (10), 1372–1396.
Pratiwi, D., Nurosyid, F., Supriyanto, A., & Suryana, R. (2016). Optical properties of natural dyes on the dyesensitized solar cells (DSSC) performance. In Journal of physics: Conference series . IOP Publishing. https://doi.org/10.1088/1742-6596/776/1/012007
Puri Sikka, M., Sarkar, A., & Garg, S. (2022). Artificial intelligence (AI) in textile industry operational modernization. Research Journal of Textile and Apparel . https://doi.org/10.1108/RJTA-04-2021-0046
Rani, N., & Jajpura, L. (2021). Sustainable dyeing of wool by natural dyes in conjunction with natural mordants. In Recent trends in traditional and technical textiles (pp. 127–144). Springer.
Reddy, A., Moniruzzaman, M., Madhavi, V., & Jaafar, J. (2020). Chapter 8 – Recent improvements in the extraction, cleanup, and quantification of bioactive flavonoids. In Atta-ur-Rahman (Ed.), Studies in natural products chemistry (Vol. 66, pp. 197–223). Elsevier.
Rehana, M., Ibrahim, G., Mashaly, H., Hasanin, M., GamalRashad, H., & Mowafi, S. (2022). Simultaneous dyeing and multifunctional finishing of natural fabrics with hibiscus flowers extract. Journal of Cleaner Production, 374 .
Ribeiro, J., & Veloso, C. (2021). Microencapsulation of natural dyes with biopolymers for application in food: A review. Food Hydrocolloids, 112 . https://doi.org/10.1016/j.foodhyd.2020.106374
Roy Maulik, S. (2015). Application and characterization of natural dye. Asian Dyer, 12 (3), 30–37.
Saati, E., Mulandari, R., Wachid, M., & Winarsih, S. (2018). The utilization of Telang flower as healthy-natural food coloring on dawet drink. In SolarPACES 2017: International conference on concentrating solar power and chemical energy systems . AIP Publishing. https://doi.org/10.1063/1.5064356
Sadeghi-Kiakhani, M., Tehrani-Bagha, A. R., Safapour, S., Eshaghloo-Galugahi, S., & Etezad, S. M. (2021). Ultrasound-assisted extraction of natural dyes from hawthorn fruits for dyeing polyamide fabric and study its fastness, antimicrobial, and antioxidant properties. Environment, Development and Sustainability, 23 , 9163–9180.
Samanta, A. K., & Konar, A. (2011). Dyeing of textiles with natural dyes. Natural Dyes (Ed. E. Perrin Akçakoca Kumbasar), 3 , 30–56.
Saranraj, P., & Sivasakthi, S. (2014). Spirulina platensis – Food for future: A review. Asian Journal of Pharmaceutical Science & Technology, 4 (1), 26–33.
Sawhney, N., & Satapathi, S. (2016). Utilization of naturally occurring dyes as sensitizers in dye sensitized solar cells. IEEE Journal of Photovoltaics, 7 (2), 539–544.
Sen, T., Barrow, C., & Desh, S. (2019). Microbial pigments in the food industry – Challenges and the way forward. Frontiers in Nutrition, 6 . https://doi.org/10.3389/fnut.2019.00007
Shakunthala, N. (2021). Natural dyes as a photosensitizer in dye sensitized solar cells – Review. Journal of Scientific Research, 65 (8). https://doi.org/10.37398/JSR.2021.650813
Shalini, S., Prasanna, S., Mallick, T. K., & Senthilarasu, S. (2015). Review on natural dye sensitized solar cells: Operation, materials, and methods. Renewable & Sustainable Energy Reviews, 51 , 1306–1325.
Shukla, D., & Vankar, P. S. (2017). Dyeing of cotton by different new natural dyeing sources. Natural Dyes for Textiles: Sources, Chemistry, and Applications , 111.
Singh, H., & Bharati, K. (2015). Handbook of natural dyes and pigments . Woodhead Publishing India.
Singh, M., Vajpayee, M., & Ledwani, L. (2021a). Eco-friendly surface modification of natural fibres to improve dye uptake using natural dyes and application of natural dyes in fabric finishing: A review. Materials Today: Proceedings, 43 , 2868–2871.
Singh, S., Maurya, I., Sharma, S., Kushwaha, S., Srivastava, P., & Bahadur, L. (2021b). Application of new natural dyes extracted from Nasturtium flowers ( Tropaeolum majus ) as photosensitizer in dye-sensitized solar cells. Optik, 243 . https://doi.org/10.1016/j.ijleo.2021.167331
Singh, D., Kumari, K., & Ahmed, S. (2022). Chapter 17 – Natural herbal products for cancer therapy. In B. Jain & S. Pandey (Eds.), Understanding cancer (pp. 257–268). Academic.
Sivakumar, V., Lakshmi Anna, J., Vijayeeswarri, J., & Swaminathan, G. (2009). Ultrasound-assisted enhancement in natural dye extraction from beetroot for industrial applications and natural dyeing of leather. Ultrasonics Sonochemistry, 16 (6), 782–789.
Slama, N., Ticha, M., Skhiri, W., Boudokhane, C., & Dhaouadi, H. (2022). Development and optimization of a sustainable process assisted by microwave energy to dye cellulosic fabrics by Juglans regia bark residues extract. Sustainability, 14 (13), 7534.
Solymosi, K., Latruffe, N., Morant-Manceau, A., & Schoefs, B. (2015). Food colour additives of natural origin (pp. 3–34). Woodhead Publishing.
Stevens, C. V. (2023). Handbook of natural colorants . Wiley.
Book Google Scholar
Strieder, M., LandimNeves, M., Silva, E., & Meireles, M. (2020). Low-frequency and high-power ultrasound-assisted production of natural blue colorant from the milk and unripe Genipa americana L. Ultrasonics Sonochemistry . https://doi.org/10.1016/j.ultsonch.2020.105068
Ticha, M., Slama, N., Dhouibi, N., Boudokhane, C., & Dhaouadi, H. (2022). Valorization of Juglans regia . L bark residues as a natural colorant based on response surface methodology: A challenging approach to a sustainable dyeing process for acrylic fabrics. Sustainability, 14 (7), 4134.
Trotman, E. (1994). Dyeing and chemical technology of textile fibres (1st ed.). B I Publications Pvt. Ltd.
Tupuna-Yerovi, D., Paese, K., Floˆres, S., Guterres, S., & Rios, A. (2020). Addition of norbixin microcapsules obtained by spray drying in an isotonic tangerine soft drink as a natural dye. Journal of Food Science and Technology, 57 (3), 1021–1031.
Vadood, M., & Haji, A. (2022a). A hybrid artificial intelligence model to predict the color coordinates of polyester fabric dyed with madder natural dye. Expert Systems with Applications, 193 . https://doi.org/10.1016/j.eswa.2022.116514
Vadood, M., & Haji, A. (2022b). Application of ANN weighted by optimization algorithms to predict the color coordinates of cellulosic fabric in dyeing with binary mix of natural dyes. Coatings, 12 . https://doi.org/10.3390/coatings12101519
Verma, S., & Gupta, G. (2017). Natural dyes and its applications: A brief review. International Journal of Research & Analytical Reviews, 4 (4), 57–60.
Vinha, A., Rodrigues, F., Nunes, M., & Oliveira, M. (2018). 11 – Natural pigments and colorants in foods and beverages. In C. M. Galanakis (Ed.), Polyphenols: Properties, recovery, and applications (pp. 393–427). Woodhead Publishing.
Winarti, S., & Firdaus, A. (2010). Stability of red color rosella extract for food and beverage colorant. Jurnal Teknologi Pertanian, 11 (2), 87–93.
Wirasuta, I., Triastuti, N., Deviyanthi, K., Sartika, D., & Utari, P. (2018). Formulation of the body scrub cream from purple sweet potato (Ipomoea batatas L.). Indonesian Journal of Pharmaceutical Science and Technology, 5 (1), 26–30.
Witek-Krowiak, A., Chojnacka, K., Podstawczyk, D., Dawiec, A., & Pokomeda, K. (2014). Application of response surface methodology and artificial neural network methods in modelling and optimization of biosorption process. Bioresource Technology, 160 , 150–160.
Wongcharee, K., Meeyoo, V., & Chavadej, S. (2007). Dye-sensitized solar cells using natural dyes extracted from rosella and blue pea flowers. Solar Energy Materials and Solar Cells, 91 (7), 566–571.
Wu, M., Chena, Y., Penga, P., Zuo, D., Wang, Q., Liu, L., et al. (2022). Optimization of dark dyeing poly(lactic acid) yarn with indigo dyes applied to denim fabric. The Journal of The Textile Institute . https://doi.org/10.1080/00405000.2022.2047320
Xia, G., Ji, X., Xu, Z., & Ji, X. (2022). Transparent cellulose-based bio-hybrid films with enhanced anti-ultraviolet, antioxidant and antibacterial performance. Carbohydrate Polymers, 298 . https://doi.org/10.1016/j.carbpol.2022.120118
Yadav, S., Tiwari, K. S., Gupta, C., Tiwari, M. K., Khan, A., & Sonkar, S. P. (2022). A brief review on natural dyes, pigments: Recent advances and future perspectives. Results in Chemistry, 5 , 100733.
Yu, D., Liu, Y., Ren, Y., Liu, X., & Qu, H. (2022). One-pot strategy to simultaneously prepare dyed, flame retardant and UV-resistant silk fabric based on a safflower yellow derivative. Royal Society of Chemistry, 12 , 26147–26159.
Yusuf, M., Shabbir, M., & Mohammad, F. (2017). Natural colorants: Historical, processing, and sustainable prospects. Natural Products & Bioprospecting, 7 , 123–145.
Zhai, X., Li, Z., Zhang, J., Shi, J., Zou, X., Huang, X., et al. (2018). Natural biomaterial-based edible and pH-sensitive films combined with electrochemical writing for intelligent food packaging. Journal of Agricultural & Food Chemistry, 66 (48), 12836–12846.
Zhang, Y., Zhou, Q., Xia, W., Rather, L., & Li, Q. (2022). Sonochemical mordanting as a green and effective approach in enhancing cotton bio-natural dye affinity through soy surface modification. Journal of Cleaner Production, 336 .
Zhou, H., Wu, L., Gao, Y., & Ma, T. (2011). Dye-sensitized solar cells using 20 natural dyes as sensitizers. Journal of Photochemistry and Photobiology A: Chemistry, 219 (2–3), 188–194.
Download references
Author information
Authors and affiliations.
Department of Silpa-Sadana, Visva-Bharati (A Central University), Sriniketan, India
Silpinwita Das & Sankar Roy Maulik
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Sankar Roy Maulik .
Editor information
Editors and affiliations.
Chief Sustainability Officer, Green Story Inc., Kowloon, Hong Kong
Subramanian Senthilkannan Muthu
Rights and permissions
Reprints and permissions
Copyright information
© 2023 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this chapter
Das, S., Roy Maulik, S. (2023). Recent Approaches and Advancements in Natural Dyes. In: Muthu, S.S. (eds) Natural Dyes and Sustainability. Sustainable Textiles: Production, Processing, Manufacturing & Chemistry. Springer, Cham. https://doi.org/10.1007/978-3-031-47471-2_4
Download citation
DOI : https://doi.org/10.1007/978-3-031-47471-2_4
Published : 24 January 2024
Publisher Name : Springer, Cham
Print ISBN : 978-3-031-47470-5
Online ISBN : 978-3-031-47471-2
eBook Packages : Earth and Environmental Science Earth and Environmental Science (R0)
Share this chapter
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Publish with us
Policies and ethics
- Find a journal
- Track your research
Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
- View all journals
- Explore content
- About the journal
- Publish with us
- Sign up for alerts
- Open access
- Published: 05 December 2022
Sustainable fashion: eco-friendly dyeing of wool fiber with novel mixtures of biodegradable natural dyes
- Lina Lin 1 , 2 na1 ,
- Tiancheng Jiang 1 , 2 na1 ,
- Lexin Xiao 1 , 2 ,
- Md. Nahid Pervez 3 ,
- Xiaobo Cai 4 ,
- Vincenzo Naddeo 3 &
- Yingjie Cai 1 , 2
Scientific Reports volume 12 , Article number: 21040 ( 2022 ) Cite this article
3523 Accesses
17 Citations
11 Altmetric
Metrics details
- Biomaterials
- Materials science
Natural materials, especially natural colorants, have achieved global prominence and might be regarded as an environmentally beneficial alternative to hazardous synthetic dyes. The color limitation of natural dyes hinders their application in textiles. The present work aims to prepare more color shades of wool yarns via dyeing with ternary natural dye mixtures without adding mordants. In this study, a sustainable dyeing approach for wool yarn was evaluated with three natural dyes, madder red (MR), gardenia blue (GB), and gardenia yellow (GY), by following an industrial dyeing procedure in the absence of a mordant. In the beginning, a preliminary assessment of dye stabilities was carried out, and it was found that the three natural dyes were sensitive to temperature and acid (degradation tendency). Then, the dyeing behavior was systematically evaluated, including a single natural dye, a binary natural dye mixture, and a ternary natural dye mixture. The results of wool yarn dyeing with a single natural dye show that the dye exhaustion percentage (E%) of MR, GY, and GB was in the ranges of 78.7–94.1%, 13.4–44.1%, and 54.8–68.5%, respectively. The dyeing results of wool yarns dyed with binary and ternary natural dye mixtures (a color triangle framework of dyed wool yarn) were characterized by colorimetric values (L*, a*, b*, C*, h, and K/S), and are presented to enlighten various colorful shades. Finally, color uniformity and colorfastness tests confirmed the vital contribution of natural dyes toward wool yarn coloration. Particularly, colorfastness to washing confirmed the stability of natural dyes with reference to the lower amount of dyes released into the effluent, which is beneficial for the environment. Overall, this study provides a good background for enhancing the industrialization trend of natural dyes by modulating their dyeing scheme.
Similar content being viewed by others

Dyeing of soybean protein/flax blended yarns with reactive dyes and subsequent dye-fixation
Sustainable traditional grass cloth fiber dyeing using the Taguchi L16 (4^4) orthogonal design

Green extraction and application of yellow natural curcumin colorant from Curcuma aromatica rhizomes for silk dyeing
Introduction.
Synthetic dyes are commonly used for the coloration of textiles owing to their huge hues and easy application. However, the synthetic dyes and auxiliaries present in dyeing wastewater discharged into the environment cause toxic and allergic reactions 1 , 2 , 3 , 4 , 5 , 6 , 7 to creatures, resulting in adverse effects on the ecosystem. With an increase in the concern on environmental issues, the use of natural dyes for textile coloration has recently increased owing to their biodegradability and compatibility with the environment 8 , 9 , 10 . The natural dyes not only color but also provide beneficial properties to the textiles 11 , 12 , such as antibacterial activity, antioxidant activity, and UV resistance. However, the disadvantages of low dye exhaustion percentage and fixation efficiency, especially weak light fastness, hinder its wide application 13 , 14 , 15 .
To overcome the inherent drawbacks of natural dyeing on a larger scale, mordants (metallic salts) 16 , 17 such as aluminum potassium sulfate (KAl(SO 4 ) 2 ), stannous chloride (SnCl 2 ), potassium dichromate (K 2 Cr 2 O 7 ), and sodium chromate (Na 2 CrO 4 ) are extensively added in dyeing by pre, simultaneous, or post-mordanting pattern 18 , 19 to form complexes between the natural dye and fiber. After complexation, the natural dye is stably precipitated in fiber 20 , promoting dye exhaustion, fixation, and colorfastness to washing and rubbing 21 , 22 , 23 . However, the residual heavy metal ions of mordants in the discharge are harmful to the environment 24 , 25 . Besides, the color hue of natural dye is shifted after complexing with mordants, and different mordants result in different color changes, causing problems in controlling the color stability in batch production 26 , 27 .
Natural dyes are encouraged to be used in textile dyeing 20 owing to their benefits to the ecosystem. However, the source of natural colorants limits their industrial application because the harvest time is dependent on the season 28 , 29 . Although the color range of natural dyes covers red, olive, burgundy, green, yellow, brown, blue, and black from the plant origin of bark, roots, leaves, fruits, and flowers 30 , 31 , it is still not comparative with that of synthetic dyes. In other words, the color hue of natural dye also challenges a natural dyeing product. In dyeing textiles with a synthetic dye, the color shade of a substance is generally prepared with ternary dye mixtures; hence, huge color shades of textiles are achieved. To overcome the color limitation of natural dyes, dyeing textile with ternary natural dye mixtures is a practical way to prepare more color shades of textiles. However, notably the addition of mordants in dyeing cause a color shift, and different natural dyes may need different mordants 32 . Thus, it is necessary to eliminate the addition of mordants in natural dyeing.
This study presents a new insight into the sustainable dyeing of wool yarn with mixtures of madder red (MR), gardenia blue (GB), and gardenia yellow (GY) dyes to prepare different color shades (color triangle) with various dye mass ratios without the addition of mordants. Dyeing with a binary and ternary mixture of natural dyes is still limited in natural dyeing plants 33 . In this study, not only colorful wool fibers were successfully produced using natural dye mixtures with satisfactory wash colorfastness, but also the research guidance on the coloration of fiber with natural dye mixtures is provided.
Experimental
Scored wool yarn (48 Nm/2, 100%) was a gift from a local dyeing plant. The MR dye was obtained from Zhongda Hengyuan Biotechnology Stock Co., Ltd. (China). The GB and GY dyes were obtained from Wuhan Green Food Biological Engineering Co. Ltd. (China). The chemical structures of the main components of MR 34 , GB 35 , 36 , and GY 37 are shown in Fig. 1 . Sodium acetate trihydrate (AR) and acetic acid (AR) were purchased from Sinopharm Chemical Reagent Co., Ltd (China). Nonionic detergent (Luton 500) was purchased from Dalton UK Company (Shanghai, China).
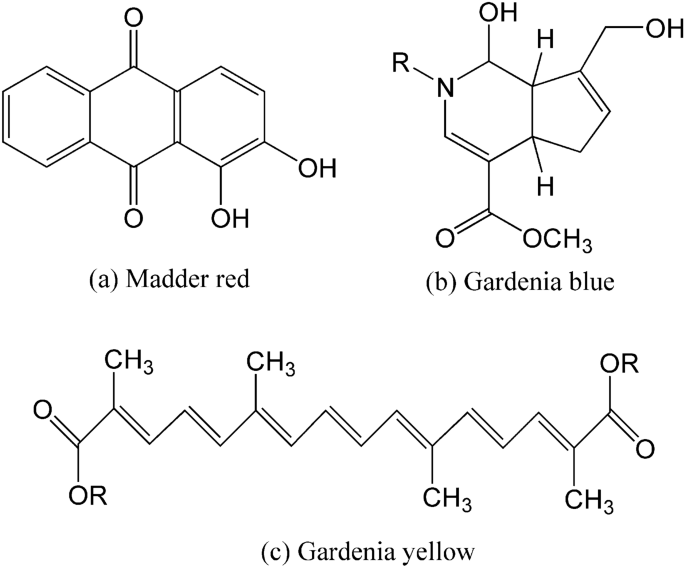
Chemical structures of main components of ( a ) madder red, ( b ) gardenia blue, and ( c ) gardenia yellow dyes.
Stability of natural dyes
To study the thermostability of these three natural dyes, various acidic buffer solutions with pH 3–6 were prepared using sodium acetate trihydrate and acetic acid in deionized water. The dye was added to the acidic buffer solution to prepare a dye solution with a concentration of 80 mg L −1 at pH 3–6. Subsequently, the dye solution was heated at a rate of 1 °C min −1 from 30 °C to a target temperature (60–100 °C), and then maintained at this level for a period (110–150 min) to make 180 min of total treatment time. The treatment process was completed in a rotary infrared radiation laboratory-dyeing machine (Automatic Prototype, Model: A-12, AQUA, China). The dye solution at various treatment periods was measured using a UV–Vis spectrophotometer (Cary 100, Agilent Technologies, Australia) 38 . The degradation percentage of dye (D%) was calculated using Eq. ( 1 ), where A 0 and A 1 are the light absorbance of the dye solution at the maximum absorbance wavelength (λ max ) before and after treatment.
Dyeing of wool yarn
In dyeing with a single natural dye, the wool yarn (2 g) was dyed with various 2% o.m.f (on the mass of fiber) of natural dye in an acidic buffer solution at pH 3–6 with a liquor ratio of 25:1 at 60–100 °C for 180 min in a rotary infrared radiation laboratory-dyeing machine (Automatic Prototype, Model: A-12, AQUA, China) 39 . The dyeing processes are shown in Fig. 2 . In dyeing with natural dye mixtures, the wool yarn (2 g) was dyed with 3% o.m.f of dye mixtures (varying weight ratios are shown in Fig. 3 ) at pH 3 and 90 °C with a liquor ratio of 25:1 and maintained at 90 °C for 40 min. After dyeing, the dyed sample was squeezed by hand to remove the excess dye solution in the wool yarns, followed by drying in an oven. Subsequently, the dried sample was treated in a soap solution containing 2 g L −1 of nonionic detergent at 95 °C for 15 min at a liquor ratio of 50:1. Consequently, the soaped dyed wool yarns were dried in an oven at 60 °C.

Dyeing of wool yarn with natural dye.
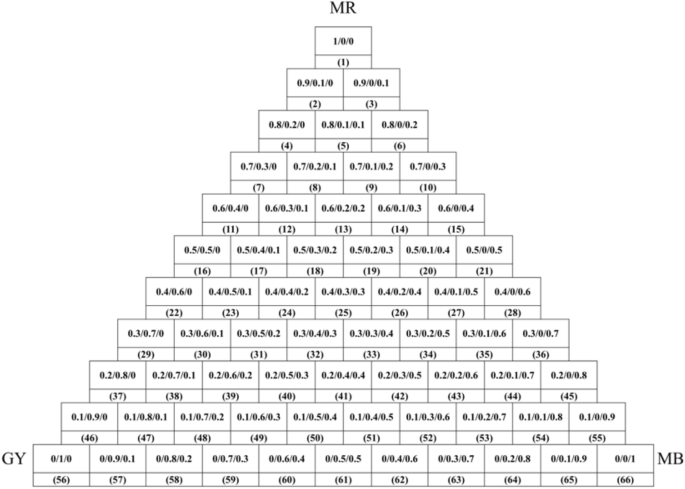
Color triangle of wool yarn dyeing using MR, GY, and GB dye mixtures with various dye mass ratios.
Characterization
The chromatic values (L*, a*, b*, C*, h, and K/S) of dyed wool yarns were detected using a spectrophotometer (CHN-Spec CS-650A, Hangzhou Color Spectrum Technology Company, China) in the range of 350–760 nm of wavelength with 10 nm interval at 20 random positions. The mean of 20 detections was used to determine the chromatic values of each sample. The color difference (∆E) of dyed samples before and after treatment was calculated using Eq. ( 2 ), where subscripts a and b refer to those after and before treatment, respectively. The color uniformity of dyed wool yarn was represented by the standard deviation (σ) of the K/S values 40 . The wash colorfastness was evaluated using the ISO 105-C06:1997 (Test number: C2S). The light fastness of dyed wool yarn was tested using a Xenon Test Chamber (Q-SUN Xe-1, Q-LAB, USA) with irradiation for 24 h 41 . The color difference (∆E, Eq. 2 ) of the dyed sample before and after irradiation was used to assess the light fastness 42 .
Results and discussion
The light absorbances of MR, GB, and GY dyes from 350 to 760 nm are shown in Fig. 4 . The maximum absorbance wavelengths of MR dye (0.048 g L −1 ), GB dye (0.18 g L −1 ), and GY dye (0.1 g L −1 ) are 519 nm, 594 nm, and 441 nm, respectively. This indicates that at the same concentration of the used dyestuffs, the tinctorial strength of MR is the highest, followed by GY, and GB has the lowest tinctorial strength.
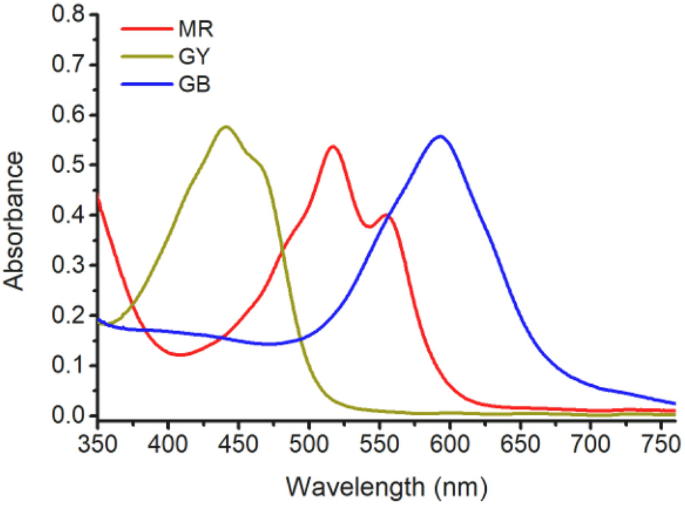
Light absorbance of MR, GB, and GY dye solutions.
The stability of these three natural dyes was evaluated at various temperatures (60–100 °C), pH of dye solution (pH 3–6), and treatment time; their D% values are shown in Figs. 5 and 6 . In the temperature variation study, the three natural dyes showed that the value of D% increased with an increase in temperature for these three dyes. This dye degradation tendency was also claimed in the case of cotton fabric dyeing with watermelon rind saps 43 . For 180 min treatment of GY dye, the D% values were 16.2, 19.1, 24.9, 32.6, and 46.8% for treating at 60, 70, 80, 90, and 100 °C, respectively. The other two dyes also exhibited a similar increasing tendency; the D% values of MR and GB increased from 15.1 to 28.0% and from 6.9 to 27.1% with increasing temperature from 60 to 100 °C for 180 min treatment, respectively. Besides, the D% values increased with an increase in treatment time for each treatment temperature 44 . These three natural dyes are sensitive to temperature, especially GY dye.
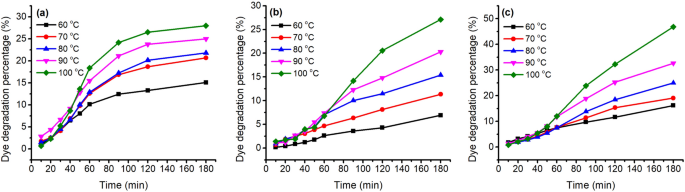
Thermostability of 80 mg L −1 of ( a ) MR, ( b ) GB, and ( c ) GY dyes in deionized water at pH 4 and 60–100 °C.
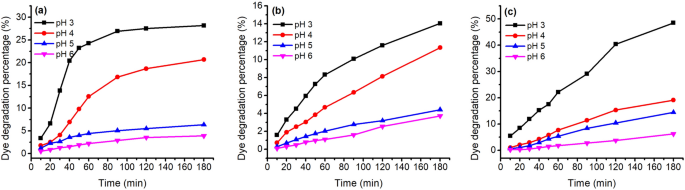
Thermostability of 80 mg L −1 of ( a ) MR, ( b ) GB, and ( c ) GY dyes in deionized water at pH 3–6 and 70 °C.
In the study of pH condition variation, the stability of these natural dyes decreased with a decrease in pH from pH 6 to pH 3, which is consistent with the previous study 45 . With the decrease in pH from pH 6 to pH 3, the D% values increased from 6.2 to 48.5% for GY dye (Fig. 6 c), from 3.9 to 28.2% for MR dye (Fig. 6 a), and from 3.7 to 14.0% for GB dye (Fig. 6 b). Hence, the order of stability in pH sensitivity was GY > MR > GB. In addition, the D% almost linearly increased with the increase in treatment time for GB and GY dyes, while for MR dye, the increase in D% gradually reduced when the temperature reached the target temperature.
Dyeing of wool yarn with a single natural dye
The results of the dye stability study indicate that during the dyeing, the natural dyes were partially degraded; hence, it is incorrect to use the light absorbance of the dye solution before and after dyeing to detect the dye amount absorbed in the wool yarn. Therefore, the description of the K/S values of dyed wool yarn is an alternative method to describe the real dye adsorption behavior in dyeing, and the higher K/S stands for, the higher dye adsorbed in wool yarn 46 .
The K/S values of dyed samples before soaping are shown in Figs. 7 and 9 with a variation in dyeing temperature (60–100 °C) and pH of the dye bath (pH 3–6), respectively. In the variation in dyeing temperature (Fig. 7 ), the K/S values increased with increasing dyeing temperature from 60 to 100 °C, except for the GY dyeing because its highest K/S was at 90 °C. The results can be attributed to the better swelling of fiber and dye migration in fiber at higher dyeing temperatures 47 . Thus, a higher dyeing temperature is beneficial for dye absorption in wool fiber.
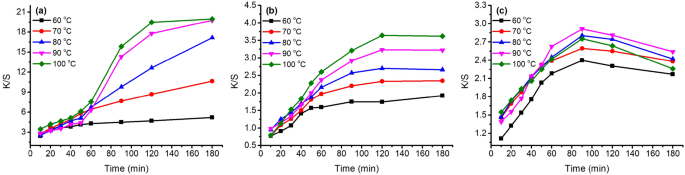
Color strength (K/S) of dyed wool fiber with 2% o.m.f of ( a ) MR, ( b ) GB, and ( c ) GY dyes in water at pH 4 and 60–100 °C.
Besides, after reaching the target temperatures, the dyes were still adsorbed in the wool yarns, except for the GY dye. In the dyeing with GY dye, after reaching the target temperatures, the dye was still adsorbed in the wool yarn first, but the GY dye mass decreased after 90 min of dyeing time in all dyeing temperatures, especially for the dyeing at 100 °C, which reduced to 2.3 at 180 min from 2.8 at 90 min dyeing time. The dramatic dye degradation can be attributed to the decrease in the K/S value. The dye degraded in the dye bath broke the previous dye adsorption balance, resulting in the dye adsorbed in wool fiber being desorbed and transferred to the dye bath. Therefore, the optimum dyeing temperature and dyeing time for GY were 90 °C and 90 min, respectively.
In the dyeing with MR dye, the dyeing equilibrium was 120 min after dyeing at 100 °C with a K/S of 19.5, and the K/S was 17.8 at 90 °C. In the dyeing with GB dye, the dye adsorptions were close to equilibrium for 120 min after dyeing. Hence, the optimum dyeing temperature and dyeing time for both MR and GB dyes were 100 °C and 120 min, respectively.
In the variation of dye bath pH study, Fig. 9 shows that a decrease in dye bath pH was effective in promoting dye adsorption, i.e., K/S value for all these three dyes, which was proved by the previous study 48 . Wool fiber contained free carboxylic acid (–COOH) and amino (–NH 2 ) groups; both groups exist in the zwitterion form when the wool is in water 49 . Thus, the lower the pH of the medium in which wool was present, the more the generated terminal amino groups 49 . Herein, the natural dyes were used as acidic dyes, forming ionic bonds with wool fiber; to be exact, the ionic bond between the anionic group of natural dye and the cationic terminal amino group of wool (Fig. 8 ). The cationic sites of terminal amino groups decreased with the increase in the pH of the dyeing medium 50 , and decreased the dye adsorption during dyeing, i.e., lowering the K/S value of dyed wool yarn.
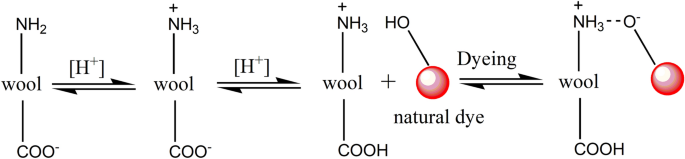
Mechanism of wool dyeing with natural dye in acidic conditions.
In the dyeing with MR dye, the K/S values increased with increasing dyeing time under various pH conditions. Whereas, in the dyeing with GB dye, the K/S values increased for 60 min and then slightly increased in the last period of dyeing for pH 3 and 4 of the dye bath. In the dyeing with GY dye, the maximum K/S values for pH 3 to pH 5 were at 90 min after dyeing, with a slight decrease in the last period of dyeing, which can be ascribed to intensive degradation. Figure 9 shows that the highest dye adsorption present in the dyeing at pH 3 hinted that the partial dye degradation during dyeing might not influence the tendency of dyeing performance.
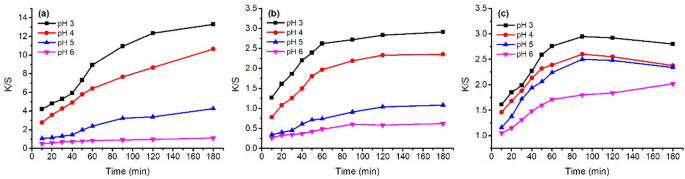
Color strength (K/S) of dyed wool fiber with 2% o.m.f of ( a ) MR, ( b ) GB, and ( c ) GY dyes in water at pH 3–6 at 70 °C.
Dyeing of wool yarn with natural dye mixtures
The dyed wool yarns with binary and ternary natural dye mixtures before and after soaping are shown in Figs. 10 and 11 , respectively. The chromatic values, K/S values, and dye fixation rates of wool yarns dyed with binary dye mixtures before and after soaping are shown in Tables 1 , 2 , and 3 , and those with ternary dye mixtures are shown in Table 4 .
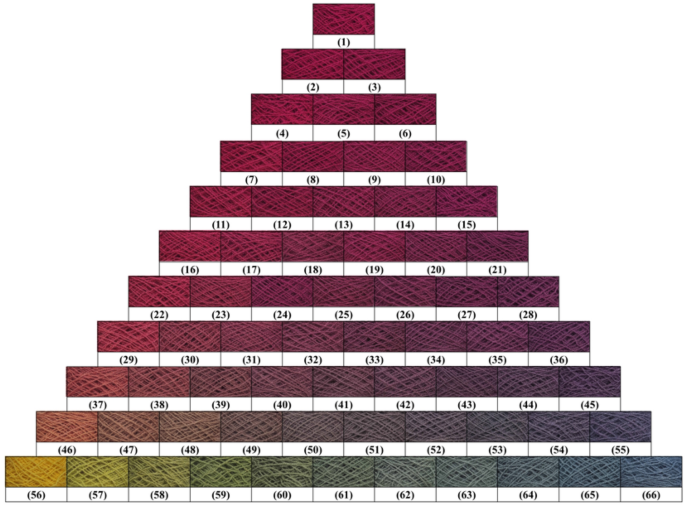
Dyed wool yarn with ternary mixtures of MR, GY, and GB before soaping.
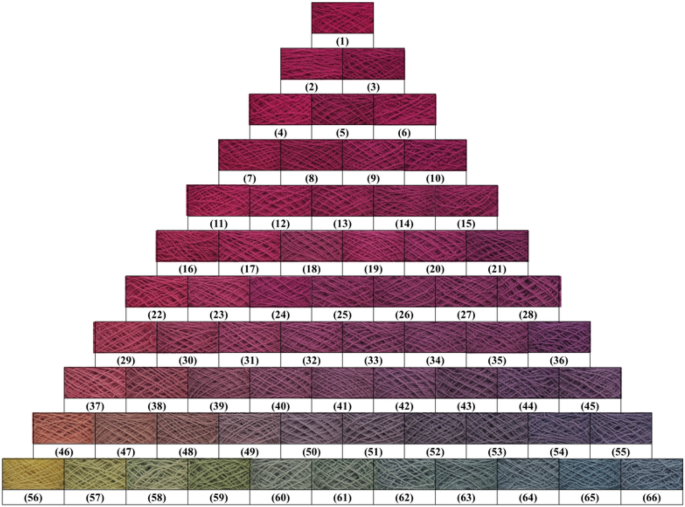
Dyed wool yarn with ternary mixtures of MR, GY, and GB after soaping.
In the dyeing with binary mixtures, the color shades of dyed samples before and after soaping regularly changed due to the inerratic change in dye mass ratio. As shown in Table 1 , with a decrease in MR and an increase in GY mass ratios in mixtures among the dyed samples before soaping, a decreased tendency of a* values of CIE lab color space and increased tendencies of L* and b* values of CIE lab color space is presented, because MR (red color) mainly contributes to the positive a* value, while GY (yellow color) mainly affects the positive b* value, according to the color coordinate.
In Table 1 , the dye mass ratio was 0.2:0.8 (MR:GY) for Sample 37. However, the λ max of K/S was 530 nm, and h was 33.5°, indicating that the main color hue of the sample is reddish. The photograph of Sample 37 in Fig. 10 also identified the color hue. It was possibly caused by the dye mass adsorbed in the wool fiber and the tinctorial strength of dyes. The results of the thermostability study of these three natural dyes clearly show that these three dyes degraded at 90 °C (Fig. 5 ) and pH 3 (Fig. 6 ). Thus, the natural dyes were adsorbed in wool yarn during dyeing and suffered from degradation simultaneously. Notably, the dye concentration during the dyeing was reduced more dramatically than in the dye degradation treatment because of the dye adsorption in wool fiber, i.e., the dye degradation percentage in dyeing was less than in degradation.
Figure 12 shows the results of dyeing with MR (Sample 1), GY (Sample 56), and GB (Sample 66) and their respective control dyeing (without the addition of wool yarn). The dye degradation percentage (D%), residual percentage (R%), maximum dye exhaustion percentage (E max %), and minimum dye exhaustion percentage (E min %) were calculated using Eqs. ( 1 ), ( 3 )–( 5 ), respectively.
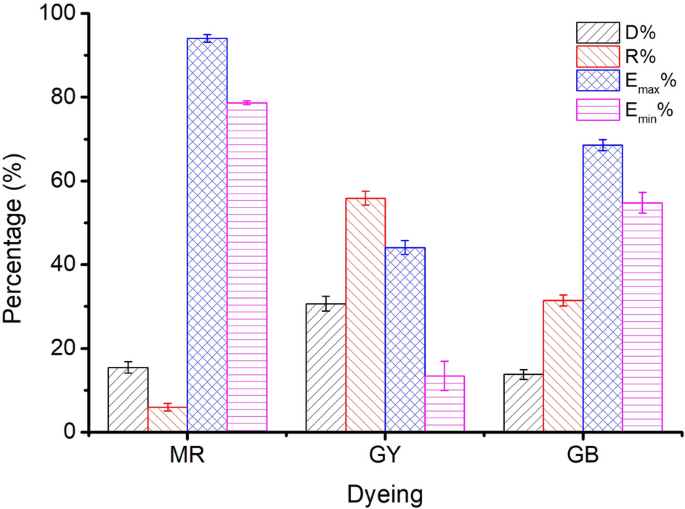
Dye residual percentage (R%), degraded percentage (D%), and the maximum and minimum dye exhaustion percentages (Emax% and Emin%) in dyeing with MR (Sample 1), GY (Sample 56), and GB (Sample 66).
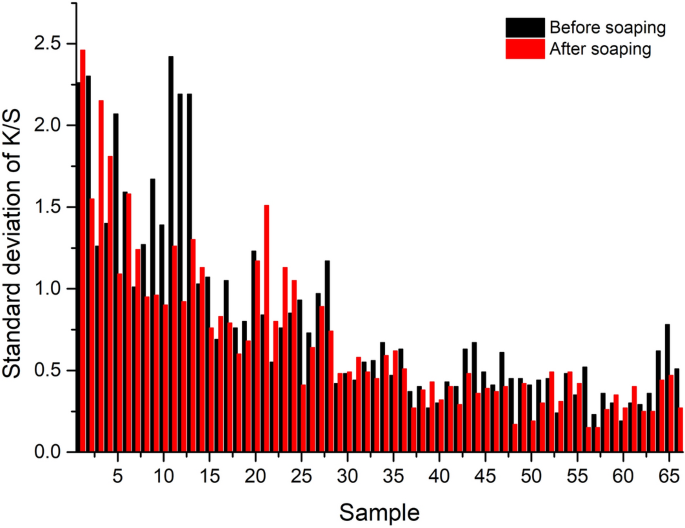
Standard deviation of K/S values of dyed wool yarns.
where A 2 is the light absorbance of the residual dye bath at λ max .
When the natural dyes did not degrade during dyeing, i.e., the D% was null, the E% was maximum (E max %); or during the dyeing of wool yarn, the tendency of dye degradation was similar to that in the control dyeing, i.e., the D% was maximum, so the E% was minimum (E min %). However, actually, partial natural dyes degraded during the wool fiber dyeing, and the dye adsorption in wool fiber reduced the amount of dye degradation in comparison to control dyeing. Thus, the E% should be within a range of E min % to E max %.
Figure 12 shows that the E% values of MR, GY, and GB were in the ranges of 78.7–94.1%, 13.4–44.1%, and 54.8–68.5%, respectively, and without overlapping. In other words, in the dyeing with a single dye, MR dyeing had the highest E%, GB dyeing was the next, and GY dyeing had the lowest E%, because of the highest R% and D% for GY. Thus the higher E% of MR than GY contributed to the reddish hue for Sample 37. Moreover, the GY has a lighter tinctorial strength than MR; hence, with the increased ratio of GY, the L* values increased. Besides, the maximum wavelength of the dyed samples’ K/S changed to 450 nm from 530 nm while the dye mass ratio was 0.1:0.9 (Sample 46), indicating that GY primarily contributed to the color shade.
In the description of color shade using the CIE Lch color space, the C* values (color saturation) generally decreased with the decrease in MR dye ratios for the dyed samples with or without soaping, indicating that MR primarily reduced the C* in the dyeing with binary mixtures of MR and GY dyes. C* value is dependent on the a* and b* values, and it is determined using Eq. ( 6 ). Because the higher E% and stronger tinctorial strength of MR than GY, the decrease of MR component in the binary mixture of MR and GY decreased the C* values. The h value was used to assess the hue of the sample. The maximum wavelength of K/S shifted to 450 nm from 530 nm, whereas the dye mass ratio was 0.1:09 (MR:GY), accompanied by an h value of 55.0° for the dyed sample before soaping, indicating that the dyed samples had more yellowness.
The color of the dyed sample changed by the soaping process, and the color difference (∆E) was within the range of 3.4–8.4 for the dyeing of wool yarns with binary mixtures of MR and GY dyes. The ∆E for the single MR dye was 4.2, while the ∆E for the single GY jumped to 18.5. Because the dyeing was executed at pH 3, an excessive GY was possibly adsorbed in wool yarn. Excessive adsorption usually occurs in the dyeing of wool with an acidic dye under strongly acidic conditions. Thus, the unfixed natural dyes in wool fiber were washed off after soaping, accompanied by color change. Owing to a temperature of 95 °C during soaping, it was impossible to detect the dye fixation rate by the light absorbance of the soaping solution because these natural dyes are easily degraded at high temperatures (Figs. 5 and 6 ). Besides, in the dyeing with binary mixtures of dyes, the dye amount removal ratio of each dye from the dyed wool yarn was occasionally different, for example, in Sample 55 in Table 2 , shifting the λ max of K/S after soaping. Furthermore, these natural plant dyes contain many compounds 51 , 52 , 53 , 54 , which are also adsorbed in the substance and contribute to the color shade of the dyed sample in its application. Hence, after soaping the wool yarn dyed with a single dye, the color shift was caused by the change in the amount of all the compounds in the wool yarn, although the λ max of K/S was the same. Therefore, the dye fixation rate cannot be calculated from the ratio of the K/S value at the λ max before soaping to that after soaping.
In the dyeing with binary MR and GB mixtures, the chromatic values of dyed wool yarns obtained before and after soaping are shown in Table 2 . The chromatic values of L*a*b* show a similar change tendency in comparison with the dying using binary MR and GY mixtures. The blue color of GB contributed a negative b* value. Thus, with an increase in GB ratio in the mixture of MR and GB, the b* value of the dyed samples changed to a minus value, and its absolute value became larger, indicating that the GB dye was adsorbed more in the wool yarns. The λ max of K/S was 530 nm for Sample 28 with a ratio of 0.4:0.6 (MR:GB). Then, the λ max of K/S shifted to 570 nm at a ratio of 0.3:0.7 (Sample 36) and 600 nm at a ratio of 0.1:0.9 (Sample 55) due to the higher E% of GB. After soaping, the unfixed dye wash-off weakened the color strength and increased the lightness because the L* values of dyed samples were promoted.
The chromatic values of dyed wool yarns with binary mixtures of GY and GB are shown in Table 3 . GY mainly contributed to positive b* value, while GB mainly contributed to negative b*. Hence, with the increase in GY ratio, the b* decreased and shifted to a minus value, and the L* values decreased. However, the a* values slightly fluctuated within the range of − 7.30 to − 11.17. Both of these decreases indicate that the color of the dye sample became darker with the increase in GB content. Notably, the a* value (5.25) of Sample 56 immediately changed to minus (− 7.30) at a ratio of 0.9:0.1, indicating that GB had a green tint and a vital effect on the a* value. Thus, Sample 56 showed slight greenish color (Fig. 10 ). The λ max of K/S was 450 nm for Sample 61 with a ratio of 0.5:0.5 (MR:GB), and it changed to 610 nm for Sample 62 with a ratio of 0.4:0.6. It was caused by the higher amount of GB in the dyed wool yarn because of its high E% (Fig. 12 ) and high ratio in the binary mixtures, although the tinctorial strength of GY is higher than GB (Fig. 4 ). The main hue of the dyed sample did not shift by soaping, except for Sample 61 The λ max of K/S changed to 610 nm from 450 nm because more amount of GY was washed off by the soaping, in contrast with GB. The ∆E values of Samples 56 and 66 were 18.5 and 5.8, respectively, indicating that the fixation property of GY was poorer than GB. This result supports the explanation of the λ max of K/S shift in Sample 61 after soaping.
The chromatic values of dyed wool yarn with ternary mixtures of MR, GY, and GB are shown in Table 4 . The different chromatic values of dyed samples after soaping indicate that wool yarns with many color shades were prepared with different dye mass ratios. The color shades are shown in Fig. 11 . Besides, the ∆E values had a small change, indicating that the dye fixation properties of these dyes were acceptable.
Color uniformity
The color uniformity of dyed wool yarns expressed by the standard deviation of K/S values is shown in Fig. 13 . Generally, most of the standard deviation values are lower than 1.0 for the dyed samples (Figs. 10 and 11 ) before and after soaping, indicating that the color on the surface of dyed samples was even. Meanwhile, the soaping process was beneficial for the color uniformity of dyed samples, because the standard deviation of dyed samples was reduced after soaping. Therefore, it can be concluded that in the dyeing with binary and ternary dyes mixtures, the natural dyes were equally distributed in the wool yarns and exhibited uniform color.
Colorfastness to washing and lighting
The samples dyed with a single dye, binary dye mixture, and ternary dye mixture were selected to assess their wash colorfastness, as shown in Fig. 14 . The wash colorfastness of these samples in staining the multifiber fabric was Grade 5 for all six fibers. Although the light color was present in the solution during testing, the multifiber fabric was clean without any staining, possibly because the testing conditions (pH 10.5) prevented the removal of dye from the multifiber. Furthermore, after testing, the fade color grade of wash colorfastness was found to be Grade 5 for all the tested samples. Therefore, the results of these selected samples indicate that the wash colorfastness of the dyed samples with a single dye and mixtures was Grade 5 for both the staining and fade colorfastness. Overall it shows that less amount of color is released during washing, which is very crucial for maintaining environmental sustainability.

Wash colorfastness assessment of dyed wool yarns using singe dye, binary dye mixtures, and ternary dye mixtures.
The photographs of selected dyed wool yarns before and after light colorfastness testing are shown in Fig. 15 , along with their K/S curves, and the color difference of dyed wool yarns before and after the light colorfastness testing is shown in Table 5 . Undoubtedly, the colors of dyed wool yarns faded apparently due to the natural dye's inherent defect of unstable color by light irradiation 55 . After light irradiation, the K/S of dyed samples decreased, especially for GY and GB dyes. The color of the dyed sample with single GY (Sample 56) or GB (Sample 66) almost tended to be a grey color. In contrast, MR exhibited a relatively better light resistance 56 , although it also slightly faded after light irradiation. In the dyed wool yarn with binary and ternary mixtures, the MR component showed its vital contribution to light resistance. The color difference of dyed wool samples in Table 5 shows a significant trend of MR for light resistance because the ∆E of the dyed sample with MR was lower than that without the MR component (Samples 56, 61, and 66). Meanwhile, because the color faded, the L* values of all the samples increased.
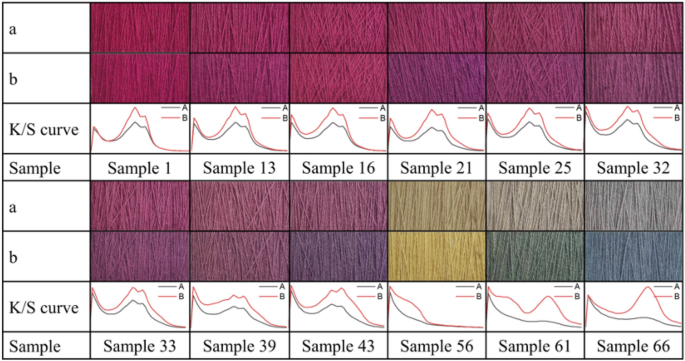
Photographs of dyed wool yarns ( a ) after and ( b ) before light colorfastness testing and their K/S curves.
Conclusions
The sustainable dyeing of wool yarn in the presence of three representative natural dyes, MR, GB, and GY, was carried out without the addition of conventional mordants. Initially, the light absorbance data show that the tinctorial strength of MR is the highest, followed by GY and GB, which is also supported by their thermostability. In a single bath of natural dyeing, K/S values were measured to determine the real dye adsorption behavior, and it found the K/S values increased with increasing dyeing temperature from 60 °C to 100 °C, except for the GY dyeing because its highest K/S was at 90 °C. Then, a color triangle dyeing recipe (binary and ternary dyeing scheme) was formulated using around 66 samples; no overlapping tendency was obtained with the E% values of MR (78.7–94.1%), GY (13.4–44.1%), and GB (54.8–68.5%). Additionally, a color uniformity test confirmed that in the dyeing with binary and ternary dyes mixtures, the natural dyes were equally distributed in the wool yarns (uniform color), and the colorfastness properties were satisfactory in terms of less color reduction during the washing process especially. As a result, natural dye mixtures may be deemed as a suitable approach for coloring textile materials, with no problematic processes or negative environmental repercussions. Hence, in future research, attention will be paid to mixing natural colors to find natural dyes with green color and investigate their dyeing performance on wool fiber in mixtures, thereby expanding the range of colors for dyed wool yarn with a mixture of natural dyes.
Data availability
The datasets generated during the current study are available from the corresponding author on reasonable request (Prof. Yingjie Cai, Y. Cai).
Pervez, M. N. et al. A critical review on nanomaterials membrane bioreactor (NMs-MBR) for wastewater treatment. NPJ Clean. Water 3 , 43. https://doi.org/10.1038/s41545-020-00090-2 (2020).
Article CAS Google Scholar
Pervez, M. N., Stylios, G. K., Liang, Y., Ouyang, F. & Cai, Y. Low-temperature synthesis of novel polyvinylalcohol (PVA) nanofibrous membranes for catalytic dye degradation. J. Clean. Prod. 262 , 121301. https://doi.org/10.1016/j.jclepro.2020.121301 (2020).
Pervez, M. N. et al. Efficient degradation of Mordant Blue 9 using the Fenton-activated persulfate system. Water 11 , 2532. https://doi.org/10.3390/w11122532 (2019).
Hasan, K. M. F. et al. A novel coloration of polyester fabric through green silver nanoparticles (G-AgNPs@PET). Nanomaterials 9 , 569. https://doi.org/10.3390/nano9040569 (2019).
Morshed, M. N. et al. Statistical modeling and optimization of heterogeneous Fenton-like removal of organic pollutant using fibrous catalysts: A full factorial design. Sci. Rep.-UK 10 , 16133. https://doi.org/10.1038/s41598-020-72401-z (2020).
Article CAS ADS Google Scholar
Pervez, M. N., He, W., Zarra, T., Naddeo, V. & Zhao, Y. New sustainable approach for the production of Fe 3 O 4 /Graphene Oxide-activated persulfate system for dye removal in real wastewater. Water 12 , 733. https://doi.org/10.3390/w12030733 (2020).
Su, S. et al. Anhydrous dyeing processes of ramie fiber in liquid ammonia. Cellulose 26 , 8109–8120. https://doi.org/10.1007/s10570-019-02630-7 (2019).
Hossain, M. Y. et al. Effluent-free deep dyeing of cotton fabric with cacao husk extracts using the Taguchi optimization method. Cellulose 28 , 517–532. https://doi.org/10.1007/s10570-020-03525-8 (2021).
Habib, M. A., Pervez, M. N., Mahmud, S., Khan, M. R. M. & Heng, Q. Macadamia integrifolia: A new source of natural dyes for textile colouration. Asian J. Chem. 29 , 1543–1548. https://doi.org/10.14233/ajchem.2017.20560 (2017).
Hossain, M. Y. et al. Adsorption, kinetics, and thermodynamic studies of cacao husk extracts in waterless sustainable dyeing of cotton fabric. Cellulose 28 , 2521–2536. https://doi.org/10.1007/s10570-020-03662-0 (2021).
Shabbir, M., Rather, L. J. & Mohammad, F. Economically viable UV-protective and antioxidant finishing of wool fabric dyed with Tagetes erecta flower extract: Valorization of marigold. Ind. Crops Prod. 119 , 277–282. https://doi.org/10.1016/j.indcrop.2018.04.016 (2018).
Shafiq, F. et al. Extraction of natural dye from aerial parts of argy wormwood based on optimized taguchi approach and functional finishing of cotton fabric. Materials. 14 , 5850. https://doi.org/10.3390/ma14195850 (2021).
Xu, X. et al. Mordant free dyeing and functionalization of wool fabrics with biocolorants derived from Apocynum venetum L. Bast. ACS. Sustain. Chem. Eng. 8 , 12686–12695. https://doi.org/10.1021/acssuschemeng.0c04757 (2020).
Shahid, M., Shahidul, I. & Mohammad, F. Recent advancements in natural dye applications: A review. J. Clean. Prod. 53 , 310–331. https://doi.org/10.1016/j.jclepro.2013.03.031 (2013).
Hossain, M. Y. et al. Green and sustainable method to improve fixation of a natural functional dye onto cotton fabric using cationic dye-fixing agent/D5 microemulsion. J. Nat. Fibers 19 , 1–16. https://doi.org/10.1080/15440478.2021.2024933 (2022).
Shukla, D. & Vankar, P. S. Natural dyeing with black carrot: New source for newer shades on silk. J. Nat. Fibers 10 , 207–218. https://doi.org/10.1080/15440478.2012.757031 (2013).
Yi, E. & Cho, J. Y. Color analysis of natural colorant-dyed fabrics. Color. Res. Appl. 33 , 148–157. https://doi.org/10.1002/col.20390 (2008).
Article Google Scholar
Feiz, M. & Norouzi, H. Dyeing studies of wool fibers with madder ( Rubia tinctorum ) and effect of different mordants and mordanting procedures on color characteristics of dyed samples. Fiber. Polym. 15 , 2504–2514. https://doi.org/10.1007/s12221-014-2504-x (2014).
Telegin, F. Y. et al. Structure and properties of dyes in coloration of textiles: Application of fragment approach. Key. Eng. Mat. 703 , 261–266. https://doi.org/10.4028/www.scientific.net/KEM.703.261 (2016).
Jahangiri, A. et al. Natural dyeing of wool by Madder ( Rubia tinctorum L.) root extract using tannin-based biomordants: Colorimetric, fastness and tensile assay. Fiber. Polym. 19 , 2139–2148. https://doi.org/10.1007/s12221-018-8069-3 (2018).
Burkinshaw, S. M. & Kumar, N. The mordant dyeing of wool using tannic acid and FeSO 4 , Part 1: Initial findings. Dyes Pigments 80 , 53–60. https://doi.org/10.1016/j.dyepig.2008.05.008 (2009).
Zheng, G. H., Fu, H. B. & Liu, G. P. Application of rare earth as mordant for the dyeing of ramie fabrics with natural dyes. Korean J. Chem. Eng. 28 , 2148–2155. https://doi.org/10.1007/s11814-011-0090-9 (2011).
Cai, Y. et al. Improved reactive dye fixation on ramie fiber in liquid ammonia and optimization of fixation parameters using the Taguchi approach. Dyes Pigments 183 , 108734. https://doi.org/10.1016/j.dyepig.2020.108734 (2020).
Bechtold, T., Turcanu, A., Ganglberger, E. & Geissler, S. Natural dyes in modern textile dyehouses—how to combine experiences of two centuries to meet the demands of the future?. J. Clean. Prod. 11 , 499–509. https://doi.org/10.1016/S0959-6526(02)00077-X (2003).
Glover, B. & Pierce, J. H. Are natural colorants good for your health?. J. Soc. Dyers. Colour. 109 , 5–7. https://doi.org/10.1111/j.1478-4408.1993.tb01491.x (1993).
Amutha, K. & Sudhapriya, N. Dyeing of textiles with natural dyes extracted from Terminalia arjuna and Thespesia populnea fruits. Ind. Crops. Prod. 148 , 112303. https://doi.org/10.1016/j.indcrop.2020.112303 (2020).
İşmal, Ö. E. & Yıldırım, L. In The Impact and Prospects of Green Chemistry for Textile Technology (eds Shahid ul, I. & Butola, B. S.) 57–82 (Woodhead Publishing, 2019).
Samanta, A. K. & Konar, A. In Natural Dyes vol. 3 (ed Emriye, A. K.) 134 (IntechOpen, 2011).
Mussak, R. A. & Bechtold, T. In Handbook of Natural Colorants. (eds Bechtold, T. & Mussak, R.A.M. ) 315–338 (Wiley, 2009).
Mongkholrattanasit, R., Kryštůfek, J., Wiener, J. & Viková, M. UV protection properties of silk fabric dyed with eucalyptus leaf extract. J. Text. I (102), 272–279. https://doi.org/10.1080/00405001003722369 (2011).
Shahidul, I. & Sun, G. Thermodynamics, kinetics, and multifunctional finishing of textile materials with colorants extracted from natural renewable sources. ACS. Sustain. Chem. Eng. 5 , 7451–7466. https://doi.org/10.1021/acssuschemeng.7b01486 (2017).
Zhang, Y., Shahidul, I., Rather, L. J. & Li, Q. Recent advances in the surface modification strategies to improve functional finishing of cotton with natural colourants—a review. J. Clean. Prod. 335 , 130313. https://doi.org/10.1016/j.jclepro.2021.130313 (2022).
Samanta, P., Singhee, D. & Samanta, A. K. Fundamentals of natural dyeing of textiles: Pros and cons. Curr. Tren. Fash. Tech. Text. Eng. 2 , 001–008. https://doi.org/10.19080/CTFTTE.2018.02.555593 (2018).
Derksen, G. C. H. et al. Development of a process for obtaining non-mutagenic madder root ( Rubia tinctorum ) extract for textile dyeing. Ind. Crops Prod. 164 , 113344. https://doi.org/10.1016/j.indcrop.2021.113344 (2021).
Li, W. et al. Expression of heat-resistant β-glucosidase in Escherichia coli and its application in the production of gardenia blue. Synth. Syst. Biotechnol. 6 , 216–223. https://doi.org/10.1016/j.synbio.2021.08.002 (2021).
Tsutsumiuchi, K. et al. Molecular structure of gardenia blue pigments by reaction of genipin with benzylamine and amino acids. J. Agric. Food Chem. 69 , 3904–3911. https://doi.org/10.1021/acs.jafc.0c07948 (2021).
Ozaki, A. et al. Genotoxicity of gardenia yellow and its components. Food Chem. Toxicol. 40 , 1603–1610. https://doi.org/10.1016/S0278-6915(02)00118-7 (2002).
Cai, Y. et al. Cationic modification of ramie fibers in liquid ammonia. Cellulose 25 , 4463–4475. https://doi.org/10.1007/s10570-018-1905-1 (2018).
Zhang, P. et al. Dyeing of raw ramie yarn with Reactive Orange 5 dye. Ind. Crops. Prod. 176 , 114315. https://doi.org/10.1016/j.indcrop.2021.114315 (2022).
Lin, L. et al. Combination of wet fixation and drying treatments to improve dye fixation onto spray-dyed cotton fabric. Sci. Rep.-UK 11 , 15403. https://doi.org/10.1038/s41598-021-94885-z (2021).
Inamdar, U. Y., Pervez, M. N., Navik, R. G., Peng, X. & Cai, Y. Low-temperature bleaching of cotton fabric by activated peroxide system. Emerg. Mater. Res. 6 , 387–395. https://doi.org/10.1680/jemmr.16.00148 (2017).
Liang, Y. et al. Post-treatment of reactive dyed cotton fabrics by caustic mercerization and liquid ammonia treatment. Cellulose 28 , 7435–7453. https://doi.org/10.1007/s10570-021-03984-7 (2021).
Liman, M. L. R., Islam, M. T., Hossain, M. M., Sarker, P. & Dabnath, S. Coloration of cotton fabric using watermelon extract: Mechanism of dye-fiber bonding and chromophore absorption. J. Text. I (112), 243–254. https://doi.org/10.1080/00405000.2020.1738036 (2021).
Steet, J. A. & Tong, C. H. Degradation kinetics of green color and chlorophylls in peas by colorimetry and HPLC. J. Food. Sci. 61 , 924–928. https://doi.org/10.1111/j.1365-2621.1996.tb10903.x (1996).
Ryan-Stoneham, T. & Tong, C. H. Degradation kinetics of chlorophyll in peas as a function of pH. J. Food. Sci. 65 , 1296–1302. https://doi.org/10.1111/j.1365-2621.2000.tb10600.x (2000).
Guesmi, A., Hamadi, N. B., Ladhari, N. & Sakli, F. Dyeing properties and colour fastness of wool dyed with indicaxanthin natural dye. Ind. Crops. Prod. 37 , 493–499. https://doi.org/10.1016/j.indcrop.2011.07.026 (2012).
Zhang, P. et al. Toward improved performance of reactive dyeing on cotton fabric using process sensitivity analysis. Int. J. Cloth. Sci. Tech. https://doi.org/10.1108/IJCST-03-2021-0035 (2022).
Kamel, M. M., Abdelghaffar, F. & El-Zawahry, M. M. Eco-friendly dyeing of wool with a mixture of natural dyes. J. Nat. Fibers. 8 , 289–307. https://doi.org/10.1080/15440478.2011.627114 (2011).
Lewis, D. M. & Rippon, J. A. The Coloration of Wool and Other Keratin Fibres (Wiley, 2013).
Book Google Scholar
Mehrparvar, L., Safapour, S., Sadeghi-Kiakhani, M. & Gharanjig, K. A cleaner and eco-benign process for wool dyeing with madder, Rubia tinctorum L., root natural dye. Int. J. Environ. Sci. Tech. 13 , 2569–2578. https://doi.org/10.1007/s13762-016-1060-x (2016).
Derksen, G. C. H. et al. Development of a process for obtaining non-mutagenic madder root ( Rubia tinctorum ) extract for textile dyeing. Ind. Crops. Prod. 164 , 113344. https://doi.org/10.1016/j.indcrop.2021.113344 (2021).
Hobbs, C. A. et al. Genotoxicity evaluation of the naturally-derived food colorant, gardenia blue, and its precursor, genipin. Food. Chem. Toxicol. 118 , 695–708. https://doi.org/10.1016/j.fct.2018.06.001 (2018).
Kwon, O. O. et al. Photovoltaic performance of TiO 2 electrode adsorbed with gardenia yellow purified by nonionic polymeric sorbent in dye-sensitized solar cells. Spectrochim. Acta. A. 136 , 1460–1466. https://doi.org/10.1016/j.saa.2014.10.037 (2015).
Yusuf, M. et al. Eco-dyeing of wool using aqueous extract of the roots of indian Madder ( Rubia cordifolia ) as natural dye. J. Nat. Fibers. 10 , 14–28. https://doi.org/10.1080/15440478.2012.738026 (2013).
Yatagai, M., Magoshi, Y., Becker, M. A., Sano, C., Ikuno, H., Kohara, N. & Saito, M. In Historic Textiles, Papers, and Polymers in Museums vol. 779 ACS Symposium Series (eds Jeanette M. C. & Mary, T. B.) Ch. 7, 86–97 (American Chemical Society, 2000).
Manhita, A. et al. Enlightening the influence of mordant, dyeing technique and photodegradation on the colour hue of textiles dyed with madder—a chromatographic and spectrometric approach. Microchem. J. 98 , 82–90. https://doi.org/10.1016/j.microc.2010.12.002 (2011).
Download references
Acknowledgements
This work was financially supported by the Opening Project of Hubei Key Laboratory of Biomass Fibers and Eco-Dyeing & Finishing, Project Number: STRZ202115.
Author information
These authors contributed equally: Lina Lin and Tiancheng Jiang.
Authors and Affiliations
Hubei Provincial Engineering Laboratory for Clean Production and High Value Utilization of Bio-Based Textile Materials, Wuhan Textile University, Wuhan, China
Lina Lin, Tiancheng Jiang, Lexin Xiao & Yingjie Cai
Engineering Research Centre for Clean Production of Textile Dyeing and Printing, Ministry of Education, Wuhan Textile University, Wuhan, China
Sanitary Environmental Engineering Division (SEED), Department of Civil Engineering, University of Salerno, 84084, Fisciano, Italy
Md. Nahid Pervez & Vincenzo Naddeo
TST Group Holding Ltd., Guangzhou, 510620, China
You can also search for this author in PubMed Google Scholar
Contributions
L.L. T.J. L.X. M.N.P. X.C. Y.C. done all the experimental work and wrote the experimental and results parts. L.L. M.N.P. X.C. V.N. Y.C. written and edited the abstract, introduction and literature review. L.L. M.N.P. L.X. X.C. V.N. Y.C. and all authors reviewed the manuscript.
Corresponding authors
Correspondence to Xiaobo Cai , Vincenzo Naddeo or Yingjie Cai .
Ethics declarations
Competing interests.
The authors declare no competing interests.
Additional information
Publisher's note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ .
Reprints and permissions
About this article
Cite this article.
Lin, L., Jiang, T., Xiao, L. et al. Sustainable fashion: eco-friendly dyeing of wool fiber with novel mixtures of biodegradable natural dyes. Sci Rep 12 , 21040 (2022). https://doi.org/10.1038/s41598-022-25495-6
Download citation
Received : 13 September 2022
Accepted : 30 November 2022
Published : 05 December 2022
DOI : https://doi.org/10.1038/s41598-022-25495-6
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
This article is cited by
Screening of vegetal dyes for wool dyeing: selection of feasible alternatives for industrial applications.
- Uğur Ergünay
Fibers and Polymers (2024)
By submitting a comment you agree to abide by our Terms and Community Guidelines . If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.
Quick links
- Explore articles by subject
- Guide to authors
- Editorial policies
Sign up for the Nature Briefing: Translational Research newsletter — top stories in biotechnology, drug discovery and pharma.
Beyond Natural Dyes: Embracing Sustainable Innovations in Industrial Textile Dyeing
![research paper about natural dye Author ORCID: We display the ORCID iD icon alongside authors names on our website to acknowledge that the ORCiD has been authenticated when entered by the user. To view the users ORCiD record click the icon. [opens in a new tab]](https://chemrxiv.org/engage/assets/public/chemrxiv/images/logos/orcid.png)
Version History
Version notes, author’s competing interest statement.
- Open access
- Published: 14 July 2015
Extraction of eco-friendly natural dyes from mango leaves and their application on silk fabric
- Mohammad Gias Uddin 1
Textiles and Clothing Sustainability volume 1 , Article number: 7 ( 2015 ) Cite this article
56k Accesses
29 Citations
1 Altmetric
Metrics details
The aim of the study was to evaluate the performance of dyes extracted from mango leaves in silk dyeing. Extraction medium was optimized by extracting dyes from fixed quantity of crushed leaves under pH values from 3 to 12. The maximum relative color strength of the extracted dye liquor was found to be at pH 10. The optimum dye extraction conditions i.e., the temperature, time, and material-to-liquor ratio were found to be 98 °C, 60 min, and 1:10, respectively. Dyeing was carried out with the optimized dye extract on mordanted and unmordanted silk fabrics. The dyed materials were evaluated by measuring the color yield and fastness properties. It was concluded that the color values were found to be influenced by the addition of mordants, consequently different fashion hues were obtained from the same dye extract using different mordants. It can also be said that mango leaves have good potentiality for dyeing of silk fabric.
Textile dyeing industry at present uses excessive amount of synthetic dyes to meet the required coloration of global consumption of textiles due to cheaper prices, wider ranges of bright shades, and considerably improved fastness properties in comparison to natural dyes (El-Nagar et al. 2005 ; Iqbal et al. 2008 ). But the production of synthetic dyes is dependent on petrochemical source, and some of these dyes contain carcinogenic amines (Hunger 2003 ). The application of such dyes causes serious health hazards and influences negatively the eco-balance of nature (Bruna and Maria 2013 ; Goodarzian and Ekrami 2010 ; Jothi 2008 ). Moreover, many countries already imposed stringent environment standards over these dyes. For instance, Germany has banned the azo dyes (Almahy et al. 2013 ). In this situation, a higher demand is put towards the greener alternatives or agricultural residues (Ammayappan et al. 2014 ). As a result, natural dyes are among the promising options for developing a greener textile dyeing process and such interest is reflected to the increased number of recent publications. Plant leaves are potential sources of natural dyes because of their easy availability and abundant nature.
Silk has been known as the “queen of fibers” since its discovery. Clothes made from silk are luxurious and have many excellent qualities including the material’s luster, light weight, superior mechanical performance, fine and smooth texture, excellent moisture transportation, and excellent draping quality (Cai et al. 2001 ). Mango bark has been reported to be used on silk and cotton materials as a source of natural dyes, and a wide range of colors have been produced using different mordants (Bains et al. 2003 ; Win and Swe 2008 ). On the other hand, the use of acid activated mango leaf powder (MLP) has been reported in another study for the removal of the Rhodamine B (RB) dye from aqueous solution (Khan et al. 2011 ). However, apart from this application of mango leaves, different leaves such as peach, poinsettia, acalypha, and parthenium leaves have also been reported to extract colors which were used in dyeing of silk materials (Mahajan et al. 2005 ; Rawat et al. 2006 ; Saravanan et al. 2013 ; Suneeta and Mahale 2002 ) while mango leaves have been reported to be used in batik painting technique on silk fabric in comparison with other four natural dyes (Klaichoi and Padungtos 2010 ). There is scope to extract color from mango leaves for the use in dyeing of silk fabric in order to get different fashion hues. The aim of the research was to evaluate the performance of dyes extracted from mango leaves in silk dyeing. The specific objectives were to analyze the aqueous extraction process of the dyes, to explore the possibilities of producing fashionable hues from the dyes using different mordants, to compare between unmordanted and mordanted dyed fabrics, to analyze the color values, and to assess the color fastness properties of dyed fabric.
Mango leaves used for the extraction purpose was collected from Roads & Highways Department, Dhaka, Bangladesh. Mangiferin as shown in Fig. 1 (1,3,6,7-tetrahydroxyxanthone-C-2-β-D-glucoside) was the chemical responsible (Luo et al. 2012 ) for providing color from mango leaves.
Chemical structure of mangiferin
Plain weave (1/1) raw silk fabric (22 g/m 2 fabric) purchased from Sopura Silk Limited, Dhaka, was used for this study.
The leaves were washed thoroughly with water to remove dirt. They were dried under direct sunlight and grinded into very small units with the help of a grinding machine. The wastages are removed using a fine strainer, and finally, weight was taken. After drying, crushing, and removing wastages, the weight of 1 kilogram leaves was found to be 318 gram. Raw, dried, and crushed leaves are shown in Fig. 2 .
( a ) Raw, ( b ) dried, and ( c ) crushed mango leaves
The color component was extracted from the leaves in aqueous extraction process. Extraction was carried out with fixed quantity of crushed leaves (10 gram) under ten different pH values from pH 3 to 12 with a liquor ratio of 1:10 (Weight of crushed leaves in gram; amount of water in milliliter) at 98 °C for 60 min to optimize extraction medium. In each process of extraction, the mixture was cooled down and finally the dye extracts were filtered with fine filter paper three times to ensure clear dye solution.
The dye extracts obtained at different pH values were used for obtaining standard calibration curves through their absorbance values found using a dual beam reflectance spectrophotometer. The dilution of the extracts was carried out for the linear dependence on the concentration-absorbance relation at an absorbance peak ( λ max ). The absorbance values of extracted dye liquors under alkaline (pH 8–12) and acidic (pH 3–6) conditions were considered as batches, and relative color strength values of these batch solutions were measured from the spectrophotometer by comparing with the absorbance value of extracted dye liquor under neutral condition (pH 7) which was considered as standard.
Again, the dye extract which gave the maximum color strength was utilized to optimize the extraction levels of temperature, time, and material-to-liquor ratio. An orthogonal design of experiments was undertaken for this purpose.
Raw silk fabric was degummed in an aqueous solution containing soap (15 g/L), sequestering agent (1 g/L), and wetting agent (1 g/L) maintaining the bath at pH 9. The material-to-liquor ratio during the treatment was maintained at 1:50. The temperature was gradually raised to 80 °C and run for 60 min. The degummed fabric was washed with 2 g/L detergent at 65 °C for 10 min.
The degummed fabric was bleached by treating with 35 % hydrogen peroxide (3 mL/L), sequestering agent (1 g/L), wetting agent (1 g/L), and trisodium phosphate (2 g/L), maintaining a material-to-liquor ratio of 1:50 at pH 9 and temperature 60 °C for 60 min followed by washing with 2 g/L detergent at 65 °C for 10 min. CIE whiteness index of the bleached fabric was found to be 63.26.
Pre-mordanting was carried out on silk fabric using 5 % (on fabric weight) of ferrous sulfate, alum (potassium aluminum sulfate), and tin (stannous chloride) mordants individually and using four different combinations of mordants such as ferrous sulfate-alum (2.5 + 2.5 %), ferrous sulfate-alum-tin (2 + 2 + 1 %), alum-tin (2.5 + 2.5 %), and alum-tin-tannic acid (2 + 2 + 1 %) at 60 °C for 60 min keeping a material-to-liquor ratio of 1:30. Again, cream of tartar (CT) was used as a mordant assistant (Mortazavi et al. 2012 ) with stannous chloride, written as tin-CT.
Dyeing was carried out IR sample dyeing machine with the optimized dye extract as per standard parameters recommended for silk fabric, reported in Clariant manual, i.e., at 80 °C for 60 min under pH 5, keeping a material-to-liquor ratio of 1:50. Opticid PSD (1.5 g/L) was used as a buffering agent in the extracted dye liquor.
Color yield of dyed fabrics
Dyed samples were analyzed by measuring the reflectance curve between 350 and 750 nm with the spectrophotometer with illuminant D 65 at 10 0 observer. The minimum of the curve ( R min ) was used to determine the ratio of light absorption ( K ) and scatter ( S ) via the Kubelka-Munk function (Mcdonald 1997 ).
Color coordinates of dyed fabrics
The color coordinates of the dyed samples were determined based on the CIELab system via the spectrophotometer. In addition, ∆E CMC value was determined to show the color difference between mordanted and unmordanted samples.
Color fastness
Washing and light fastness tests were carried out in ISO 105 C02 and ISO 105 B02 method, respectively.
Results and discussion
Color strength of extracted dye liquors.
Optimum pH was selected based on the relative color strength value of the extracted dye liquor at which maximum color was extracted. Changes in color strength were found due to changes in pH as shown in Table 1 .
It can be seen from the extraction results that the extracted solution showed maximum color strength at pH 10 which was 108.5. It was also found that from the neutral condition (pH 7), relative color strength values gradually decreased up to pH 4 and then increased at pH 3. The reason of extracting more coloring component in alkaline medium was due to the presence of acidic phenolic groups in mangiferin which reacted with alkali and formed more soluble salts in water as shown in Fig. 3 . The solubility of the coloring component was increased due to the increased ionization of hydroxyl (phenoxide) groups in alkaline medium (Ali 2007 ).
Reaction of mangiferin with caustic soda
Again, increasing the pH from neutral condition improved the color strength of the extracted dye liquors up to a certain point. A further increase in alkaline pH resulted in decrease in the color strength of the extract. This decline in color strength was due to the high reactivity of mangiferin in concentrated alkaline medium (Spyroudis 2000 ).
Furthermore, the cell wall of leaves is composed of cellulosic material which gains anionic charge under alkaline medium. Because of these anionic repulsive forces among the cell walls, they lose their strength and ruptured easily (Ali 2007 ). In addition, as the observed leaf dyes have polyphenolic chromophoric structure, hence better extractions were observed using aqueous method (Sivakumar et al. 2009a , b ).
Study of pH stability of dye extracts
It was noticeable during extraction that pH of the extraction bath changed gradually with time. Table 2 shows the pH variation after filtration and with time elapsed.
pH was found to be decreased in all the extraction baths from pH 3 to 12. This was due to the release of acidic color components from the leaves during extraction. From the neutral pH bath where the pH was set 7, the higher the alkalinity of the extraction bath, the greater was the pH drop rate. The drop rate became gradually slower while gradually approaching to more acidic bath from neutral bath. The pH was also measured after 24 h of the filtration process to notice the stability of the extracted bath at acidic pH, and no major noticeable change was reported.
Again, the dyes can show resonating form and give different tones with the change in pH because for natural dyes, pH changes very often. Furthermore, silk dyeing is recommended to be carried out in acidic medium as silk is sensitive to alkaline medium of dyeing, but extraction of the mangiferin dyes was optimized at alkaline pH. Therefore, the stability of the dyes after extraction is of importance.
Optimization of aqueous extraction conditions
The levels for each of the three factors in the orthogonal design of experiments are shown in Table 3 . The extraction experiments were performed under optimum pH condition (pH = 10). The results of the orthogonal design of experiments are shown in Table 4 .
Optimum factors: A 3 B 2 C 1 ,
Absorbance = 1.041.
The optimum extraction conditions were 98 °C for temperature factor, 60 min for time factor, and 1:10 for material-to-liquor ratio. It has been found that dye liquor extracted under optimum conditions had the maximum absorbance value, which was 1.041. From the range analysis of the average absorbance results as shown in Table 4 , the most influential factor of extraction was material-to-liquor ratio, while extraction time factor was the least influential.
Dyed samples
The use of mordants and their combinations produced different shades on silk fabric which are shown in Table 5 .
Color measurements of dyed fabrics
The results of color measurements of the dyed silks are shown in Table 6 .
K / S value of the unmordanted dyed sample was found to be 11.85. This dye uptake on the silk fiber is attributed to the structural features of the fiber. However, in the mordanting method, mordant resulted in improved color yield of the dyed fabrics, except tin. Ferrous sulfate as a mordant significantly increased the color yield of silk. The K / S value was found to be 17.46 using ferrous sulfate which showed the maximum relative surface color strength value of 147.4 % considering the unmordanted dyed sample as reference. Besides, using alum with ferrous sulfate, and tin and alum with ferrous sulfate as a combination, color strengths were found to be 140.3 % ( K / S = 16.62) and 121.7 % ( K / S = 14.42), respectively.
In single mordanting process of silk, the order of color yield was found to be ferrous sulfate > alum > tin. It was obvious that color yield gradually decreased when approached from ferrous sulfate to tin. Again, among the four different combinations of mordants, the order was found to be ferrous sulfate-alum > alum-tin-tannic acid > ferrous sulfate-alum-tin > alum-tin.
The addition of ferrous sulfate mordant increased the greenness quality 21.58 % when compared with the reference dyed sample. Tin reduced 22.23 % redness while tin-CT increased 34.05 % redness of the reference dyed sample. Again, from b * values, it was noticed that all the ferrous sulfate mordanted samples were bluer than the reference samples while tin-CT and alum-tin-TA mordanted samples increased yellowness of dyed fabric. The color saturation value ( C *) were found to be least in ferrous sulfate mordanted sample (8.7) whereas the values were found to be maximum in the case of alum (32.3) and tin-CT (32.6) mordanted samples. Moreover, the hue angles lie within 67.8° to 83.7°, so all of the dyed samples were closer to yellowish shade than the reddish. Higher color difference (∆ E CMC ) was noticeable between reference and ferrous sulfate mordanted samples, and the difference reduced from ferrous sulfate to alum and then alum to tin as shown in Table 6 .
The presence of hydroxyl or carbonyl groups in dye structure is capable to form metal complex with the positively charged metals. Dye anions and metal cations have strong attraction towards positively charged amino and negatively charged carboxyl groups of silk, respectively. Hence, they form ionic bonding between dye and fiber, metal and fiber, and finally dye and metal ions. The dye-metal complex also forms coordinate bonds with the uncharged amine (−NH 2 ) groups of silk as shown in Fig. 4 . In addition, one molecule of dye can form a bond with one site of fiber molecule while one molecule of mordant can form bonds with two or more molecules of dyes. Therefore, these are some of the different features indicating application of mordants increased the color yield (Bhattacharya and Shah 2000 ; Temani et al. 2011 ; Uddin 2014 ).
Structure of mangiferin with ferrous sulfate on silk
Again, ferrous sulfate as a transition metal having coordination number 6 forms a large number of complexes with the dye molecules (Mongkholrattanasit and Punrattanasin 2012 ). As a result, when they interact with the silk fiber, some coordination sites remain free, and at that time, amino and carboxylic groups on the fiber can occupy these free sites. Thus, ferrous sulfate can form a ternary complex on one site with the fiber and in another site with the dye (Fig. 4 ). This strong coordination tendency can enhance interaction between the fiber and the dye (Bhattacharya and Shah 2000 ). This resulted in higher dye uptake as well as shade change due to mordanting with ferrous sulfate (Uddin 2014 ).
In contrast, aluminum and tin salts formed weak coordination complexes with the dyes. This tends to form quite strong bonds with the dye molecule but not with the fiber (Cotton and Wilkinson 1972 ). Thus, they block the dye and reduce its interaction with the fiber. This is the reason behind the lower K / S values in the case of aluminum and tin salts than those obtained from ferrous sulfate. Moreover, CT as an assistant increased the color yield from 11.16 to 13.19 when used with tin. CT is chemically potassium hydrogen tartrate which can be used in addition to dyes and mordants to change the pH in order to change colors and to help the absorption of the mordant metal (Mortazavi et al. 2012 ).
Fastness results
Washing fastness.
The results of washing and light fastness of the dyed fabrics are shown in Table 7 .
The unmordated dyed silk showed color change rating of 4. This can be explained that the good fastness to washing for the sample dyed without mordant was due to the affinity of coloring component through H-bonding and van der Waals forces. Using mordants, the color change ratings were found to be within 3/4 to 5, where a rating of 5 (excellent) was found using tin-CT mordant. The ratings were found to be 4/5 in the case of using alum and alum-tin. So it can be said that the overall ratings of color change were good. As wash fastness is influenced by the rate of diffusion of dye molecules and state of dyes inside the fiber, dyes has a tendency to aggregate inside the fiber. Thus, their molecular size is increased resulting in good wash fastness. In addition, in the case of mordanted samples, complexing with mordant also has the effect of insolubilizing the dye, making it color fast.
On the other hand, the color staining ratings were found to be from 4/5 to 5 for all the dyed fabrics, except when ferrous sulfate and its combinations were used as mordant. There were very slight staining observed on to the adjacent wool fiber of the multifiber fabric in the case of ferrous sulfate and its combination samples where the ratings were 4 and almost no staining on the other fibers of the multifiber fabric.
Light fastness
Light fastness as shown in Table 7 was found to be better, and among those, the lowest ratings attained were 5 in the case of tin and alum-tin combination while the unmordanted dyed fabric showed a rating of 6.
In the case of metallic mordants, ferrous sulfate mordanted samples dyed with the mango leaf extracts showed excellent light fastness. This happened due to the formation of a complex with transition metal which protected the chromophore from photolytic degradation. The photons sorbed by the chromophoric group dissipated their energy by resonating within the six-member ring thus formed and, hence, protecting the dyes. Thus, ferrous sulfate can bind with more dye molecules than alum or tin. During exposure to light, the fabrics mordanted with ferrous sulfate, alum, or tin may have the same number of dye molecules destroyed. But as the fabrics mordanted with ferrous sulfate had deeper shades due to bonding with more number of dye molecules, it seemed to fade less compared to the fabric mordanted with alum or tin.
Conclusions
This study was planned in search of greener alternative to satisfy the consumers’ growing demand of eco-friendly products, and progress has been made with this study in the use of mango leaves extracts. The maximum relative color strength of the extracted dye liquor was found to be at pH 10. But the extracted dye liquors have shown good pH stability in acidic conditions. It was shown that different fashion hues were obtained on silk fabric from the same dye extract using mordants and their combinations. Again, color yields were found to be influenced by the addition of mordants. In single mordanting, the order of color yield was ferrous sulfate > alum > tin. In combined mordanting, the order was ferrous sulfate-alum > alum-tin-tannic acid > ferrous sulfate-alum-tin > alum-tin. Other color values were also found to be influenced due to mordanting. Washing and light fastness properties were found to be from good to excellent in most of the cases. Thus, on the basis of the results, it can be said that mango leaves have good scope for application on silk fabrics.
Ali, S. (2007). Evaluation of cotton dyeing with aqueous extracts of natural dyes from indigenous plants (PhD Thesis) (pp. 62–63). Pakistan: University of Agriculture, Faisalabad. 96.
Google Scholar
Ali, MA, Almahy, HA, & band Ali, AA. (2013). Extraction of carotenoids as natural dyes from the Daucus carota Linn (carrot) using ultrasound in Kingdom of Saudi Arabia. Research Journal of Chemical Sciences, 3 (1), 63–66.
CAS Google Scholar
Ammayappan, L, Shakyawar, DB, Chhagan, L, Sharma, M, & Wani, SA. (2014). Extraction of natural colourants from agricultural residues and their application on woolen fabric: part 2. Man Made Textiles In India, 57 (6), 212–215.
Bains, S, Singh, OP, Goraya, G, & Kang, M. (2003). Dyeing of cotton with mango ( Mangifera indica ) dye. Journal of the Textile Association, 64 (1), 23–26.
Bhattacharya, SD, & Shah, AK. (2000). Metal ion effect on dyeing of wool fabric with catechu. Color Technol, 116 (1), 10–12.
Article CAS Google Scholar
Bruna, CV, & Maria, AMM. (2013). Azo dyes: characterization and toxicity—a review. Textiles and Light Industrial Science and Technology, 2 (2), 85–103.
Cai, Z, Jiang, G, & Yang, S. (2001). Chemical finishing of silk fabric. Color Technol, 117 , 161.
Clariant manual. Recommendations for dyeing silk. Sandoz Chemicals Ltd., Switzerland, ID 05543.00.94.
Cotton, FA, & Wilkinson, G. (1972). Advanced inorganic chemistry: a comprehensive text (3rd ed.). New York: John Wiley & Sons.
El-Nagar, K, Sanad, SH, Mohamed, AS, & Ramadan, A. (2005). Mechanical properties and stability to light exposure for dyed Egyptian cotton fabric with natural and synthetic dyes. Polymer-plastics Technology and Engineering, 44 (7), 1269–1279. doi: 10.1080/03602550500207816 .
Goodarzian, H, & Ekrami, E. (2010). Extraction of dye from madder plant ( Rubia tinctorium ) and dyeing of wool. World Applied Sciences Journal, 9 (4), 434–436.
Hunger, K. (2003). Industrial dyes . Darmstadt: WILEY-VCH Verlag GmbH & Co. KGaA.
Iqbal, J, Bhatti, IA, & Adeel, S. (2008). Effect of UV radiation on dyeing of cotton fabric with extracts of henna leaves. Indian Journal of Fiber & Textile Research, 33 , 157–162.
Jothi, D. (2008). Extraction of natural dyes from African marigold flowers ( Tagetes ereectal ) for textile coloration. AUTEX Research Journal, 8 (2), 49.
Khan, TA, Sharma, S, & Ali, I. (2011). Adsorption of Rhodamine B dye from aqueous solution onto acid activated mango ( Magnifera indica ) leaf powder: equilibrium, kinetic and thermodynamic studies. Journal of Toxicology and Environmental Health Sciences, 3 (10), 286–297.
Klaichoi, C, & Padungtos, W. (2010). Development of batik painting technique silk fabric via natural dyes. In The 2nd RMUTP International Conference, Green Technology and Productivity (pp. 382–387).
Luo, F, Lv, Q, Zhao, Y, Hu, G, Huang, G, Zhang, J, et al. (2012). Quantification and purification of mangiferin from Chinese mango ( Mangifera indica L.) cultivars and its protective effect on human umbilical vein endothelial cells under H 2 O 2 -induced stress. Int J Mol Sci, 13 , 11260–11261. doi: 10.3390/ijms130911260 .
Mahajan, S, Sidhu, SP, & Grewal, J. (2005). Dyeing of silk with peach ( Prunus persica ) dye using combination of mordants. Journal of the Textile Association, 66 (2), 85–89.
Mcdonald, R (Ed.). (1997). Color physics for industry (2nd ed.). Soc . Bradford: Dyers and Colorists.
Mongkholrattanasit, R, & Punrattanasin, N. (2012). Properties of silk fabric dyed with eucaliptus, quercetin, rutin and tannin using padding techniques . Bangkok, Thailand: RMUTP International Conference on Textiles & Fashion.
Mortazavi, SM, Kamali Moghaddam, M, Safi, S, & Salehi, R. (2012). Saffron petals, a by-product for dyeing of wool fibers. Prog Color Colorants Coat, 5 , 75–84.
Rawat, B, Jahan, S, Grover, E, & Yadav, S. (2006). Color fastness properties of silk fabric dyed with poinsettia leaves. Asian Textile Journal, 15 (4), 43–45.
Saravanan, P, Chandramohan, G, Mariajancyrani, J, & Shanmugasundaram, P. (2013). A study on extraction and application of eco-friendly natural dye extracted from leaves of Acalypha indica Linn on silk fabric. International Journal of Textile and Fashion Technology, 3 (5), 1–8.
Sivakumar, V, Jayapriya, J, Shriram, V, Srinandini, P, & Swaminathan, G. (2009a). Ultrasound assisted enhancement in wattle bark ( Acacia mollissima ) vegetable tannin extraction for leather processing. J Am Leather Chem Assoc, 104 (11), 375–383.
Sivakumar, V, Lakshmi, A, Vijayeeswaree, J, & Swaminathan, G. (2009b). Ultrasound assisted enhancement in natural dye extraction from beetroot for industrial applications and natural dyeing of leather. UltrasonSonochem, 16 (6), 782–789. doi: 10.1016/j.ultsonch.2009.03.009 .
Spyroudis, S. (2000). Hydroxyquinones: synthesis and reactivity. Molecules, 5 , 1291–1330. doi: 10.3390/51201291 .
Suneeta, MB, & Mahale, G. (2002). Dye from Parthenium leaves. Man-made Textiles in India, 45(5), 198–200.
Temani, P, Shakyawar, DB, Ammayappan, L, Goyal, V, & Wani, SA. (2011). Standardization of dyeing condition of cochineal extract on pashmina yarn. Journal of the Textile Association, 72 (2), 90–92.
Uddin, MG. (2014). Effects of different mordants on silk fabric dyed with onion outer skin extracts. Journal of Textiles, Article ID 405626 , 1–8. doi: 10.1155/2014/405626 .
Article Google Scholar
Win, ZM, & Swe, MM. (2008). Purification of the natural dyestuff extracted from mango bark for the application on protein fibers. World Acad Sci Eng Technol, 22 , 536.
Download references
Author information
Authors and affiliations.
Department of Textile Engineering, Faculty of Engineering, Ahsanullah University of Science and Technology, Dhaka, 1208, Bangladesh
Mohammad Gias Uddin
You can also search for this author in PubMed Google Scholar
Corresponding author
Correspondence to Mohammad Gias Uddin .
Additional information
Competing interests.
The author declares that he has no competing interest.
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License ( https://creativecommons.org/licenses/by/4.0 ), which permits use, duplication, adaptation, distribution, and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
Reprints and permissions
About this article
Cite this article.
Uddin, M.G. Extraction of eco-friendly natural dyes from mango leaves and their application on silk fabric. Text Cloth Sustain 1 , 7 (2015). https://doi.org/10.1186/s40689-015-0007-9
Download citation
Received : 06 April 2015
Accepted : 02 June 2015
Published : 14 July 2015
DOI : https://doi.org/10.1186/s40689-015-0007-9
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Ferrous Sulfate
- Silk Fabric
- Light Fastness
Open Access is an initiative that aims to make scientific research freely available to all. To date our community has made over 100 million downloads. It’s based on principles of collaboration, unobstructed discovery, and, most importantly, scientific progression. As PhD students, we found it difficult to access the research we needed, so we decided to create a new Open Access publisher that levels the playing field for scientists across the world. How? By making research easy to access, and puts the academic needs of the researchers before the business interests of publishers.
We are a community of more than 103,000 authors and editors from 3,291 institutions spanning 160 countries, including Nobel Prize winners and some of the world’s most-cited researchers. Publishing on IntechOpen allows authors to earn citations and find new collaborators, meaning more people see your work not only from your own field of study, but from other related fields too.
Brief introduction to this section that descibes Open Access especially from an IntechOpen perspective
Want to get in touch? Contact our London head office or media team here
Our team is growing all the time, so we’re always on the lookout for smart people who want to help us reshape the world of scientific publishing.
Home > Books > Chemistry and Technology of Natural and Synthetic Dyes and Pigments
Fundamentals of Natural Dyes and Its Application on Textile Substrates
Submitted: 15 May 2019 Reviewed: 30 September 2019 Published: 22 December 2019
DOI: 10.5772/intechopen.89964
Cite this chapter
There are two ways to cite this chapter:
From the Edited Volume
Chemistry and Technology of Natural and Synthetic Dyes and Pigments
Edited by Ashis Kumar Samanta, Nasser S. Awwad and Hamed Majdooa Algarni
To purchase hard copies of this book, please contact the representative in India: CBS Publishers & Distributors Pvt. Ltd. www.cbspd.com | [email protected]
Chapter metrics overview
4,582 Chapter Downloads
Impact of this chapter
Total Chapter Downloads on intechopen.com
Total Chapter Views on intechopen.com
The meticulous environmental standards in textiles and garments imposed by countries cautious about nature and health protection are reviving interest in the application of natural dyes in dyeing of textile materials. The toxic and allergic reactions of synthetic dyes are compelling the people to think about natural dyes. Natural dyes are renewable source of colouring materials. Besides textiles it has application in colouration of foods, medicine and in handicraft items. Though natural dyes are ecofriendly, protective to skin and pleasing colour to eyes, they are having very poor bonding with textile fibre materials, which necessitate mordanting with metallic mordants, some of which are not eco friendly, for fixation of natural dyes on textile fibres. So the supremacy of natural dyes is somewhat subdued. This necessitates newer research on application of natural dyes on different natural fibres for completely eco friendly textiles. The fundamentals of natural dyes chemistry and some of the important research work are therefore discussed in this review article.
- colour fastness
- extraction of natural dyes
- natural dyes
Author Information
Virendra kumar gupta *.
- M.L.V. Textile and Engineering College, Bhilwara, Rajasthan, India
*Address all correspondence to: [email protected]
1. Introduction
No health hazard
Easy extraction and purification
No effluent generation
Very high sustainability
Mild dyeing conditions
Renewable sources
Mostly applicable to natural fibres (cotton, linen, wool and silk)
Poor colour fastness properties
Poor reproducibility of shades
No standard colour recipes and methods available.
Use of metallic mordants, some of which are not eco friendly.
Hill [ 1 ] had given his views that research work with natural dyes is inadequate, and there is need of significant research work to explore the potentials of natural dyes before its important application to textile substrate.
In India initially Alps Industries Ghaziabad (Uttar Pradesh, India) and later Ama Herbals, Lucknow, and Bio Dye Goa done extensive work for industrial research and production of natural dyes and natural dyed textiles. Textile-based handicraft industries in many countries engaged local people to dye textile yarn with natural dyes and weave them to produce specialty fabrics. Printing of textile fabrics with natural dyes in India are specially done in Rajasthan and Madhya Pradesh.
Vegetable origin
Animal origin
Mineral origin
Natural dyes are having wide application in the colouration of most of the natural fibres, e.g. cotton, linen, wool and silk fibre, and to some extant for nylon and polyester synthetic fibre. However, the major issues for natural dyed textiles are reproducibility of shade, non availability of well-defined standard procedure for application and poor lasting performance of shade under water and light exposure. To achieve good colour fastness to washing and light are also a challenge to the dyer. Several researchers had proposed different dyeing methods and process parameters, but still these information are inadequate, so this calls for the need of research to develop some standard dye extraction technique and standardisation of whole process of natural dyeing on textiles. Here there are examples of few important natural dyes [ 17 ] which are widely used in the dyeing of textile materials, described below.
1.1 Jack fruits ( Artocarpus heterophyllus Lam)
It is a very popular fruit of south India and other parts of India. The wood of the tree is cut into small chips and crushed into dust powder and then subsequently boiled in water to extract the dye. After mordanting treatment of dyed fabrics, yellow to brown shades are obtained. The cotton and jute fabrics are dyed by this dye. It belongs to the family of Moraceae. The dye consists of morin as colouring molecule ( Figure 1 ).

Molecular structure of morin (3,5,7,2′,4′pentahydroxy-flavone).
1.2 Turmeric ( Curcuma longa )
The dye is obtained from the root of the plant. The turmeric root is dried, crushed in powder form and boiled with water to extract the dye. It can be used in the dyeing of cotton, wool, and silk. Proper mordanting treatment improves colour fastness to wash. The brilliant yellow shade is obtained after dyeing with turmeric natural dye. Turmeric is a rich source of phenolic compounds known as curcuminoids. The colouring ingredients in turmeric are called curcumin. Curcumin is diarylheptanoid existing in keto-enol form. Turmeric is a member of Curcuma botanical group ( Figure 2 ).

Molecular structure of curcumin (diarylheptanoid).
1.3 Onion ( Allium cepa )
The papery skin of onion is the main source of the dye. Onion skin is boiled to extract the colour and subsequently can be dyed with or without mordanting the fabric. The resulting colour is from orange to brown. It contains colouring pigments called pelargonidin (5,5,7,4 tetrahydroxy antocyanidol). The amount of colouring pigment present varies from 2.0 to 2.25% ( Figure 3 ).

Molecular structure of pelargonidin (5,5,7,4 tetrahydroxy antocyanidol).
1.4 Hina ( Lawsonia inermis L)
It is the leaf of the plant that is traditionally used in making the coloured design on the hands of women. The leaf of the plant is dried, crushed and subsequently boiled with water to extract the dye from leaf. The mordanted fabric gives colour from brown to mustard yellow. This is the dispersed dye type colour; hence, polyester and nylon can be dyed by hina. However, it stains wool and silk giving a lighter brown colour. Hina is commonly known as lawsone. The chief constituent of hina leaves is hennotannic acid; it is a red orange pigment. Chemically hennotannic acid is 2-hydroxy-1,4-naphthoquinone. The colouring molecules have strong substantivity for protein fibre ( Figure 4 ).

Molecular structure of lawsone (2-hydroxyl-1,4-naphthoquinone).
1.5 Indigo ( Indigofera tinctoria )
It is the seed of the plant. The full matured plant has 0.4% colour on weight of the plant. The plants are steeped in the water until the fermentation start. When the hydrolysis of glucoside is completed, the liquor is separated from the plant debris. The extract is aerated which converts indoxyl to indigotin which separates out as a precipitate. The shade of natural indigo is difficult to reproduce exactly. The variety of blue shade on cotton can be obtained by the application of natural Indigo. It is kind of vat dye and hence need reductive vatting with liquid jiggery and citric acid or dithionate.
The precursor to indigo is indican which is a colourless water-soluble compound. Indican hydrolyzes in water and releases β-D-glucose and indoxyl. The oxidation of indoxyl resulted in indigotin. The average yield of indican from an indigo plant is 0.2–0.8%. Indigo is also present in molluscs. The molluscs contain mixture of indigo and 6,6′-dibromo indigo (red), which together produce a colour known as Tyrian purple. During dyeing due to air exposure, dibromo indigo is converted into indigo blue, and the mixture produces royal blue colour ( Figure 5 ).

Molecular structure of natural indigo.
1.6 Madder or manjistha or Rubia ( Rubia tinctorum )
The dye is obtained from the root of the plant. The root is scrubbed, dried in sunlight and finally boiled in the water to extract the dye in solution. The dye has red colour. The cotton, silk and wool fibre can be dyed with madder at a temperature of 100°C for time period of 60 min, and subsequently dye solution is cooled. Bright red shade is produced on wool and silk and red violet colour on cotton. This is a mordantable type of acid dye having phenolic (-OH) groups. The colouring matter in madder is alizarin of the antharaquinone group. The root of the plant contains several polyphenolic compounds, which are 1,3-dihydroxyanthraquinone, 1,4-dihydroxyanthraquinone, 1,2,4-trihydroxyanthraquinone and 1,2-dihydroxyanthraquinone ( Figures 6 and 7 ).

Molecular structure of alizarin and purpurin.

Molecular structure of 1,4-dihydroxyanthraquinone and 1,8-dihydroxyanthaquinone.
1.7 Tea waste ( Camellia sinensis )
India is one of the biggest consumer of tea. The left over waste of tea is collectable in large quantity. The extract of tea waste can be used as a natural dye in combination with different mordants, which can produce yellowish brown to brown shade. This is a mordantable dye. Flavonoids, flavonols and phenolic acids are the main colouring components in waste of the tea. Polyphenols, which are mostly flavonols, are known as catechins with epicatechin and its derivatives.
1.8 Safflower ( Carthamus tinctorius )
The safflower petals are soaked in distilled water and subsequently boiled with water for more than 2 h, and it is repeated two times. The solution is filtered and the filtrate is vacuum dried. The obtained powder is having strength of 20–30%. In dyeing it produces cherry red to yellowish red shade. Safflower contains natural pigment called carthamine. The biosynthesis of carthamine takes place by chalcone (2,4,6,4-tetrahydroxy chalcone) with two glucose molecules and that resulted in the formation of safflor A and safflor B ( Figure 8 ).

Molecular structure of carthamine (safflower).
1.9 Sappan wood ( Caesalpinia sappan )
Aqueous extraction is used to extract the dye from sappan wood. Alkali extraction can also be used. It produces bright red colour. It produces an orange colour in combination with turmeric and maroon shade with catechu. The sappan wood tree is found in India, Malaysia and the Philippines. The colouring pigment is similar to logwood. The same dye is also present in Brazil wood.
1.10 Logwood ( Haematoxylon compechianum )
The dye is extracted from the stem of the tree. The stems are broken into small pieces and steepened in cold water for several hours followed by boiling. The extracted dye solution is strained. The logwood natural dye is used to produce black shade on the wool. The logwood trees are found in Mexico, Central America and the Caribbean islands. It is also known as compeachy wood. The colouring matter in logwood natural dye is haematoxylin, which after oxidation forms haematein during isolation ( Figures 9 and 10 ).

Molecular structure of haematoxylin and brazilin.

Molecular structure of haematein.
1.11 Saffron ( Crocus sativus )
The dye is extracted from the stigma of flower, which is boiled in water, and the colour is extracted. It imparts a bright yellow colour to the textile material. The wool, silk and cotton can be dyed with saffron. Alum mordant produces orange yellow shade which is also called saffron yellow. This is also used as food colouring. Saffron is a perennial plant which belongs to the Iridaceae family. The aqueous extract of saffron petals contains 12% colourant. The colouring matter of saffron contains phenolic compounds, flavonoids and anthocyanins. Anthocyanidins (pelargonidin) is responsible for the colour in saffron petals. The oxidation of anthocyanidins produces flavonol ( Figure 11 ).

Molecular structure of pelargonidin (anthocyanidin) purple and kaempferol (flavonol) yellow.
1.12 Pomegranate rind ( Punica granatum )
Rind of pomegranate fruit waste is used as a natural dye. Pomegranate fruit is rich in natural tannins. The anar peel produces a yellow colour dye. This natural dye is used in dyeing of wool, silk and cotton fibre. The colouring molecule in pomegranate rind is flavogallol which is called granatonine. It exists in alkaloid form (N-methyl granatonine). The pomegranate rind is rich in tannin content; therefore, it is also used as tanning material ( Figure 12 ).

Chemical structure of granatonine.
1.13 Lac insect ( Laccifer Lacca Kerr)
It is a resinous protective secretion from the insect lac which work as a pest on a number of plants. Lac dye can be obtained by extracting stick lac (shellac) with water and sodium carbonate solution and precipitating with lime. Lac contains a water-soluble red dye. It produces scarlet to crimson red shade after dyeing. The lac dye is obtained from an insect named as coccus lacca. Resin which produced by insect is called stick lac. The lac dye contains laccaic acid A and B which are responsible for the colour of the dye. The amount of colouring matter (laccaic acid) is 0.5 to 0.75% on the weight of the resin ( Figures 13 and 14 ).

Chemical structure of laccaic acid A.

Chemical structure of laccaic acid B.
1.14 Cochineal ( Dactylopius coccus )
Cochineal is obtained from an insect. It produces beautiful crimson, scarlet and pink colour on cotton, wool and silk. After mordanting with alum, chromium, iron and copper; the colour from purple to grey are produced. Cochineal is a scale insect from which natural colourant carmine is derived. Carminic acid is extracted from female cochineal insects. The body of insect is 19–22% carminic acid ( Figure 15 ).

Chemical structure of carminic acid.

1.15 Mineral sources
Some kinds of mineral ores, red clay and ball clay can yield light colours along with mineral salts. But colour composition is not constant and depends on source.
2. Classification of natural dyes
2.1 by chemical constitution, 2.1.1 indigoid class.
Two important dyes in this class are indigo blue and Tyrian purple. It occurs as glucoside indicant in the plant. Another blue dye is woad having the same chemical class. The chemical structure which belongs to indigoid class is shown in Figure 16 .

Indigoid structure.
2.1.2 Anthraquinone class
Dyes that belong to this class are having anthraquinone structure and obtained from plant and insect. The red shade is specific to this class. Madder, lac, kermes and cochineal are some of the examples. The general chemical structure of this class is shown in Figure 17 .

Anthraquinoid structure.
2.1.3 Alpha naphthoquinone
The dyes are having alpha naphthoquinone structure such as 2-hydroxy 1-4-naphthoquinone. Hina, lawsone and juglone are examples of this class. The chemical structure of this class is shown in Figure 18 .

Naphthoquinone structure.
2.1.4 Flavones
The dyes are having yellow shade. The natural dye weld belongs to this category. Most of the dyes are derivatives of hydroxyl and methoxy substituted flavones or isoflavones. The chemical structure of this class of dye is shown in Figure 19 .

Flavones structure.
2.1.5 Carotenoids
The natural dyes saffron and annatto belong to this class. The dye structure of this class has long-chain conjugated double bonds. The chemical structure of this class is as shown in Figure 20 .

Carotenoid structure.
2.1.6 Dihydropyrans
The dyes which belong to this category are logwood and sappan wood. Logwood, a natural dye, produces dark black shade on silk, wool and cotton.
2.1.7 Anthocyanidins
The natural dye carajurin belongs to this category. The blue and orange shades are obtained from this class.
2.2 Chemistry of natural dyes
Different natural colourants contain different chromophoric and auxochromic groups. Depending on the presence of a particular group in the dye structure, the chemistry of the dyes can be explained in terms of their chromophoric groups. The different dye structures and chromophoric groups are as explained.
2.2.1 Quinoid-based structure
The quinoid-based dye structure can be overviewed as three chemical structures (a) benzoquinone, (b) naphthoquinone and (c) anthraquinone. The natural colourant carthamine belongs to benzoquinone group, and juglone and lawsone are having naphthoquinone structure. Alizarine dye possesses anthraquinone structure.
2.2.1.1 Benzoquinone dyes
In this dye structure the л electron system is small, and the dye contains another unsaturated group in conjugation to л electron system ( Figure 21 ). The red colourant carthamine is present in safflower (Natural Red 26). Safflower ( Carthamus tinctorius ) is a subtropical plant and cultivated in India, China, North and South America and Europe. In dyeing, the water-soluble yellow dye (safflor yellow) is extracted [ 18 ] by cold water, and then red safflorcamin is extracted by diluted sodium carbonate solution. After the neutralisation of extracted solution, it can be used in dyeing of wool, silk and cotton.

Structure of carthamine.
2.2.1.2 Naphthoquinone dyes
Lawsone and juglon natural dye belongs to this category. Lawsone is extracted from hina plant; the leaves also contain flavonoid colourants lutcolin. It is cultivated in countries like India, Africa and Australia. Naphthoquinone is present in glycosidic [ 19 , 20 ] form named as Hennosid A, B and C. The quantitative analysis of lawsone can be performed by high-performance liquid chromatography on reverse-phase C 18 column. Chloroform extracted hina leaves were analysed by high-performance thin layer chromatography ( Figures 22 and 23 ).

Lawsone (2-hydroxy, 1,4 naphthalene).

Juglone (5-hydroxy, 1,4 naphthoquinone).
2.2.1.2.1 Lawsone
Lawsone form 1:2 complex with Fe(II) and Mn (II) and useful in dyeing of wool and silk fibre. The better dye uptake is obtained at pH 3.0. Agarwal et al. [ 21 ] studied the effect of different mordants and different mordanting methods to get the different shades. Hina can be used for dyeing of cotton, polyester, polyamide and cellulose triacetate as the structure of dye molecules are similar to disperse dyes [ 22 , 23 , 24 ].
2.2.1.2.2 Juglone
Juglone is representative of natural dye with naphthoquinone structure. The dyestuff is extracted from different part of nut trees. Juglone is present as a glycoside form in trees and plants. Wool dyed with juglone are having good resistance with moths and insects. Mordanting treatment further enhances the fastness properties. Dyeing of textile materials with aqueous walnut extract yields brown shade. Wide range of textile fibre, e.g. wool, silk, nylon and polyester, can be dyed with juglone.
2.2.1.3 Antharaquinone
It possess biggest group of anthraquinone dyes. Rhubarb (CI Natural Yellow 23) is extracted from the root of the plant. The extracted dye contains emodin, chrysophenol, aloe emodin and pyscion ( Figure 24 ). Rhubarb extract is used in dyeing of wool fibre [ 25 ]. It produces yellow to orange shade after mordanting with alum. The mordanting treatment improves light fastness of dyed materials.

Different representative structurers of anthraquinone group-based dye molecules.
Natural dye alizarin, pseudo purpurin and purpurin ( Figure 25 ) belongs to plant of Rubiaceae family and has an anthraquinone structure [ 26 ]. The dye is obtained from the root of plant.

Structures of alizarin, pseudo purpurin and purpurin.
Madder (C.I Natural Red 8) natural dye produces red colourant; the cultivation of madder is done as a source material for red colour in Europe, Asia and Northern and Southern America. The dyestuff is extracted from the dried roots of the plant. The roots of the plant contain 2–3.0% of di- and tri-hydroxyl anthraquinone glucosides.
2.2.2 Carotenoids
Hydrocarbon carotenoid
Oxygen containing called xanthophylls

Structure of β-carotene.
Structural changes by hydrogenation, double bond migration, isomerization and chain lengthening and shortening resulted in many carotenoid structure. Carotenoids possess strong UV light resistance, and β carotene ( Figure 26 ) is a typical structure generally found in natural colourants.
2.2.2.1 Pyron dyes
Pyron dyes contain flavonoids and anthocyanins having structure as shown in Figures 27 and 28 . The pyron structure is bound to various sugars by glycosidic bonds [ 17 ]. Flavonoids are classified as flavonols, flavones, anthocyanidins, isoflavones, flavon-3,4-diols and coumarins. Yellow flavones and flavonols are used as vegetable dyes. The valuable and very popular flavonoid is yellow quercetin which possess several bio effect.

Structure of anthocyanins.

Structure of quercetin.
2.2.2.2 Anthocyanins
Anthocyanins are found in fruits and vegetables; some are grape wine, sweet and sour cherries, red cabbage, hibiscus and different varieties of oranges. There are more than 500 varieties of anthocyanins that produces red, pink, violet and orange colours. There are some important anthocyanins which are cyaniding, delphinidin, pelargonidin, malvidin, peonidin and petunidin. Many plants besides anthocyanins also contain quercetin and chlorophylls, and the resulted colour is a mixture of all these.
2.2.3 Dyes from lichens and mushrooms
Violet and purple colours were generally obtained from molluscs and shellfish, and they were source of dyestuff from ancient to the beginning of the Middle Ages. Royale purple and Tyrian purple were the name of the colour obtained originally from molluscs [ 27 ]. Lichens and mushrooms are source of natural dyes, and they produce violet and purple colours. Lichens are found in coastal areas and were easier to collect. The dyeing methods with lichens are easy; however, disadvantage associated with lichens is poor light fastness. Therefore, the dyeing of lichens are limited to cheap quality fabrics. Fungi are also used for dyeing of textiles. In America and India, red colour is obtained from fungus Echinodontium tinctorium . In Italy and France, fungi obtained from Polyporales were used to dye the wool.
The colourants in lichens and fungi are benzoquinone derivatives, especially terphenylquinone. Some of these species possess compounds such as Sarcodon , Phellodon , Hydnellum and Thelephora [ 28 , 29 ]. Orchil and litmus are the colourants that are responsible for the colour in lichens. The lichens’ colour are produced through pre-compounds of orchil and litmus by consecutive enzymatic, hydrolysation, decarboxylation and oxidation [ 30 ] reactions, respectively. Then some pre-compounds are lecanoric acid, atranorin and gyrophoric acid which take part in the formation of orchil and litmus as shown in Figure 29 .

Structures of different colourants occurring in fungi and lichens.
In the past, the extraction of colourants from lichens were performed by keeping the lichens in water with ammonia for several days. The reaction occurred through enzymatic hydrolysis in which non coloured compounds such as lecanoric acid are converted into orcinol by hydrolysis and decarboxylation. Orcinol after oxidation forms purple orceins or litmus. The colour of both litmus and orchil depend on the pH of the solution [ 30 ]. In acidic pH dyestuff forms red cation, and in basic pH, it forms bluish violet anion. The lichens which belong to species Parmelia , Xanthoria parietina , Ochrolechia tartarea and Lasallia pustulata are capable to produce yellowish, brownish and reddish brown colours in dyeing of wool with lichens [ 31 ]. The dyeing is done by boiling the wool with lichen solution either premordanted or without mordanted wool in presence of ammonia.
The mushrooms which belongs to species Sarcodon , Phellodon and Hydlnellum contain terphenylquinone compounds as a main colourants which produce blue colour in mushrooms. They are benzoquinone derivatives. The Cortinarius species mushrooms are richly coloured in brown, red, olive green and violet. They are anthraquinone derivatives.
2.2.4 Tannins
Tannins are polymeric polyphenols with typical aromatic ring structure with hydroxyl constituents and have relatively high molecular weight. In plants two different groups of tannins are found, (a) hydrolysable tannins and (b) proanthocyanidins (condensed tannin) [ 32 , 33 ]. Tannins are present in plant cell and are concentrated in epidermal tissues. Tannins are found in wood, leaves, buds, stems, florals and roots [ 34 ]. The hydrolysable tannins are concentrated in the roots of several plants. The plants are the source of different variety of tannins. The three major tannins (hydrolysable tannins) are grouped as gallotannins [ 35 ] or ellagitannins and which are gallic acid or ellagic acids. The most widespread gallotannins are pentagalloyl glucose. Ellagitannins are esters of hexahydroxydiphenic acids. Gallic acid and hexahydroxydiphenic acid occur together in some hydrolysable tannins [ 36 ].
Condensed tannins are polymers of 15-carbon polyhydroxyflavan-3-ol monomer units such as (−) epicatechin or (+) catechin. The complex chemical nature of tannins makes the biosynthesis and polymerisation a difficult task; however, there are some established pathways for bio synthesis. The precursor for biosynthesis of hydrolysable tannins is shikimic acid. The direct aromatization of 3-dehydroshikimic acid produces gallic acid, which upon esterification forms polyol.
The bio synthesis of condensed tannins occurs through two different ways (a) by phenylpropanoid and (b) by polyketide. The polyketide pathway takes malonyl moieties for aromatic ring formation in flavonoid biosynthesis. The phenylpropanoid pathway takes aromatic amino acid, L-phenylalanine, which is non-oxidatively deaminated to E-cinnamate by phenylalanine ammonia-lyase.
2.3 By hue or colour produced
Red: Colour index has 32 red natural dyes. The prominent members are maddar, manjistha, Brazil wood, Morinda , cochineal and lac dyes.
Blue: There are four natural blue dyes. Some prominent colours are indigo, Kumbh and flowers of Japanese Tsuykusa. Natural indigo blue is known from very ancient time to dye cotton and wool.
Yellow: There are 28 yellow natural dyes available which are used in dyeing of wool, silk and cotton. Prominent examples are barberry, tesu flowers, Kamala, turmeric and marigold.
Green: Plants that yield green natural colour are very rare; they are made by mixing yellow and blue primary colours. Woad and Indigo produce green colour.
Black and brown: There are six black natural dyes. Cutch is used to produce brown shade; for getting black shade lac, carbon and caramel are used.
Orange: Natural dyes which produce red and yellow colour are used to produce orange shade. Barbeny and annatto are the examples of orange colour.
2.4 Application based classification
Vat dyes: Indigo is a water-insoluble dye, and before application it is solubilised in water. The solubilisation of natural indigo is done with the help of sodium hydrosulphite and sodium hydroxide. After solubilisation, it is applied on cellulosic fibre, and after dyeing the development of colour is done by oxidation with hydrogen peroxide. Indigo dye is the representative of indigoid class of vat dyes
Direct dyes: The natural dyes which are water soluble and have a long and planar molecular structure and presence of conjugated (single and double bonds) bonds can be applied by direct dyeing method. The dye molecules may contain amino, hydroxyl and sulphonic groups. Turmeric, Harda, pomegranate rind and annatto can be applied by direct dyeing method. Common salt is used to get better exhaustion of dyes. The dyeing temperature is kept at 100°C
Acid dyes: The dye molecules possess sulphonic or carboxylic groups in their structure, which produce affinity for wool and silk fibre. The dyeing is done at acidic pH of 4.5–5.5. After dyeing the fastness improvement is done with tannic acid. The dyeing of wool and silk with saffron is done by acid dyeing method. The presence of common salt in dye bath produces levelling effect
Basic dyes: The dye molecules produce coloured cation after dissolution in the water at acidic pH. The dye molecules contain –NH 2 groups and react with –COOH groups of wool and silk. The dye bath pH is kept 4–5 by adding acetic acid
3. Extraction of natural dyes
The amount of natural dyes present in natural products are very less [ 11 , 37 ]. They need specific technique to remove dye from their original source. Here there are some methods which are suitable for extraction of natural dyes from their source materials [ 28 ]; the different extraction methods are as follows:
3.1 Aqueous extraction
In this method, the dye containing materials are broken into small pieces or powdered and then soaked in water overnight. It is boiled and filtered to remove non-dye materials. Sometimes trickling filters are also used to remove fine impurities. The disadvantages of this technique are that during boiling, some of the dye decompose. Therefore, those dyes which do not decompose at boiling temperature are suitable by this method. The molecules should be water soluble.
3.2 Acid and alkali extraction
Most of the natural dyes are glycosides; they can be extracted under acidic or alkaline conditions. Acidic hydrolysis method is used in extraction of tesu natural dye from tesu flower. Alkaline solution are suitable for those dyes which contain phenolic groups in their structure. Dyes from annatto seeds can be extracted by this method. The extraction of lac dye from lac insect and red dye from safflower is also done by this method.
3.3 Ultrasonic microwave extraction
Microwave and ultrasonic waves are helpful in extraction of natural dyes. This technique is having several advantages over aqueous extraction. In this technique less quantity of solvent (water) is required in extraction. The treatment is done at lower temperature and less time as compared to aqueous extraction. Ultrasonic and microwaves are sent in aqueous solution of natural dye, which accelerate the extraction process.
3.4 By fermentation
In the presence of bio enzymes the fermentation of natural colour bearing substances becomes faster, and the extraction of natural dyes takes place. Indigo extraction is the best example of fermentation method of extraction. Enzymes break glucoside indican into glucose and indoxyl by the indimulsin enzyme. Amatto natural dye extraction is also done by enzyme method. Cellulose, amylose and pectinase are having application in the natural dye extraction from the bark, stem and roots.
3.5 Solvent extraction
There is use of organic solvents such as acetone, petroleum, ether, chloroform and ethanol in the extraction of natural dyes. It is a very viable technique as compared to aqueous extraction. The yield of dye is good, and the quantity of water requirement is less. The extraction is done at lower temperature.
4. Characterisation of natural dyes
For successful commercial use of natural dyes, there is need of standardized dyeing technique for which characterisation of natural dyes is essential.
4.1 UV-visible spectroscopy
It is useful in characterising the colour in terms of the wavelength of maximum absorption and dominating hue. The application of UV-characterization is to identify the ability of dye molecules to absorb UV wavelength and fading characteristics of dyes. Some researchers [ 38 ] had done UV analysis of natural dyes. Mathur et al. [ 9 ] studied UV spectra of neem bark, and it has two absorption maxima at 275 and 374 nm. Beat sugar [ 39 ] shows their absorption bands at 220, 270 and 530 nm. Gulrajani et al. [ 40 ] studied the absorption bands of ratanjot and observed that at acidic pH, the absorption occurs at 520–525 nm, and in alkaline pH, it occur at 610–615 nm. Red sandal [ 41 ] wood shows strong absorption peak at 288 nm and maximum absorption at 504 and 474 nm in methanol solution at pH 10. Gomphrena globosa flower has peak at 533 nm. The dye does not have difference in peak value at pH 4 and 7 in visible region; however it shifted towards 554 nm [ 42 ]. Bhuyan et al. studied the dye absorption extracted from Mimusops elengi and Terminalia arjun and concluded that dye absorbed by the fibre varies from 21.94 to 27.46% and from 5.18 to 10.78%, respectively, depending on bath concentration [ 43 , 44 , 45 ]. He also reported absorption of colour extracted from the roots of Morinda angustifolia Roxb using benzene extract. The colour shows absorption at 446, 299, 291, 265.5 and 232 nm.
| Name of the dye | Wavelength of maximum absorption |
|---|---|
| Neem bark extraction | 275 and 374 nm |
| Beet sugar | 220, 280 and 530 nm |
| Ratanjot at acidic pH | 520 and 525 nm |
| Alkaline pH | 570, 610 and 615 nm |
| Red sandal wood | 288 nm |
The value of the wavelength of the maximum absorption for a particular dye depends on the chemical constitution of the dye molecules which is variable and depends on the growth environment of a particular natural dye. The characterisation of a particular dye is helpful in deciding the hue of the dye.
4.2 Chromatographic technique
Thin layer chromatography is used to identify different colour components in natural dyes. Koren [ 46 ] analysed insect dye, madder and indigoid. Guinot [ 47 ] analysed plants containing flavonoids colour compounds. Balakina [ 48 ] analysed quantitatively and qualitatively red dyes such as alizarin, purpurin and carminic acid by high-performance liquid chromatography. Mc Goven [ 49 ] et al. identified the dyes stripped from wool fibre by HPLC with C18 column. Szostek [ 50 ] et al. studied the retention of carminic acid, indigotin, corcetin, gambogic acid, alizarin, flavonoid, anthraquinone and purpurin. He studied examination of faded dyes through emission and absorption spectra by non destructive method. Cristea [ 51 ] et al. had reported quantitative analysis of weld by HPLC and informed that after 15 min. Extraction in methanol/water mixture, 0.448% luteolin, 0.357% luteolin 7-glucoside and 0.233% luteolin 3′7 diglucoside were obtained. Son et al. [ 52 ] reported analysis of longer dyeing time in indigo dyeing and their effect on structural change in dye molecules through HPLC analysis. The derivative spectroscopy and HPLC were used to analyse annatto dyestuff; the sample preparation involved extraction with acetone in the presence of HCl and removal of water by evaporation with ethanol. The residue was dissolved in chloroform and acetic acid mixture for derivatives spectroscopy or with acetone for HPLC.
5. Theory of dyeing
Natural dyes are very suitable for dyeing of protein fibres as compared to cellulosic fibres. Synthetic fibres which contain polar groups such as nylon, acrylic and viscose are also accessible to natural dyes. Natural dyes are thermo unstable and have poor chemical stability, which make the natural dyes unfit for dyeing at high temperature and pressure. The presence of hydrogen bond and Van der Waals force of attraction play important role in the fixation of natural dyes on the fibre. Natural dyes are having poor exhaustion value due to subdued affinity for fibre materials, so to increase the exhaustion of dyes, common salt/Glauber’s salt are added in the dye bath. The isotherm of the natural dyes sorption obeys Nernst isotherm [ 17 , 53 , 54 ].
Natural dyes are having poor affinity and substantivity [ 55 , 56 ] for cellulosic fibres such as cotton and viscose. The absence of reactive groups in fibres and dyes does not allow for bond formation, so they need mordanting treatment to fix the dye on fibre surface. Protein fibres are having bond-forming groups in fibre structure, and the presence of carboxylic groups in natural dyes provides opportunity for bonding and gets bonded with fibre and shows good fastness properties. Natural dyes are having smaller molecular size, and they are not having conjugated linear structure [ 57 ]. Therefore, natural dyes are having inferior exhaustion behaviour. Sometimes salt sodium chloride is also used to improve the dye exhaustion % ( Figure 30 ).

Sorption isotherm of dyeing of silk fabric (without mordant) with eucalyptus leaves extract at three different temperature 30, 60 and 90°C [ 17 ].
6. Application of natural dyes
Different researchers had proposed different methods of dyeing of natural and synthetic fibres with natural dyes. The dyeing of textile substrates depends on dyeing parameters which are fibre structure, temperature, time and pH of the dye bath and dye molecule characteristics. The fastness properties of dyes on textile substrates depend on bonding of dyes with fibre. Since natural dyes are lacking in the presence of active groups to make bonds with textile fibres, the fastness properties are not very good. The cellulosic fibres are difficult to dye with natural dyes as they have poor affinity and substantivity. The lack of bonding of natural dyes with cellulosic fibre requires mordanting treatment. Protein fibres have ionic groups and get bonded with natural dyes possessing ionic groups in dye structure.
The dyeing of proteins fibre can be done by exhaust method of dyeing. The dyeing process parameters in wool and silk dying is pH at 4.5–5.5 and dyeing temperature 80–90°C. The exhaustion % of dyes in dyeing is very poor. The longer liquor ratio may be preferred because of poor solubilities of natural dyes in water. Stainless steel-made dyeing machines are suitable in dyeing of wool and silk.
Since natural dyes are having poor affinity for cellulosic fibre and due to poor exhaustion, mordanting treatment [ 29 , 58 ] is done to fix the dyes on cellulosic fibre. The dyeing of cellulosic fibre can be done at temperature of 80–90°C.The exhaustion of dyes can be increased by adding exhausting agents, sodium chloride or Glauber’s salt in dye bath. Most of the dyeing is done at neutral pH. Dyeing of cotton with natural indigo is done at alkaline pH in the presence of sodium hydrosulphite in a container made of stainless steel. The copper container gives deeper shade in dyeing of cellulosic fibre. The mordanting treatment improves the washing fastness of dyed samples. There are three methods of mordanting [ 44 , 45 ].
6.1 Conventional method of dyeing
In the state of Maharashtra, Gujrat and Rajasthan [ 59 ], the people follow conventional method of dyeing of cotton fabric with natural dyes which may be explained with the following process sequences. The fabric is pretreated before dyeing to get the absorbency. The grey fabrics are given dunging treatment followed by washing. The bleaching treatment is given to make the fabric white, after that it is steamed and stepped into alkaline solution, and finally rinsing and washing treatment is given. After thorough pretreatment the fabric is soaked into solution of harda/myrobolan and dried. The dried fabric is premordanted with alum and subsequently dipped into natural dye solution at boiling temperature. After dyeing the fabric is given washing and rinsing treatment and dried in the sun light. Water is sprayed on the fabric to brighten the shade. The process is repeated 2 to 4 days. The dyeing method differs from place to place. Here are some examples:
6.1.1 In Bengal
The commonly used natural dyes are haldi, babul, madder, pomegranate rind and marigold [ 59 ]. In the dyeing of fabric with sappan wood, the fabric is dipped in aqueous extract of sappan wood with or without alum solution and boiled for 2–3 hours. In the dyeing of Indian madder, the madder is extracted either from the stem or root and boiled with water to extract the natural colourants. The pretreated fabric is boiled with dye extract solution. Mordanting treatment may be given either before dyeing or after dyeing with alum solution.
6.1.2 In Orissa
The sappan wood chips are boiled with alum and turmeric and after boiling it was cooled. In cooled solution of dye, the fabric materials are kept for 3–4 h. It is a premordanting process. At some places the cold solution of natural dye is taken with sufficient quantity of water, and the fabric is dipped in cold solution for 24 h and finally boiled for 2 h.
6.1.3 In Uttar Pradesh
The application of natural indigo on cotton fabric is done by two methods which are called Khari Mat and Mitha Mat.
6.1.4 Khari Mat
In Khari Mat’s process to dissolve natural indigo, 40 gallon of water is taken in an earthen vessel, and in that water there are addition of 2.0 lbs. indigo, 2.0 lbs. of lime, 2 lbs. of sajji mati and 1.0 ounce of gur (molasses). After 24 h of fermentation, the indigo dye became water soluble. The indigo dye solution is ready for dyeing. This technique is successful in hot weather.
6.1.5 Mitha Mat
In this technique, the solubilisation of natural indigo is done by taking 60 gallon of water; in that water there are addition of 4 lb. of lime, and after 1 day again 4 lb. of lime is added. After 4–5 days natural indigo dye became fully soluble. During application this mitha vat is added with old mitha vat with continuous string. The fabric is dyed in the dissolved indigo dye solution at temperature of 50–60°C.
6.2 Dyeing of cotton fabric with natural dyes
Dyeing time = 60–120 min. (depends on depth % of shade)
Temperature of dyeing = 70–100°C
M:L ratio of the bath = 1:20–1:30
Amount of dye in bath = 10–50% (on weight of the material)
Concentration of common salt = 5–20 g/l
pH of the dye bath = 10–11
After dyeing, soaping treatment is given to remove any residual/unreacted dyes and auxiliary chemicals from the surface of the fabric. An after treatment with natural dye, fixing agent may be desirable.
6.3 Dyeing of protein fibres
Wool and silk are protein fibre; both fibres have complex chemical structure and susceptible to alkali treatment. Alkaline pH of aqueous solution damage the fibre. At isoelectric pH of 5.0, the wool is neutral and the silk is slightly positive. The wool and silk can be dyed with natural dyes through premordanting or after mordanting. Mordanting is done with tannin-rich natural source chemical like harda or metal salt aluminium sulphate or ferrous sulphate.
The pH of the dye bath = 4–5
Temperature of dyeing = 80–90°C.
Time of dyeing = 50–60 min.
After dyeing, soaping treatment is given to remove any residual/unreacted dyes and auxiliary chemicals from the surface of the fabric. An after treatment with natural dye fixing agent may be desirable.
6.4 Dyeing of synthetic fibres
Different synthetic fibres like nylon, polyester and acrylic can be dyed with natural dyes like onion skin extract, babool bark extract and hina. The dyeing can be done either by padding (cold pad batch) method or exhaust method with or without mordanting. Dyeing is carried out at acidic pH. High-temperature high-pressure dyeing gives better results in terms of colour strength than other dyeing methods.
6.5 Fixation of natural dyes
Most controversial are lead salts and chromates (potassium, sodium, ammonium dichromate).
The salt SnCl 2 also works as mordant. It is water soluble, having reducing agent properties. It is toxic in nature.
Copper sulphate (CuSO 4 5H 2 O) and ferrous sulphate (FeSO 4 7 H 2 O) molecules are also used as a mordant. They are good chelating agents.
Tannins are poly phenolic compounds and able to form complexes with metals and bind with organic substances such as proteins, alkaloids and carbohydrates. The tannins are also called bio mordants. Tannins can be used either alone or in association with metal salts. The phenolic groups of tannins can form effective bonds with fibre and natural dye molecules.
6.5.1 Metallic mordants
Metal salts of aluminium, chromium, iron and copper are used as a mordants. The important mordants are potassium dichromate, ferrous sulphate, copper sulphate, stannous chloride and stannic chloride.
6.5.2 Tannins and tannic acid
Tannins are obtained from the excretions of bark and other parts, e.g. leaves and fruits of the plant. Extractions are either used directly or in concentrated form. Large number of tannin containing substances are employed as a mordant in textile fibre dyeing.
6.5.3 Oil mordants
Oil mordants are used in dyeing of madder. Oil mordants make a complex with alum used in mordanting treatment. Metal atom combined with carboxylic groups of oil and bound metal then makes bond with the dye molecules, and in this way, superior wash fastness can be achieved.
6.6 Mordanting process
Premordanting: In premordanting process, mordanting is done before dyeing; subsequently the fabric is dyed with natural dye in aqueous media. It is a two-bath process in which the first bath is used for mordanting of fabric and in the second bath, dyeing is done with natural dyes. Dyeing and mordanting are done at the same temperature of 60–70°C. the mordants are complexing agents, and if they are taken in the same bath, they may react to each other, and precipitation of dyes may occur. That deteriorate fastness properties of dyed fabrics
Metamordanting: In metamordanting treatment, the mordant chemicals are added with natural dye in the same dye bath; dyeing and mordanting take place simultaneously. The mordanting and dyeing temperature are 80–90°C
After mordanting: In after mordanting treatment [ 53 , 54 ], the dyeing of fabric is done first; after that in the same bath mordanting compounds are added. The temperature of chroming is 80–90°C. after chroming, the temperature is dropped to 60°C, and goods are run for 15 minutes after that liquor is drained
The application of natural dyes on cellulosic materials are done by the pad-dry-washing and pad-dry-steaming-washing method. High-temperature curing is not suggested as dye molecules are susceptible to decompose. Fibre and yarn dyeing can also be done with natural dyes similar to synthetic dye application.
7. Fastness properties of natural dyes
The quality parameters in dyeing is fastness properties. Several test methods are described to access the colour fastness. The fastness properties give idea about the quality of dyeing. In natural dyes, the fastness properties are strongly related to substrate type and mordant used for dyestuff fixation. Besides the dyestuff itself, there are many factors such as water, chemicals, temperature, humidity, light, pretreatments, after treatments, dyestuff distribution in fibre and fixation of dyestuff affect the fastness properties. In natural dyeing the colour and fastness of natural dyes need special attention for careful selection of materials and process. Natural dyes were in use up to end of the nineteenth century. At that time the dyeing with natural dyes were at peak with excellent fastness properties; however, after commercialization of synthetic dyes in the nineteenth century, the proficiency in natural dyeing started to decrease. The different fastness properties of dyes show the resistance of dyes towards different external environment in which fabric containing dyes are exposed. The fastness properties of dyes depend on the structure of dyes, exposure on the environment and fastness improvers and type of mordant used. There is need to explore some natural after treatment agents to improve the light and washing fastness.
7.1 Light fastness
Yellow dyes (old fustic and Persian berries), light fastness rating 1–2
Reds (cochineal with tin mordent, alizarin with alum mordant, lac with tin mordant), rating 3–4
Blue (indigo depends on mordants), rating 4–5 and 5–6
Black (logwood), rating 4–5
Effect of various additives on photo fading of carthamin in cellulose acetate film.
Critical examination of fading process of natural dyes to reproduce original colour of the fabric after fading.
The rate of photo fading effect is effectively suppressed in the presence of nickel hydroxyl-arylsulphonate. The addition of UV absorbers in bath has small effect in reducing photo fading effect.
7.2 Washing fastness
The washing fastness of natural dyes is poor to medium. The bonding of dye with fibre is very poor, and due to that dyes are not very fast with detergent solutions. Duff et al. [ 29 ] studied the effect of alkalinity of washing solution in washing of natural dyes dyed fabrics. The alkaline pH of the detergent solution changes the colour value in terms of the hue and value. Logwood and indigo are having good fastness value as compared to others. The mordanting treatment improves the washing fastness of dyes. Samanta et al. [ 68 ] reported some improvement in washing fastness by use of fixing agent.
7.3 Rubbing fastness
The rubbing fastness of most of the natural dyes are moderate to good. Samanta et al. [ 8 , 58 ] reported that jackfruit wood, manjistha, red sandal wood, babool and marigold having good rubbing fastness on jute and cotton fabric.
8. Advantages of natural dyes
8.1 uv-protective fabrics.
UV-protected fabrics are required to protect the skin and body of the human being from sunburns, tannings, premature skin burns and skin ageing. Researchers had done the work on to produce fabrics which had sun-protecting effect by the application of natural dyes in dyeing. Sarkar [ 69 ] evaluated ultraviolet protection factor (UPF) value of cotton fabric dyed with madder, indigo and cochineal with reference to fabric parameters. Grifani [ 70 , 71 ] studied the effect of natural dyes on cotton, flax, hemp and ramie and got good results. Metallic mordants [ 72 ] have potential to improve the UPF value of wool, silk and cotton. Orange peel extract natural dye applied on wool increased the UPF value of dyed wool fabric considerably.
8.2 Insect proof
Cellulosic materials and woollen are susceptible to moth and fungus attack in humid and warm conditions. Koto et al. [ 73 ] studied the effect of natural dyes on wool. The anthraquinone-based natural dyes cochineal, indigo and madder are able to produce insect proof and repellent fabric when used as a dyes in dyeing of wool.
9. Summary and conclusions
Natural dyes due to its unique character of natural origin are known as ecofriendly dyestuff; however the bonding of dye molecules with fibre-active sites are very poor, and they need some bridging chemicals to anchor the dye molecules with fibre, and mordanting agents are helpful in bridging the dye molecules with fibre. The synthetic mordanting agents are not very eco friendly, and some are toxic which depress the efficacy of natural dyes and sometime become matter of debate.
Natural dye does not have any shade card to match the samples or reproducing the shade. So there is need of collection of spectral data of natural dyes so that any shade can be reproduced.
There is need of awareness about natural dyes dyed fabric in people so that it can be popular in big way. and due to that demand and consumption of natural dyed fabric will increase.
Natural dyes are costly as compared to synthetic dyes. So some research work should be done to reduce the cost of production.
Big production houses, technical institutions and research houses should organised workshops and symposia to spread the advantages of natural dyes.
The government should promote the production of natural dyes by giving financial incentives to small manufactures of natural dyes.
There must be some very strong research and development work to improve the quality of natural dyes in terms of low cost, use of natural mordent and widespread applications.
Acknowledgments
I am very thankful to Prof. A.K. Samanta for inspiring me and giving very excellent suggestions for preparing this review paper. I am very thankful to the editor for his remarkable patience and monitoring.
- 1. Hill DJ. Is there a future for natural dyes? Review of Progress in Coloration and Related Topics. 1997; 27 :18
- 2. Dedhia EM. Natural dyes. Colourage. 1998; 45 (3):45
- 3. Chavan RB. Chemical Processing of Handloom Yarns and Fabric. Delhi: Department of Textile Technology, IIT; 1999. p. 6
- 4. Ghosh P, Samanta AK, Das D. Effect of selective pretreatments and different resin post-treatments on jute-viscose upholstery fabric. Indian Journal of Fibre and Textile Research. 1994; 19 :298
- 5. Gulrajani ML, Deepti G. Natural Dyes and their Application to Textiles. Delhi: Department of Textile Technology, IIT; 1999. p. 23
- 6. Senthil P, Umasankar P, Sujatha B. Ultrasonic dyeing of cotton fabric using with neem leaves. Indian Textile Journal. 2002; 112 (6):14
- 7. Saxena S, Iyer V, Shaikh AI, Shenai VA. Dyeing of cotton with lac dye. Colourage. 1997; 44 :23
- 8. Samanta AK, Preeti A, Siddhartha D. Dyeing of jute and cotton fabrics using Jackfruit wood extract: Part I—Effects of mordanting and dyeing process variables on colour yield and colour fastness properties. Indian Journal of Fibre and Textile Research. 2007; 32 :466
- 9. Mathur P, Metha A, Kanwar R, Bhandari CS. Use of neem bark as wool colourant—Optimum conditions of wool dyeing. Indian Journal of Fibre and Textile Research. 2003; 28 :95
- 10. Gulrajani ML, Gupta DB, Agarwal V, Jain V, Jain M. Some studies on natural yellow dyes. Indian Textile Journal. 1992; 102 (4):50
- 11. Mahale G, Sakshi, Sunanda RK. Silk dyed with Acalypha ( Acalypha wilkesiana ) and its fastness. Indian Journal of Fibre and Textile Research. 2003; 28 :86
- 12. Katti MR, Kaur R, Shrihari N. Dyeing of silk with mixture of natural dyes. Colourage. 1996; 43 (12):37
- 13. Lokhande HT, Vishnu A, Dorngade, Nayak SR. Application of natural dyes on polyester. American Dyestuff Reporter. 1998; 40
- 14. Rathi DR, Padhye RN. Studies on application of natural dyes on polyester. Colourage. 1994; 41 (12):25
- 15. Paul R, Jayesh M, Naik SR. Natural dyes: Classification, extraction and fastness properties. Textile Dyer & Printer. 1996; 29 (22):16
- 16. Teli MD, Paul R, Pardesi PD. Natural dyes, classification, chemistry and extraction methods. Colourage. 2000; 60 :43
- 17. Krizova H. Natural dyes. In: Kryštůfek M, Vik W, editors. Chapter 18: Textile Dyeing—Theory and Applications. 1st ed. TUL: Vysokoškolskýpodnik Liberec s.r.o., Studentská 2. Liberec. pp. 317-334
- 18. Oda H. Improvement of light fastness of natural dyes. Part 2: Effect of functional phenyl esters on the photo fading of carthamin in polymeric substrate. Coloration Technology. 2001; 117 (5):257
- 19. Badan BM, Burkinshans SM. Dyeing of wool and nylon 6.6 with henna and lawsone. Dyes and Pigments. 1993; 221 :15
- 20. Kawamura T, Hisata Y, Okuda K, Noro Y, Takeda, Tanka T. Quality evaluation of plant dye henna with glycosides. Natural Medicine. 2000; 54 (2):86
- 21. Agarwal A, Grag A, Gupta KC. Development of suitable dyeing process for dyeing of wool with natural dye henna ( Lawsonia inerma ). Colourage. 1992; 39 (10):43
- 22. Gupta DB, Gulrajani ML. Kinetic and thermodynamic studies on 2-hydroxy-1,4-naphthoquinone (lawsone). JSDC. 1994; 110 :112
- 23. Singh K, Karr V, Mehra S, Mahajan A. Solvent-assisted dyeing of polyester with henna. Colourage. 2006; 53 (10):60
- 24. Bechtold T, Turcann E, Ganglberger, Geisslers. Natural dyes in modern textile dyehouse. Journal of Cleaner Production. 2003; 11 :499
- 25. Rita M, Bechtold T. Natural colourants in dyeing. In: Thomas B, Mussak R, editors. Handbook of Natural Colourants. United Kingdom: John Wiley and Sons Ltd.; 2009. p. 316
- 26. Bechtold T. Natural colourants. In: Thomas B, Mussak R, editors. Handbook of Natural Colourants. United Kingdom: John Wiley and Sons Ltd.; 2009. p. 154
- 27. Cardon D. Natural Dyes, Tradition, Technology and Science. London: Archetype Publications; 2007
- 28. Casselman KD. Lichen Dyes and Dyeing: The New Source Book. Mineola, New York: Dover Publications; 2001
- 29. Grierson S, Duff DG, Sinclair RS. Natural dyes of the Scottish high lands. Textile History. 1985; 16 :23
- 30. Gill M, Steglich W. In: Herz W, Grisebach H, Kirby GW, Ch T, editors. Progress in the Chemistry of Organic Natural Products. Vol. 51. 1987. p. 125
- 31. Raisanen R. Dyes from lichens and mushrooms. In: Bechtold T, Mussak R, editors. Handbook of Natural Colourants. United Kingdom: John Wiley and Sons Ltd.; 2009. p. 2003
- 32. Porter CJ. Tannins. In: Harborne JB, editor. Methods in Plant Biochemistry. Vol. 1, 389. London: Academic Press; 1989
- 33. Seigler DS. Plant Secondary Metabolism. Bostan: Kluwer Academic Publications;
- 34. Waterman PG, Mole S. Analysis of Phenolic Plant Metabolites. Oxford: Blackwell Scientific Publications; 1994
- 35. Ayres MP, Class JP Jr, Macclean SF, Redman AM, Reichart PB. Diversity of structure and antiherbivore activity in condensed tannins. Ecology. 1989; 78 :1696
- 36. Julkunen R, Tiitto, Haggman H. Tannins and tanning agents. In: Bechtold T, Mussak R, editors. Handbook of Natural Colourants. United Kingdom: John Wiley and Sons Ltd.; 2009. p. 2003
- 37. Agarwal A, Paul S, Gupta KK. Effects of mordants on natural dyes. Indian Textile Journal. 1993; 1 :110
- 38. Erica J, Jiedemann, Yang Y. Fiber-safe extraction of red mordant dyes from hair fibers. Journal of the American Institute for Conservation. 1995; 34 (3):195
- 39. Mathur JP, Bhandari CS. Use of beet sugar as wool colourant. Indian Journal of Fibre and Textile Research. 2001; 26 :313
- 40. Gulrajani ML, Gupta D, Maulik SR. Bio polishing of tasar silk. Indian Journal of Fibre and Textile Research. 1994; 24 :294
- 41. Gulrajani ML, Bhaumiks S, Oppermann W, Handman G. Dyeing of red sandal wood on wool and nylon. Indian Journal of Fibre and Textile Research. 2003; 28 :221
- 42. Sankar R, Vankar PS. Dyeing wool with Gomphrena globosa flower. Colourage. 2005; 52 (4):35
- 43. Bhuyan R, Sai Kai CN, Das KK. Extraction and identification of colour components from the barks of Mimusops elengi and Terminalia arjuna and evaluation of their dyeing characteristics on wool. Indian Journal of Fibre and Textile Research. 2004; 29 (12):470
- 44. Samanta AK, Priti A. Application of natural dyes on textiles. Indian Journal of Fibre and Textile Research. 2009; 34 :384
- 45. Kharbade BV, Agarwal OP. Identification of natural red dyes in old Indian textiles. Journal of Chromatography. 1985; 347 :447
- 46. Zvic K. HPLC analysis of the natural scale insect, madder and indigoid dyes. JSDC. 1994; 110 (9):273
- 47. Guinot P, Roge A, Aradennec A, Garcia M, Dupont D, Lecoeur E, et al. Dyeing plants screening: An approach to combine past heritage and present development. Coloration Technology. 2006; 122 :93
- 48. Balankina GG, Vasiliev VG, Karpova EV. HPLC and molecular spectroscopic investigations of the red dye obtained from an ancient Pazyryk textile. Dyes and Pigments. 2006; 71 :54
- 49. Mc Goven PE, Lazar J, Michel RH. The analysis of indigoid dyes by mass spectrometry. JSDC. 1990; 106 (1):22
- 50. Szostek S, Grwrys JO, Surowiec I, Trojanowicz M. Investigation of natural dyes occurring in historical coptic textiles by high performance liquid chromatography with UV–Vis and mass spectrometric detection. Journal of Chromatography. 2003; A1012 :179
- 51. Cristea D, Bareau I, Vailarem G. Identification and quantitative HPLC analysis of the main flavonoids present in weld ( Reseda luteola L.). Dyes and Pigments. 2003; 57 :267
- 52. Son A-Y, Heng PJ, Kim KT. Dyes and Pigments. 2007; 61 (3):63
- 53. Patel KJ, Patel BH, Naik JA, Bhavana AM. Eco-friendly dyeing with Tulsi leave extract. Man Made Textiles. 2002; 45 (11)
- 54. Bhattacharya SD, Shah AK. Metal ion effect on dyeing of wool fabric with catechu. SDC. 2000; 116 (1):10
- 55. Maulik SR, Bhowmik KI. Studies on application of some vegetable dyes on cellulosic and lignocellulosic fibre. Man-Made Textiles in India. 2006; 49 (4):142
- 56. Siddiqui I, Gous MD, Khaleq MD. Indian Silk. 2006; 145 (4):17
- 57. Samanta AK, Priti A. Application of natural dyes on textiles. International Dyer. 2008; 193 (3):37
- 58. Samanta AK, Priti A, Siddthartha D. Studies on color interaction parameters and color fastness properties for dyeing of cotton fabrics with binary mixtures of jackfruit wood and other natural dyes. Journal of Natural Fibers. 2009; 6 :171
- 59. Mohanty BC, Chandramouli KV, Nail HD. Studies in Contemporary Textiles Crafts of Indian Natural Dyeing Process of India. Calico Museum of Textiles, Ahmedabad: H.N. Patel Publication’; 1987, I and II
- 60. Cook CC. Aftertreatments for improving the fastness of dyes on textile fibres. Review of Progress in Coloration and Related Topics. 1982; 12 :78
- 61. Pad Field P, Landi S. Natural dyes of the Scottish highlands. Studies in Conservation. 1966; 11 :161
- 62. Samanta AK, Konar A, Chakarborty S, Datta S. Dyeing of jute fabric with tesu extract: Part 1—Effects of different mordants and dyeing process variables. Indian Journal of Fibre and Textile Research. 2011; 36 (3):63
- 63. Lee JJ, Lee HH, Eom SI, Kim JP. UV absorber aftertreatment to improve light fastness of natural dyes on protein fibres. Coloration Technology. 2001; 117 :134
- 64. Mussak R, Bechtold T. Renewable resources for textile dyeing–technology, quality, and environmental aspects. In: Proceedings of the IFATCC International Congress. Barcelona; 2008
- 65. Samanta AK, Konar A, Chakarborty S, Datta S. Effect of different mordants, extraction conditions and dyeing process variables on colour interaction parameters and colour fastness properties in dyeing of jute fabric with Manjistha, a natural dye. Journal of the Institute of Engineering. 2010; 91 :7
- 66. Micheal MN, NAEl Z. Colourage. 2005; Annual 83
- 67. Hofenk JH, Graff D. Conservation restoration of church textiles and painted flags. In: 4th Int Restorer Seminar. Vol. 2. Hungary; 1983. p. 219
- 68. Samanta AK, Priti A, Siddhartha D. Application of single and mixtures of red sandalwood and other natural dyes for dyeing of jute fabric: studies on colour parameters/colour fastness and compatibility. Journal of the Textile Institute. 2009; 100 (7):565
- 69. Sarkar AK. An evaluation of UV protection imparted by cotton fabrics dyed with natural colourants. BMC Dermatology. 2004; 4 (1):15
- 70. Grifani D, Bacci L, Zipoli G, Carreral G, Baronti S, Sabatini F. Laboratory and outdoor assessment of UV protection offered by Flex and Hemp fabrics dyed with natural dyes. Photochemistry and Photobiology. 2002; 85 :313
- 71. Grifani D, Bacci L, Zipoli G, Sabatini F, Albanete I. The role of natural dyes in the UV. Protection of fabrics made of vegetable fibres. Dyes and Pigments. 2011; 91 (3):279
- 72. Gulrajani ML, Gupta D. Emerging techniques for functional finishing of textiles. Indian Journal of Fibre and Textile Research. 2011; 36 :388
- 73. Koto H, Hata T, Tsukada M. Potentialities of natural dyestuff as antifeedants against varied carpet beetle, Anthrenus verbasci . Japan Agricultural Research Quarterly. 2004; 38 (4):241
© 2019 The Author(s). Licensee IntechOpen. This chapter is distributed under the terms of the Creative Commons Attribution 3.0 License , which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Continue reading from the same book
Edited by Ashis Kumar Samanta and Nasser Sayed Awwad
Published: 30 September 2020
By Pubalina Samanta
2085 downloads
By Deepali Singhee
1176 downloads
By Anupama Mishra and Sapna Gautam
1463 downloads
IntechOpen Author/Editor? To get your discount, log in .
Discounts available on purchase of multiple copies. View rates
Local taxes (VAT) are calculated in later steps, if applicable.
Support: [email protected]
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List

Recent Advancements in Natural Plant Colorants Used for Hair Dye Applications: A Review
Hongyan cui.
1 Beijing Key Laboratory of Plant Resources Research and Development, College of Chemistry and Materials Engineering, Beijing Technology and Business University, Beijing 100048, China
Wenjing Xie
Zhongjie hua, zhiqin yuan.
2 State Key Laboratory of Chemical Resource Engineering, College of Chemistry, Beijing University of Chemical Technology, Beijing 100029, China
Associated Data
Not applicable.
There is an on-going demand in recent years for safer and “greener” hair coloring agents with the global consumer awareness of the adverse effects of synthetic hair dyes. The belief in sustainability and health benefits has focused the attention of the scientific community towards natural colorants that serve to replace their synthetic toxic counterparts. This review article encompasses the historical applications of a vast array of natural plant hair dyes and summarizes the possible coloration mechanisms (direct dyeing and mordant dyeing). Current information on phytochemicals (quinones, tannins, flavonoids, indigo, curcuminoids and carotenoids) used for hair dyeing are summarized, including their botanical sources, color chemistry and biological/toxicological activities. A particular focus is given on research into new natural hair dye sources along with eco-friendly, robust and cost-effective technologies for their processing and applications, such as the synthetic biology approach for colorant production, encapsulation techniques for stabilization and the development of inorganic nanocarriers. In addition, innovative in vitro approaches for the toxicological assessments of natural hair dye cosmetics are highlighted.
1. Introduction
Nowadays, with the growing global awareness of the adverse effects of synthetic hair dyes, the demand for safer and more environmentally friendly hair dyes is increasing. Hair dye products can be grouped into three categories according to wash fastness: temporary, semi-permanent and permanent hair dyes [ 1 ]. Permanent hair dyes refer to synthetic oxidative hair dyes, by which colors are produced in the hair cortex from small primary intermediates (e.g., p -phenylenediamine and p -aminophenol) and couplers (e.g., m -aminophenol, m -hydroxyphenol and resorcinol) through oxidation reactions in the presence of hydrogen peroxide as the oxidizing agent [ 2 ]. Permanent hair dyes represent the most widely used coloring matter in commercial hair dye cosmetics due to their strong dyeing performance, predictable colors and rich range of tones [ 3 ]. However, several studies have reported allergenicity [ 4 , 5 ], mutagenicity [ 6 , 7 ], carcinogenicity [ 8 ], and environmental toxicity [ 9 ] associated with the use of synthetic hair dye ingredients and the potential health risks have attracted widespread attention. By contrast, natural dyes are temporary or semi-permanent non-oxidative hair dyes that can be adsorbed onto the cuticle and some parts of the cortex of the hair shaft to produce color. Natural dyes derived from various parts of plants (e.g., fruits, flowers, leaves, seeds and roots) are generally regarded as low-irritating, less allergenic, sustainable and eco-friendly green products with additional health benefits (e.g., antioxidant, anti-inflammatory and antimicrobial properties) [ 10 ].
Natural dyes have been used since ancient times, when they were used not only for hair coloration, but also for medicinal, decoration and religious purposes [ 11 , 12 ]. In the early days, hair dyes were obtained from metallic compounds, plant extracts, dried plants or their mixtures [ 13 ]. Before the invention of first synthetic aniline dye, mauve, in 1856, different plant extracts and herbal preparations such as mullein, birch bark, turmeric, and saffron have been used for hair dyeing. The early record of natural hair dyeing dates back to ancient Egyptian times when Rameses II reinforced his red hair color using henna [ 13 ]. The ancient Greeks used to bleach their hairs using a rinse of potassium lye solution followed by rubbing with a type of ointment made of yellow flower petals and pollen [ 14 ]. The Romans dyed their hair black by using walnut extracts [ 15 ]. Today, the renascence of natural botanical ingredients in cosmetics and health care products has led to research work into the phytochemistry and coloring potential of these traditionally used hair dye plants. Compounds including quinones, tannins, flavonoids, indigo, curcuminoids and carotenoids were identified as the dominant naturally-occurring hair coloring matters and some plants accumulating these phytochemicals, such as Lawsonia inermis (henna) [ 16 , 17 , 18 ], Juglans regia (walnut) [ 11 , 12 , 16 ], Curcuma longa (turmeric) [ 19 , 20 ], Haematoxylon campechianum (logwood) [ 16 , 19 , 21 ] were extensively investigated. Natural dyes used in commercial cosmetics are mainly extracted from plants by solvent extraction [ 19 ], ultrasonic assisted extraction [ 22 ], microwave assisted extraction [ 10 ], supercritical fluid extraction [ 23 ], and enzyme-assisted extraction [ 24 ] etc.
This review focuses on complete phytochemical information of plant-derived hair dyes including historical uses, coloring mechanisms, color chemistry, and toxicological aspects. The development of state-of-the-art techniques to produce or stabilize natural plant dyes for advanced hair dye applications as well as hair dye safety assessments are also highlighted. The purpose of this review is to facilitate further exploration of natural colorants as healthy and sustainable hair dye products.
2. Hair Coloring Mechanisms
For most natural plant hair dyes, there are two mechanisms for hair coloration: direct dyeing and mordant dyeing. Briefly, the hair dyeing process can be divided into two steps: (i) Diffusion of dye molecules from dye bath to the keratinous hair fiber; (ii) Formation of chemical bonds (hydrogen, ionic, and covalent bonds) between the carboxyl or hydroxyl groups present in the dye molecules and amino/sulfhydryl groups in hair keratin, with or without the aid of auxiliary mordanting agents [ 25 ].
Diffusion is a three-stage process [ 1 ]: (i) the first stage is the transport of dye molecules to the fiber/water interface by a combination of aqueous diffusion and agitation; (ii) in the second stage, dyestuffs are adsorbed onto the outer layer of hair cuticle; (iii) final stage is the diffusion of dye molecules of low molecular weight into inner hair structures (cuticle and cortex) and can be characterized by the change to the cell membrane complex (CMC) present in the hair cuticle. The CMC is a continuous phase of intercellular matters that binds the cuticle and cortical cells together [ 26 ]. Studies have shown that penetration though CMC is the main transport pathway for dye substances to reach the hair cortex [ 27 , 28 ]. Less ionized small molecules are more likely to penetrate through and spread over the lipid bilayer of CMC [ 29 ]. Besides, the condition of hair fibers also affects the absorption and diffusion of external dye substances. For example, the use of hydrogen peroxide in hair dye formulation can destroy the disulfide bonds of hair keratin, causing CMC breakage and damages to the cuticle and cortex components, resulting in swelling loose hair fibers and lifted cuticles, thus facilitating deeper penetration and stronger bonding of dyestuffs to the hair exterior shaft [ 12 , 19 ].
Direct dyeing, as a non-oxidative hair coloring process, is a direct formation of a dye-complex or bonding between the dyestuff and hair fiber. The color strength of directly dyed hairs depends on the affinity of dye molecules to the hair fiber surface. Generally, dyestuffs of low molecular weight (the critical sizes are 1.2–1.3 nm for anionic dyes, 1.4 nm for cationic dyes, and 0.95 nm for nonionic dyes [ 30 ]) can easily penetrate into the cuticle layer of hair fiber. Dyes of high molecular weight cannot penetrate the cuticles but may be adsorbed onto hair fiber via various types of forces, i.e., van der Waals, electrostatic, and hydrogen bonding [ 31 ]. Among the direct dyes, natural dyes extracted from henna leaves and walnut husks are popular representatives. Take henna for example, at pH 4.5–6.0, the reduced form of lawsone (2-hydroxy-1,4-naphthoquinone), its main colorant, reacts with the protonated amino groups present in hair keratin fibers ( Figure 1 a) [ 32 ]. Additionally, SEM observation finds that henna dyestuffs might be capable of recovering the cuticle damage and providing a smooth moisture-rich appearance on the dyed hair cuticles [ 33 ].

Possible interactions between hair fibers and lawsone ( a ); Possible interactions between hair fiber, polyphenol dye and mordanting iron (II) ions, where R=H, CH 3 , OH ( b ). Reprinted with permission from Ref. [ 36 ]. Copyright 2021, Multidisciplinary Digital Publishing Institute.
Mordant dyeing refers to the formation of charge-transfer complex between the dye and a mordanting agent on the dyed hair [ 34 ]. Mordants are substances that can fix dyes on hair fibers through interactions with dye molecules and hair fibers for improved color fastness [ 35 ]. Common mordants are metal salts, such as iron (II) sulfate, copper (II) sulfate, and alum, and act as a link between the dye and the hair fiber. Dative covalent bonding is the probable mechanism in binding metallic mordants to dye molecules with the oxygen-containing groups playing a key role [ 19 ]. Figure 1 b shows dative covalent bonding between an iron (II) ion and a polyphenol dye that bonds to the hair fiber by hydrogen bonding [ 36 ]. Mordant dyeing can be conducted by pre-, meta- or post-mordanting methods. The choice of mordants and mordanting methods has significant influence on the hue of the dyed colors and fastness properties [ 37 ]. For natural dyes, it is difficult to predict an optimal mordanting procedure because the results are highly dependent on the dye plant and mordant type. On the other hand, the treatment of transition metal mordants may result in the accumulation of iron and copper in human hairs, which has been reported to cause photooxidative damage of dyed hairs through Fenton chemistry [ 38 ]. In this regard, the development of bio-mordants, especially tannins and metal-rich plants, as effective alternatives to metallic mordants warrants further investigation [ 39 ]. For example, Aloe vera extract was reported as a bio-mordant to improve the hair dyeing properties [ 40 ]. Tannin-rich plant extracts from Punica granatum (pomegranate) peels, Eucalyptus maculata (eucalyptus), Rhus coriaria (sumac) and Emblica officinalis (amla) are bio-mordants widely used in the textile industry as alternatives to the metallic mordants but are rarely reported for hair dye applications [ 41 ].
3. Phytochemicals Used for Hair Dyeing
Many organic compounds have been identified as the principal coloring matters in hair dye plants and investigated for dyeing performance under experimental conditions. Natural colorants can be classified based on dye source, application method and chemical structure. Here we describe the structure-based classification since the chemical structure uniquely identifies dye molecules with specific properties ( Table 1 ).
Summary of natural plant colorants used for hair dye applications.
| Category | Colorant | Botanical Origin | Extraction Process | Dye Bath | Dyeingsubstrate | Mordant | Dyeing Process | Dyed Color | Color Fastness | Refs. |
|---|---|---|---|---|---|---|---|---|---|---|
| Quinones | lawsone | leaves of L. | ultrasound reflux extraction (sodium hydroxide solution 0.25 mol/L, solid liquid ratio 1:55, 140 min, 100 °C) | dye gels (xanthan gum, 1,2-propanediol) | gray hair | iron (II) sulfate | post-mordanting dyeing | reddish brown | / | [ ] |
| lawsone | leaves of L. | reflux extraction (distilled water, solid liquid ratio 1:6, 120 min, 100 °C) | emulsion | gray hair | iron (II) sulfate | post-mordanting dyeing | blank | resistant to 20 shampoo washes | [ ] | |
| lawsone | leaves of L. | ultrasound reflux extraction (sodium hydroxide 0.25 mol/L, solid liquid ratio 1:55, 140 min, 100 °C) | dye gels (xanthan gum, 1,2-propanediol) | yak hair | iron (II) sulfate | post-mordanting dyeing | reddish brown | resistant to 15 shampoo washes | [ ] | |
| lawsone | leaves of L. | cold maceration extraction (water, 48 h) | dye gels (carbopol-934, glycerin, sodium hydroxide solution, methyl paraben) | hair | / | direct dyeing | brownish | resistant to 5 shampoo washes | [ ] | |
| lawsone | leaves of L. | / | paste | goat hair | / | direct dyeing | reddish brown | / | [ ] | |
| juglone | husk of L. | solvent extraction with microwave-assisted (acetone–water 70% ( / ), 60 s, 180 w) and ultrasound-assisted (20 min, 90 w, 37 kHz) | solution | bleached hair | iron (II) sulfate and gel | meta-mordanting dyeing | dark brown | resistant to 15 shampoo washes | [ ] | |
| juglone | husk of L. | ultrasound reflux extraction (ethanol 50%, solid liquid ratio 1:25, 120 min, 60 °C) | dye gels (xanthan gum, 1,2-propanediol) | gray hair | iron (II) sulfate | post-mordanting dyeing | brown | / | [ ] | |
| juglone | husk of L. | Solvent extraction (dichloromethane, 60 min, 3 times) | solution | yak hair | / | direct dyeing | red brown | / | [ ] | |
| shikonin | roots of Sieb. et Zucc. | solvent extraction (ethanol and 3% acetic acid, 30 h) | solution | bleached hair | / | direct dyeing | light brown grey | resistant to 8 shampoo washes | [ ] | |
| alizarin | roots of L. | / | solution | yak hair | / | direct dyeing | red | / | [ ] | |
| benzoquinone | shoots of Rehd. | solvent extraction (phosphate buffer, pH 6.0) | solution | yak hair | / | direct dyeing | brown | / | [ , ] | |
| Tannins | gallotannin | parasitic aphids of Mill. | ultrasound reflux extraction (80% ethanol, solid liquid ratio 1:25, 160 min, 60 °C) | dye gels (Xanthan gum, 1,2-propanediol) | gray hair | iron (II) sulfate | post-mordanting | black | resistant to 13 shampoo washes | [ ] |
| gallotannin | parasitic aphids of Mill. | ultrasound reflux extraction (80% ethanol, solid liquid ratio 1:25, 160 min, 60 °C) | dye gels (Xanthan gum, 1,2-propanediol) | gray hair | iron (II) sulfate | post-mordanting | black | / | [ ] | |
| catechin | matcha tea | / | solution | unpigmented hair | iron (II) lactate | post-mordanting | dove grey | resistant to 12 shampoo washes | [ ] | |
| Flavonoids | cyanidin-3-o-rutinoside | fruit skins of L. | aqueous extraction (acidified water, 2 h) | paste | bleached hair | / | direct dyeing | blue | resistant to 12 shampoo washes | [ ] |
| cyanidin-3-glucoside | fruit of L. | solvent extraction (methanol with aq. hydrochloric acid 1%, 30 min) | solution | yak hair | iron (II) oxalate | meta-mordanting | blue | / | [ ] | |
| cyanidin-3-glucoside | beans of | solvent extraction (hydrochloric ethanol, 4 °C, 24 h) | dye gels | bleached hair | / | direct dyeing | brownish red | resistant to 4 shampoo washes | [ ] | |
| cyanidin-3-glucoside | fruit of Var. Paniala | solvent extraction (hydrochloric ethanol, 24 h) | spray | bleached hair | / | direct dyeing | red | resistant to 5 shampoo washes | [ ] | |
| cyanidin-3-glucoside | corn cobs of | aqueous extraction (80 ± 2 °C, 15 min) | solution | grey hair | / | direct dyeing | blue | / | [ ] | |
| hematoxylin | heartwood of | ultrasound reflux extraction (ethanol 80%, solid liquid ratio1:25, 160 min, 60 °C) | dye gels (Xanthan gum, 1,2-propanediol) | gray hair | iron (II) sulfate | post-mordanting | brown red | / | [ ] | |
| hematoxylin | heartwood of | aqueous extraction (pH 9,25 °C, 1:4 ( / )) | solution | bleached hair | iron (II) sulfate | meta-mordanting | reddish-brown | resistant to 15 shampoo washes | [ ] | |
| hematoxylin | heartwood of | aqueous extraction (95 °C, 50 min, 4 times) | solution | bleached hair | / | direct dyeing | light brown red | resistant to 8 shampoo washes | [ ] | |
| quercetin | leaves of Linn. F. | solvent extraction (ethanol-water, 70 °C, 3 h, 2 cycles) | solution | bleached hair | / | direct dyeing | brown | / | [ ] | |
| acacetin | bark of (Linn.) Willd. | reflux extraction with (distilled water, solid liquid ratio 1:6, 120 min, 100 °C) | solution | gray hair | direct dyeing | / | [ ] | |||
| Indigo | indigo | leaves of Isatis indigotica Fort., Polygonum tinctorium Ait. | / | paste | gray hair | direct dyeing | dark brown | Resistant to 6 shampoo washes | [ ] | |
| indigo carmine | / | / | solution | blonde hair | / | direct dyeing | blue | / | [ ] | |
| Curcuminoids | curcumin | root of Linn. | aqueous extraction (4 °C, pH 5, solid liquid ratio 1:4) | solution | bleached hair | iron (II) sulfate | meta-mordanting | orangish brown | resistant to 15 shampoo washes | [ ] |
| curcumin | root of Linn. | / | solution | yak hair | / | direct dyeing | yellow | / | [ , ] | |
| Carotenoids | zeaxanthin | aerial parts of L. | aqueous extraction (100 °C, pH 9, solid liquid ratio 1:4) | solution | bleached hair | iron (II) sulfate | meta-mordanting | brown | resistant to 15 shampoo washes | [ ] |
| peridinin | fruit of Roxb. | aqueous extraction (25 °C, pH 7, solid liquid ratio 1:4) | solution | bleached hair | iron (II) sulfate | meta-mordanting | brown | resistant to 15 shampoo washes | [ ] | |
| lutein | flower of Linn. | aqueous extraction (60 min, 100 °C) | solution | grey hair | gel | meta-mordanting | black | resistant to 5 shampoo washes | [ ] |
3.1. Quinones
Quinones are colored compounds with a basic benzoquinone chromophore consisting of two carbonyl groups. The three main classes of quinones are naphthoquinones, anthraquinones and benzoquinones [ 53 ]. Among which, naphthoquinones, widely distributed in plants and microorganisms, are the most frequently encountered quinone hair dyes. In plants, these compounds usually exist in the free form with several isomers, among which, 1,4-naphthoquinones are the most stable [ 54 ]. The light absorbance of quinone dyes depends on their skeleton structure and is affected by the presence of various substituents. The introduction of substituents, especially free or methylated hydroxyl groups, may induce a red shift of the absorption maxima. Some substituents, such as amino or substituted amino groups, may have significant influences on the color properties of quinone dyestuffs [ 55 ]. Representative naphthoquinones for hair dyeing purposes are lawsone, juglone, and shikonin ( Figure 2 ).

Molecular structures of lawsone, juglone, shikonin, 1,4-benzoquinone, alizarin and the color changes at acid–base conditions.
Lawsone (2-hydroxy-1,4-naphthoquinone) is the main coloring component of henna leaves, also known as CI Natural Orange 6, which acts as a direct or mordant dye for hair fiber to give a red-orange color [ 17 ]. In Asia and North Africa, the leaves of henna are widely used for hair and skin coloration. In a traditional manner, henna paste (powdered henna leaves mixed with warm water) is directly applied onto the hair where lawsone can gradually diffuse from the paste into the hair shaft to bind with keratin. There are many natural dye formulations based on henna on the market, usually in the powder form. For example, a gel formula consisting of powdered henna, tea, and hibiscus leaves can dye bleached hair brown [ 18 ]. Besides direct dyeing, henna can also be used with various mordants. Ali et al. [ 20 ] reported that the plant hair dyes prepared from henna, curcumin, and Tagetes erecta extracts can be used as both direct dye to color gray hair brown and as mordant dye to obtain black color when combined with iron (II) sulfate. Additionally, lawsone has a very low allergic potential. In most cases, allergic reactions are not caused by the henna itself, but by the synthetic coloring additives that are added to henna mixtures [ 56 ]. It is worth noting that, when pure henna (usually considered as a weak sensitizer) is used in combination with p -phenylenediamine, the risks for inducing sensitization and broad immune responses (including hypersensitivity) increase significantly [ 57 ]. The European Union Scientific Committee on Consumer Safety (SCCS) concluded that henna was slightly and transiently irritating to the eye but it can be safely used as a hair dye when the content of lawsone is less than 1.4% [ 58 ]. Besides lawsone, there are flavonoids, tannins, phenolic compounds, alkaloids and other active components in henna. Researchers have reported that henna extract has anti-inflammatory, anticonvulsant [ 59 ], antioxidant, immunomodulatory [ 60 ], wound healing [ 61 , 62 ], and other pharmacological activities.
Juglone (5-hydroxy-1,4-naphthoquinone) is an isomer of lawsone and can be obtained from the leaves, roots, shells, and barks of walnut plants. Juglone and juglone-containing walnut green husk extracts were used in skin coloring preparations [ 63 ], hair dyes [ 11 , 12 , 16 ] and antimicrobial agents [ 64 ]. As a hair dye, juglone gives hair a brownish color when mordanted with iron (II) ions. Beiki et al. reported the use of walnut husk extract to dye bleached hair with good washability and antibacterial activity [ 12 ]. A variety of in vitro test methods were used to evaluate the safety of plant hair dyes based on walnut husk extract, categorizing it as non-irritant to the skin but slightly irritating to the eye [ 16 ]. Additionally, some studies reported dose-dependent cytotoxicity of juglone in human fibroblasts and keratinocytes [ 65 , 66 ]. These findings indicate that cosmetic preparations containing juglone should be used with care.
Likewise, shikonin has also been used as a direct hair dye for brown color since ancient times. Shikonin ((R)-5,8-dihydroxy-2-(1-hydroxy-4-methylpent-3-en-1-yl)naphthalene-1,4-dione) is a dominant component in the roots of Lithospermum erythrorhizon (gromwell root), a perennial herbaceous plant native to China, Japan, and Korea [ 67 , 68 ]. Wang et al. reported that the dyeing performance of gromwell root extracts on bleached hairs with the most pronounced color change observed under acidic conditions [ 21 ]. Besides, shikonin is believed to endow fibers with antibacterial and anti-ultraviolet properties [ 69 ].
Anthraquinones are compounds with a central 1,4-diketo-cyclohexa-2,5-diene (quinone) structure connected to peripheral phenyl rings [ 70 ]. Anthraquinone dyes are typical donor/acceptor types and the substituent effects of which can provide a wide range of colors. A notable structural feature is the hydrogen bonding between α-substituents and the carbonyl group, which enhances light fastness [ 34 ]. Anthraquinones are found in various parts of plants (flowers, leaves, fruits, roots, and rhizomes). Rheum officinale (rhubarb), Hamamelis mollis (witch hazel), Aloe vera (aloe), and Rhamnus davurica (buckthorn) are known genera rich in these compounds [ 71 ]. In plants, anthraquinones usually exist in the form of glycosides and rarely in the free form. Rubia cordifolia (madder) is a common source of the anthraquinone dye alizarin for dyeing purposes [ 53 ]. The structure and color of alizarin are pH-dependent ( Figure 2 ). When the pH value is less than 7, alizarin is in its unionized form with both phenolic hydroxyl groups closed and appears yellow with the absorbance maximum at 430 nm [ 72 , 73 ]. At weak alkaline pH 8, alizarin is partially deprotonated only with its β-OH and appears red with two absorption peaks at 430 nm and 530 nm [ 73 ]. When pH increases (8 < pH < 13), both phenolic hydroxyl groups lost hydrogen, and the absorption peak of alizarin moves rapidly to 530 nm, showing a purple color [ 74 ]. Boga et al. investigated hair dyeing with alizarin at pH 8 (using yak hair as a model) and found it can rapidly turn white yak hair into reddish color [ 11 ].
Benzoquinone is the basic subunit of quinone compounds. 1,4-benzoquinone is a low molecular weight benzoquinone dye extracted from young shoots of the Pyrus lindleyi (pear) and can be used for direct dyeing [ 53 ]. It was reported that 1,4-benzoquinone can give yak hairs a brown color at pH 2.7 or pH 8 in a concentration dependent manner, and the color intensity does not seem to be strongly affected by pH [ 11 ].
Overall, quinones are a class of naturally abundant colorants with great potential to obtain a broad spectrum of colors on hair, ranging from deep purple to orange and yellow. Currently, the application of quinone colorants in commercial hair cosmetics is limited by poor solubility, strong odor, and photodegradation susceptibility.
3.2. Tannins
Tannins are a large group of polyphenolic molecules having several phenolic hydroxyl groups and other groups such as carbonyls to form strong complexes with various macromolecules [ 75 ] ( Figure 3 ). Tannins are broadly distributed in the plant kingdom and are the most abundant secondary metabolites [ 76 ]. They are generally classified into two types: hydrolysable tannins and condensed tannins [ 77 ]. Hydrolysable tannins contain glucose or polyhydric alcohols esterified with gallic acid (e.g., gallotannins) or hexahydroxydiphenic acid (e.g., ellagitannins) while condensed tannins consist of flavolans or polymeric parts of flavan-3-ols (catechins) and/or flavan 3:4-diols (leucoanthocyanidins) [ 75 ].

Chemical structures of tannins (gallotannin, ellagic acid, gallic acid and catechin) with a proposed mordanting mechanism when complexed with iron (II) ions.
Tannins are typical mordant dyes. One famous example used in history is the iron-gallink (tannin-iron coordination compounds) [ 78 ]. When hydrolysable tannins (pale yellow color) are complexed with iron (II) ions, a strong bluish-black color is produced and the coordination complex formed between tannin, mordant and hair fiber can enhance the color fastness [ 16 ]. Both gallotannins and gallic acids occur abundantly (tannin content over 65%) in Galla Chinensis (Chinese gallnut) and the gallnut extract was widely used as a black hair dye with iron (II) sulfate in eastern Asia [ 44 ]. Sargsyan et al. [ 36 ] studied the hair dyeing mechanism of matcha, which contains another type of hydrolysable tannin, catechin, and found a similar iron-gall complex formed between the mordanting iron (II) ions and the hydroxy groups of flavanol and keratinous hair fiber [ 79 ]. Additionally, the selection of metal ions and mordanting methods has shown to have significant effects on the color intensity and fastness of tannin dyeing [ 80 ].
On the other hand, tannins can also act as a bio-mordant to enhance the dyeing fastness of many plant dyes. Jahangiri et al. [ 81 ] investigated the effects of tannin-based bio-mordants and metallic mordant (alum) on wool dyed with madder root extract. The results showed that fibers pretreated with tannin produced very similar color and washing fastness with those pre-mordanted with alum (ΔE < 1). This was suggested to result from the formation of ionic complexes among protein fibers and organic molecules with ionizable groups at appropriate pHs [ 82 ]. In addition, due to the existence of abundant −OH groups, the application of tannins as mordants may also enhance color fastness by forming additional hydrogen bonds with both colorants and protein fibers [ 83 ].
3.3. Flavonoids
Flavonoids are formed in plants from the aromatic amino acids, i.e., phenylalanine and tyrosine, and generally occur as glycosylated derivatives. The core structure of flavonoid is flavan nucleus, which consists of 15 carbon atoms arranged in three rings [ 84 ]. Flavonoids known for hair dye applications include anthocyanins, hematoxylin, quercetin, acacetin, etc. ( Figure 4 ). Anthocyanins are the largest group of polyphenols in the plant kingdom. They are responsible for the pink, red, purple, violet and blue colors of many fruits, vegetables and flowers [ 19 ]. Several studies have demonstrated the successful use of plants containing anthocyanins for hair coloring, including Ribes nigrum (blackcurrant) [ 45 ], Morus nigra (mulberry) [ 11 ], Phaseolus mungo (bleak bean) [ 46 ] and Cleistocalyx nervosum var. paniala [ 47 ]. Their colors are determined by the number of hydroxyl groups and degree of methylation as well as the number and position of sugar moieties (glycosides) and attached aliphatic or aromatic acids [ 45 ]. Six common derivatives of anthocyanins are presented in Figure 4 . Their colors are highly influenced by the environmental acidity/alkalinity. At pH < 3, the flavan nucleus exists mainly as flavylium cation (AH + ) showing a red color [ 45 ]. When pH increases, AH + undergoes a rapid deprotonation to form a purple-colored quinonoidal base (A); and when pH > 7.5, an anionic quinonoidal base (A − ) is formed with a blue color [ 85 ]. Besides, opening of the anthocyanin ring may result in the formation of a yellow-colored E-chalcone [ 86 ]. Cyanidin-3-glucoside was successfully applied to hair coloring as a source of red colorants and can change into blue color when complexed with iron (II) oxalate [ 45 , 47 ]. Therefore, anthocyanin dyes can be used for both direct dyeing and mordant dyeing. However, anthocyanins are unstable in aqueous formula whereas the addition of vitamin E acetate at 0.04% can enhance this natural colorant’s stability [ 45 ]. Anthocyanins-based preparations have shown to be efficient semi-permanent dyestuffs for hair with the dyed colors durable up to 5 wash cycles [ 46 ].

Chemical structures of flavonoids (anthocyanins, hematoxylin, quercetin and acacetin) and the effect of pH on anthocyanin structure and resultant color.
Hematoxylin, (6aR,11bS)-7,11b-dihydroindeno[2,1-c]chromene-3,4,6a,9,10(6H)-pentaol, derived from Haematoxylon campechianum (logwood) [ 87 ], can act directly or as mordant dye for hair fiber to give a reddish-brown color [ 16 ]. Wang et al. reported the use of logwood extract to dye bleached hair and the dyeing effect was optimal at pH 7 [ 21 ]. Thermodynamic and kinetic studies have shown that the adsorption of hematoxylin on hair is a spontaneous and exothermic process [ 87 ]. In vitro toxicological tests demonstrated that logwood extract is a safe non-irritant hair dye ingredient [ 16 ].
Quercetin (2-(3,4-dihydroxyphenyl)-3,5,7-trihydroxy-4-Hchromen-4-one)) is a polyphenolic flavonoid found in tea, onions and berries [ 88 ] and can act as a direct dye for hair coloring. Tibkawin et al. reported the use of teak leaf extract (quercetin is the main colorant) as a plant hair dye for bleached human hairs wherein the color obtained is dependent on the harvest states of leaves (young leaf extract produces reddish brown while mature leaf extract produces brown color) [ 49 ]. Quercetin is also known for its medicinal bioactivities, such as anticancer and antiviral activities [ 89 ]
Acacetin (5,7-dihydroxy-4-methoxyflavone) is a naturally-occurring flavonoid in the bark of Acacia farnesiana (huizache) [ 90 ]. Ali et al. studied the use of huizache extract to dye gray hair, and the color obtained by direct dyeing was less intense than that dyed with henna extract [ 20 ]. Besides, acacetin has a variety of pharmacological properties including neuroprotective, cardioprotective, anticancer, anti-inflammatory, antidiabetic, and antimicrobial activities [ 91 ].
Collectively, flavonoid colorants may come from a wide range of sources, but their colors are easily affected by environmental pH, metal ions, light, temperature, and oxygen [ 92 , 93 ].
3.4. Indigo
Indigo, also known as indigotin (CI Vat Blue 1), has been used as a vat dye and traditional medicine for thousands of years. In ancient times, freshly picked stems and leaves of “daqingye” indigoid plants [ 94 ], i.e., Indigo Naturalis , Baphicacanthus cusia (Nees) Bremek., Polygonum tinctorium Ait. and Isatis indigotica Fort. were soaked in vat water for several days to ferment and become dark blue [ 95 ]. Thereafter, lime was added and the residues were stirred and precipitated to obtain indigo colorant, which is a glycosylated form of indole precursor (e.g. isatan A and B) [ 96 ]. During fermentation, indole precursors, i.e., hydrolyzed and released indoles, combine spontaneously to form indigo in the presence of oxygen [ 97 ]. Intra- and inter-molecular hydrogen bonding are responsible for indigo’s insolubility in water and dilute acid [ 98 ].
In the dyeing process ( Figure 5 ), indigo is first chemically reduced in alkaline medium to obtain its soluble reduced form. Sodium hydrosulfite is usually used as the reducing agent for textiles dyed with vat dyes [ 32 , 99 ]. Indigo carmine (indigo-5,5′-disulfonic acid di-sodium salt, C.I. Natural Blue 2) was considered as sulfonated indigo which can give hair a blue hue when used as an acid dye [ 51 ]. In the case of hair dyeing, indigo is usually mixed with a certain proportion of henna powders and water to make a paste which can color the hair dark brown [ 50 ]. Komboonchoo et al. [ 32 ] investigated the dyeing characteristics of indigo, lawsone and lawsone-indigo mixture under reducing and/or oxidizing conditions and found that the dyeing performance of indigo dyes strongly depends on the pH of the solution. Indigo, when dyed in a strong alkaline dyeing bath, shows enhanced dye uptake with a dark blue color due to the presence of soluble monophenol and bisphenol ions of its reduced form. Besides, some studies have reported that indigo has anti-inflammatory, antioxidant, antibacterial and immune regulatory activities [ 100 , 101 ]. Overall, indigo is a sustainable and environmentally friendly natural colorant and is widely used in commercial hair dye cosmetics with henna for dark colors.

Molecular structures of indigo, indigo carmine and the pH effect on the soluble forms of indigos.
3.5. Curcuminoids
Curcuminoids are a bis-α, β-unsaturated diketone and in solution exist in equilibrium with the corresponding enol tautomer [ 11 ]. They are abundant in the rhizome of Curcuma longa (turmeric) and can be found in other Zingiberaceae and Araceae families. Under acidic and neutral conditions, the bis-keto form predominates, whereas at >pH 8 the enol tautomer is favored. Curcumin is practically water insoluble at acidic and neutral pHs while in alkali solution the formation of more soluble anionic species takes place [ 11 ]. Curcumin and other related curcuminoids (demethoxycurcumin and bisdemethoxycurcumin) are well known yellow curry colorants and textile dyes ( Figure 6 ). It was reported that under acidic conditions, curcumin dispersed in a water/2-propanol/benzylalcohol solution can be used for direct hair dyeing, giving a distinct yellow color [ 11 ]. Curcumin can also be used for mordant dyeing. Under the action of iron (II) sulfate mordant, bleached hairs can be dyed into an orangish-brown color with resistance to 8 shampooing washes [ 19 ]. Besides, curcumin has been used in traditional medicine for treating diabetes, abdominal pains, menstrual disorders, wounds, eczema, jaundice, inflammations and for blood purification [ 52 ]. Curcumin extracted from turmeric was reported with potent antioxidant, anti-inflammatory, anticancer and hepatoprotective activities [ 102 ]. Nevertheless, poor water solubility and photostability limit the industrial use of curcuminoids in hair dye formations.

Molecular structures of the main types of curcuminoids.
3.6. Carotenoids
Carotenoids are linear conjugated polyene-terranes and the general structure usually consists of a polyene chain with nine conjugated double bonds and two groups at both ends. Highly conjugated electronic systems of carotenoids contribute to their yellow, orange, red and purple colors [ 103 ]. Three types of carotenoids, i.e., zeaxanthin, peridinin, and lutein were successfully applied for hair dyeing ( Figure 7 ). Carotenoid dyes can provide bright hues with good color fastness properties when associated with metallic mordants. Boonsong et al. reported the use of Eclipta alba (false daisy) extract (its main colorant is zeaxanthin) and Terminalia belerica (beleric myrobalan) extract (its main colorant is peridinin) as hair dye for bleached hair with ascorbic acid as a natural color developer and iron (II) sulfate as the mordant, which produced good dyeing performance and dye fastness [ 19 ]. Likewise, lutein obtained from Tagetes erecta (marigold) was used in natural hair dyeing to cover grey after mixing with Cymphomandra betacea (tamarillo) extract and Aloe vera mordant [ 40 ]. Besides, several carotenoids, such as bixin and norbixin, have been used as a good remedy for cardiac, astringent and febrifuge gonorrhea [ 102 ]. Similar to curcumin, carotenoids are hydrophobic dyes extracted using organic solvents such as hexane, acetone and ethyl acetate. Poor photostability is a hindrance to their applications in commercial cosmetic products.

Molecular structures of zeaxanthin, peridinin, and lutein.
4. Technological Innovations for Natural Hair Dyeing
4.1. colorant production by synthetic biology techniques.
Traditional methods of obtaining hair dye colorants from plants is limited by the dependence on complex extraction/purification procedures, varying botanical sources, long cultivation cycles and limited harvest seasons. In recent years, synthetic biology techniques have brought new tools for producing plant colorants by microbial fermentation. Metabolic and genetic engineering approaches have shown great potential to edit or introduce colorant-formation genes into microorganisms for enhanced production of anthocyanins, curcumin and carotenoids [ 104 , 105 ]. The first microbial synthesis of anthocyanins was reported by Yan et al. [ 106 ], wherein a four-step metabolic pathway containing heterologous plant genes was constructed in engineered E. coli and the cytosol was able to take up naringenin and mustard alcohol to produce anthocyanin 3-O-glucoside. Likewise, heterologous synthesis of curcumin was produced in engineered E. coli by using 4-coumarate-CoA ligase (4CL1) from Arabidopsis thaliana , diketide-CoA synthase (DCS) and curcumin synthase 1 (CURS1) from turmeric [ 107 ]. Additionally, a more efficient biosynthetic pathway of β-carotene was assembled in microorganisms by metabolic engineering techniques by increasing the copy numbers of the carB and carRP genes and overexpressing genes related to the mevalonate pathway [ 108 ]. However, despite the substantial advances in the fields of synthetic biology and metabolic engineering, the development of heterogenous microbial cell factories is cumbersome and time-consuming. Additionally, the potential biosafety risks in synthetic biology are a matter of great concern recently. Thus, it is still challenging for industrialization of such technologies to produce various types of natural colorants and apply them in hair dye cosmetics.
4.2. Encapsulation of Colorants for Stabilization and Detoxification
Stability issues (thermal-, light and acidity/alkalinity stability) must be considered when using natural colorants in hair dye cosmetics. Many encapsulation systems have been successfully developed over the past years to protect sensitive phytochemicals or plant extracts from environmental stresses (heat, UV and extreme pHs) [ 109 ]. Encapsulation provides a promising strategy to protect sensitive dye molecules (core material) by enclosing them in an outer shell (wall material) to ensure the stability and dyeability of natural colorants in commercial hair dye products. Microcapsules of natural colorants can be obtained by various encapsulation techniques, including both chemical (emulsion polymerization, suspension polymerization, and interfacial polymerization) and physical (spray-drying, spray-cooling, and co-extrusion) approaches [ 110 ]. Tang et al. used maltodextrin and gum Arabic as wall materials to prepare microcapsules of extracted colorants from Chinese gallnut and henna by spray drying techniques. The encapsulation of gallnut extract significantly improved the photo- and thermal-stabilities as well as formulation stability in alkaline formulation while the encapsulation of henna extract remarkedly reduced its contact toxicity without affecting the hair dyeing properties [ 17 , 44 ]. Therefore, encapsulation technology has provided a route to solve the stability and compatibility issues addressed in natural dyeing with plant colorants.
4.3. Development of Inorganic Nanocarriers for Efficient Hair Dyeing
Benefiting from small particle size, large surface area and nanostructure, and tailorable physicochemical properties, nanomaterials are ideal carriers of bioactive ingredients for cosmetic applications [ 111 ]. In recent years, various inorganic nano-carriers (e.g., nanoparticles, nanofibers and nanotubes) have shown usefulness in stabilizing hair dye plant dyes and enhancing the dyeing effect. For example, carbon nanotubes with small size and increased surface to volume ratio can easily be absorbed onto the hair cuiticles and interact with the hair fiber, leading to enhanced affinity and long-lasting coloring effects [ 112 ]. Similarly, gallic acid reduced/functionalized silver nanoparticles [ 113 ] and colored silica nanoparticles [ 114 ] have been developed as novel dyes for bleached human hairs owing to their local surface plasma resonance properties. Besides, oxidation of dopamine to eumelanin-like polydopamine and deposition on the surface of hair in the form of nanoparticles were shown as a novel biomimetic strategy to develop melanin-mimicking pigments for hair dyeing [ 115 , 116 ]. Furthermore, Panchal et al. [ 117 ] reported that halloysite nanotubes, when loaded with lawsone, were effective for the coloration of both pigmented and grey hairs with good shampooing fastness and can modify the hair surface through physical adsorption and self-assembly. There are also reports on hair dye applications of other abundant and easy-to-obtain natural clay minerals such as sepiolite, palygorskite and kaolin [ 118 ]. Collectively, inorganic nanomaterials incorporated/functionalized with hydrophilic or hydrophobic dye molecules from natural sources have provided possibilities for the development of novel natural colorants-based hair dye products.
5. Toxicological Assessments
Natural origin does not necessarily mean non-toxic or safe. Black henna, a combination of henna leaves and p -phenylenediamine (PPD), is known to cause allergic contact dermatitis [ 56 , 119 ]. Some colorants, such as juglone, are natural toxins with cytotoxic effects [ 65 , 66 ]. Additionally, the colorant contents and toxic effects of plant extracts used in hair dye preparations can be variable with different plant sources, harvest seasons and extraction techniques. The application compatibility of juglone-containing walnut husk extract as a hair dye ingredient was assessed both in vitro and in vivo, showing it is not irritating to the skin [ 12 , 16 ]. Besides colorants, metallic mordants and heavy metal contaminants from plant extracts can be accumulated in humans with frequent dyeing and may impose health impacts. For example, the presence of high levels of Fenton transition metals (e.g., iron) can cause photooxidative damage of hair fiber [ 38 ]. Heavy metals, such as cadmium, chromium and lead, can be absorbed in the skin, liver and kidney, causing allergic contact dermatitis [ 120 ], reproductive system dysfunction [ 121 ], and other adverse health effects. Several novel analytical methods, such as differential pulse anodic stripping voltammetry, were developed for the rapid detection of heavy metals [ 122 , 123 ]. Therefore, safety assessments of both natural colorant ingredients and hair dye formulations are compulsory. It is also necessary to establish a toxicological database of hair dye plants and their main colorants since currently most plant extracts used in hair dye cosmetics are lacking important toxicological information such as acute/sub-chronic systemic toxicity, irritancy, allergenicity and genotoxicity [ 124 ].
Previously, cosmetic safety evaluations were mainly conducted using animal models [ 12 , 18 ]. Nowadays, with global acceptance of the 3R principles (replacement, reduction and refinement) and the EU ban on animal testing in 2013, more in vitro methods have been developed and applied in the toxicological assessments of plant hair dye ingredients and formula. Tang et al. evaluated the eye and skin irritation potentials of four plant hair dyes (henna, Chinese gallnuts, sappanwood, and walnut husks) by using several OECD validated in vitro methods, including bovine corneal opacity and permeability (BCOP) assay in combination with histopathological analysis, Hen’s egg test on chick chorioallantoic membrane (HET-CAM), and a test on reconstructed human epidermis models [ 16 ]. Likewise, a combination of in vitro methods, i.e., micro-direct peptide reactivity assay (mDPRA), HaCaT keratinocytes-associated IL-18 assay, U937 cell line activation test (USENS)/IL-8 levels, blood monocyte-derived dendritic cell test and genomic allergen rapid detection (GARD skin) were reported to assess the sensitization potential of henna-based hair dye products containing p -phenylenediamine [ 57 ].
6. Concluding Remarks
In recent years, green and environmental-friendly plant hair dyes have become the development trend of hair dye market owing to the growing awareness of consumer health. As an alternative to the synthetic hair dyes, botanical colorants are advantageous as they are green in nature, less toxic and biodegradable. Additionally, many plant colorants are known for their health-promoting bioactivities such as antioxidant, anti-inflammatory and antimicrobial properties. Natural colorants are advantageous for sustainability and potential health benefits while their industrial utilization for natural hair dyeing still needs to address the following challenges:
- Cumbersome extraction/purification procedures.
- High susceptibility to environmental pH, metal ions, UV and temperature.
- Low dye uptake and poor color fastness on hairs, especially unbleached hairs.
- Poor color reproducibility on human hair and testing models (e.g., yak hair and wool).
- Dependence on transition metallic mordant makes the dyed hair vulnerable to photo-oxidative damage and complicates the dyeing process.
- Insufficient toxicological data.
Future studies are required to understand the mechanistic interactions between various plant-derived colorants and human hairs, to provide a theoretical basis for the design of efficient plant hair dyes as well as to broaden the sources of hair dye plants or engineered microorganisms for large-scale production. Meanwhile, the development of efficient encapsulation systems, nanocarriers and bio-mordants, to improve the dyeability and color fastness of natural colorants as well as the establishment of toxicological databases for hair dye plants also warrant further investigation.
Funding Statement
This research was funded by the National Natural Science Foundation of China (51403006 and 22074005), the Project of China Society for Drug Regulation (2022-Z-H-033), and the Natural Science Foundation of Beijing Municipality (2202038).
Author Contributions
Writing—Original Draft, H.C.; Writing—Original Draft, W.X.; Visualization, Z.H.; Investigation, L.C.; Investigation, Z.X.; Supervision, Writing—Review & Editing, Funding Acquisition, Y.T.; Conceptualization, Funding Acquisition, Z.Y. All authors have read and agreed to the published version of the manuscript.
Institutional Review Board Statement
Informed consent statement, data availability statement, conflicts of interest.
The authors declare no conflict of interest.
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations.

IMAGES
VIDEO
COMMENTS
Most of the natural dyes showed a very good fastness property in researches. The dyes can be extracted from trees, bark, leaves, flowers, and many more sources. Most of the natural dyes exhibit ...
Abstract. Dyes and pigments can make the world beautiful. They are being used since long time and find vide applications in various fields such as food, textile, artifacts and paper industries. This has resulted in an extensive research on development of natural colorants from natural sources The advantages of using natural colorants are ...
Then, the full text of 1026 papers was reviewed and assessed to check if they were eligible for this scoping review. As a result, 248 studies were included in this review. Open in a separate window. ... This review classifies natural dyes by structural features and summarizes the research progress on natural dyes in the last ten years ...
Natural dyes have some unique qualities, such as calming colors, biodegradability, safety, non-hazardousness, antimicrobial resilience, biodegradability, etc. Plant parts and occasionally their by ...
Natural Dyes Extracted from Flower Crops: A Review of Recent Advances Section A-Research paper Eur. July 2023; European Chemical Bulletin 12(5):5110-5133; July 2023; 12(5):5110-5133;
Recent research has offered a more natural way of coloring foods and providing a clean label. Natural dyes come in several pigments: flavonoids, tannins, alkaloids, indigoids, quinones, chlorophyll, and carotenoids. Carotenoids, chlorophylls, anthocyanins, and betalains are the standard pigments used for food additives.
3. Natural dyeing from a variety of plant sources and color gamut presentation. The interest in using natural dyes from various types of plant sources in textile dyeing has been revived due to their biodegradability, renewability, and environmental-compatibility features (Deveoglu et al., 2012a, Deveoglu et al., 2012b).Generally, natural dyes can be categorized into two main categories ...
Samanta and Agarwal (2009) presented a comprehensive review of studies available on different types of mordants, mordanting methods and their effects on natural dyeing for a variety of textile materials. Most commonly used mordants in natural dyeing are aluminium potassium sulphate, potassium dichromate, stannous chloride, ferrous sulphate and ...
The application of natural dyes on a large industrial scale will require the overcoming of numerous technical challenges, such as the improvement of extraction and application methods in order to increase their cost-effectiveness [34]. The advantages and disadvan-tages of natural dyes are summarized in Table 1.
Dyeing of wool yarn. In dyeing with a single natural dye, the wool yarn (2 g) was dyed with various 2% o.m.f (on the mass of fiber) of natural dye in an acidic buffer solution at pH 3-6 with a ...
The dye Tyrian purple (C.I. 75800) also known as Tyrian red, royal purple and imperial purple is a bromine-containing reddish-purple natural dye, derived from the hypobranchial glands of several marine predatory sea snails in the family Muricidae. This dye has excellent light fastness properties [ 48 ].
While natural dyes face challenges like variability and high production costs, new technologies which use synthetic dyes yet eco-friendly offer more consistent and scalable solutions. This paper highlights the limitations and potential of these methods, emphasizing the need for further research to address cost, scalability, and environmental ...
5.5 Other Applications o f Natural Dyes. In addition to the above m entioned application s, there is a growing interest. for using natural dy es to dye leather [78], to stain wood [79], pulp [80 ...
A comprehensive review on application of natural dyes on textiles and earlier research findings has been discussed in this chapter. Moreover, recently the consumers have become very much conscious about the environment, renaissance of eco-friendly products and process like dyeing textiles with natural dyes, which has thus become also important now. Thus, revival of natural dye application on ...
The aim of the study was to evaluate the performance of dyes extracted from mango leaves in silk dyeing. Extraction medium was optimized by extracting dyes from fixed quantity of crushed leaves under pH values from 3 to 12. The maximum relative color strength of the extracted dye liquor was found to be at pH 10. The optimum dye extraction conditions i.e., the temperature, time, and material-to ...
In addition, some natural dyes have insect-repellent and mildew refrain features, e.g., fungicides derived from maple or eucalyptus tree and indigo for medical applications. Articles related to the natural dyes' functional performance finishing are carefully reviewed and summarized in Table 2. The top two functions that natural plant dyes can ...
This paper has elucidated their multifaceted potential by meticulously examining the extraction methods, characterization techniques and the efficacy of natural dyes as sensitizers in DSSCs. The factors influencing the efficiency of DSSCs utilizing natural dyes have been dissected, providing a deeper understanding of the complex interplay that ...
The meticulous environmental standards in textiles and garments imposed by countries cautious about nature and health protection are reviving interest in the application of natural dyes in dyeing of textile materials. The toxic and allergic reactions of synthetic dyes are compelling the people to think about natural dyes. Natural dyes are renewable source of colouring materials. Besides ...
In this paper reviews, the natural dyes are made from plants, animals, minerals and, insects using environmentally-friendly solvents. Dyeing is a complex, specialized science. ... IJCRT2111281 International Journal of Creative Research Thoughts (IJCRT) www.ijcrt.org 528c some of these have recently been shown to exhibit the antimicrobial effect ...
The aim of this research work is to extract dye from natural source and perform an experimental analysis of the dye adherence on cotton fabric. ... This paper reports the studies available on the ...
2. Hair Coloring Mechanisms. For most natural plant hair dyes, there are two mechanisms for hair coloration: direct dyeing and mordant dyeing. Briefly, the hair dyeing process can be divided into two steps: (i) Diffusion of dye molecules from dye bath to the keratinous hair fiber; (ii) Formation of chemical bonds (hydrogen, ionic, and covalent bonds) between the carboxyl or hydroxyl groups ...
Natural dyes (in the text: N atD) fro m plants, animal (less often) and mineral. resources, has long been used for dyeing of textile, leather, body, hair, for cosmetic. purposes and craft as well ...
Research Article. Open Access. Dye-Sensitized Solar Cells (DSSCs) Based on Extracted Natural Dyes ... the dispersions were filtered through filter papers to extract the anthocyanin (dye) for use as sensitizers. ... three natural dyes were extracted from different fruits and leaves and used as sensitizers for dye-sensitized solar cells (DSSCs). ...
Definition of natural dyes/colouants. The word 'natural dye' covers all the dyes derived from the natural sources like plants, animal and minerals. Natural dyes are mostly non-substantive and ...