- × Search Close Search

How to Become a Clinical Research Associate
Actalent article about all the opportunity available for clinical research associates.
If you like to travel, the life of a Clinical Research Associate (CRA) is a blur of airports, frequent flyer miles, taxis and hotel rooms. This career requires so much travel that living near a major airport can help you land the plum jobs.
The work of a CRA requires concentration and attention to detail. It involves combing through reams of technical medical data, looking for any potential problems. At any given time, pharmaceutical and medical device companies are holding more than 100,000 clinical trials throughout the U.S. , and CRAs are the ones monitoring and overseeing them.
Because of the good pay, the travel and the rewarding work, these jobs are highly sought after.
"It's a highly coveted role that's pretty hard to break into," said Kate Keller , a strategic delivery executive who has spent seven years recruiting thousands of CRAs for positions all over the country. "It's a really high-paying job. Some people really want to travel. And the clinical research industry is pretty cool, for lack of a better word — because you can change people's lives."
How can you become a CRA? Here's what Keller had to say.
How does one get into this line of work?
Well, it's not easy.
Employers prefer job candidates who have a background in nursing or another medical field, since they're already familiar with working with patient charts and clinical health records. "They understand the data. They understand what's happening to the patient," Keller said.
One way to get into the industry is to start out as a clinical research coordinator. CRCs recruit patients for clinical trials, and they conduct the clinical trials at local hospitals and doctor's offices. In contrast, CRAs are the ones who fly in and review how the clinical trial is going.
A good CRC who does his or her job well can get a reference from the CRA they're working with. That's a good way to get an entry-level CRA job.
How's the job market for CRAs looking?
Great! "CRAs have one of the lowest unemployment rates in the country," said Keller, who now manages Strategic Recruiting Center in New Jersey.
She added that, from year to year, CRAs and the clinical research organizations that hire them take turns having the upper hand in the job marketplace.
"The CRAs are definitely more in control right now in terms of the pay rates, the travel they're willing to do, and the type of company they're willing to work for," she said. "But it changes a lot. It probably flips every other year."
Which is more important for a CRA: education or experience?
There are master's degrees and certification programs for clinical research, but experience in the field is more important for these jobs, Keller said.
"If we had a candidate who had a master's degree but no field experience, we wouldn't be able to place them anywhere," she said.
To gain that necessary field experience, it might be more realistic to start out as a clinical research coordinator who's responsible for managing one clinical trial. If you excel at that, it can be a good stepping-stone to an entry-level CRA job.
What other factors help a CRA get good job opportunities?
It's a little like the No. 1 rule in real estate sales: "Location, location, location."
"Since there's so much travel, clients typically want the CRA to be based within 30 minutes of a major airport," Keller said. "The bigger the airport, the more marketable that candidate is" — because the employer can pay less for travel costs if they can avoid connecting flights.
What skills are required in a good CRA?
Communication skills: "The ability to work with different types of people and have difficult conversations with them," Keller said.
Administrative skills: Time management is extremely important for a CRA. You can't fall behind in submitting field reports to your employer.
Technical skills: Mastering different electronic data capture (EDC) systems is crucial. You'll use these data systems to avoid leafing through thousands of pieces of paper. The industry uses a handful of prominent EDC databases with names like Medidata Rave, IMPACT Harmony or DataLabs to capture research data at clinical trial sites. A CRA who's proficient with multiple EDC systems will have a leg up on the competition.
Like your job but curious about what else is out there? Now is the time to visit our job board to find your next great opportunity. Create a free career account today to customize your search. Upload your resume and customize your job search based on your skills and interests. Finally, consider contacting an expert career advisor. Our recruiters are available to provide advice that you can use.
Relevant Insights

Learning & Development is at the forefront of company-wide communication in the aftermath of COVID-19. Actalent shares advice on what to do differently.

How to adapt your workforce in a changing environment

Actalent article about opportunities available in entry level clinical research jobs.

Clinical Research Associate: A Full Guide on Becoming A CRA

Clinical Research Associate
A complete guide on how to become a clinical research associate.
Over 1.9 million students receive a bachelors of science every year. While a few go on to PhD, Masters, and Medical programs; many are ready to start clinical research certification online to start a career in the frontiers of medical research and patient care.
As a new student applying to the science job market, you may only find internships or recognize that even entry-level science jobs requires 1-2 years of experience. More so, you may realize many of these jobs require intense labor in the lab or just did not meet your expectations for your science degree.
This is why a career as a CRA should be considered with clinical research coordinator training. We train over 500 students each month in clinical research coordinator training and clinical research associate training (depending on prior background).
For those who have always wanted a career in medicine or have a gap year before medical school; Clinical Research Training is the next step to getting a head start in your career.
Because the position is unlike actually working in the lab and more of a management role; you get 1-on-1 connections with physicians and medical staff that can lead to a better application for medical school and other medical careers later on.
Best of all; many of these positions accept remote staff (and some allow you to travel 45-75% with full expenses including travel, accommodation, meals, and other per-dime expenses covered).
Clinical Research Training can help you save money while also increasing your salary. CRA’s with our level of training can expect to make between $6,500-$12,000 a month with an estimated promotion rate of 33% a year: an amount that is uncommon in other science-degree careers.
CCRPS is one of the only major US-based ACCRE, ACCME, ANCC, ACPE, and Transcelerate Biopharma accredited CRA certification courses that accepts students with no prior background for certification. T
his is because our course is thorough and created by Senior CRAs who have been in the field for long enough to understand what you need to know to begin working and applying. The course can be completed in as little as 7 days with dedicated full-day study time.
Clinical Research Associate Certification Qualifications
Foreign Doctors Welcome : A Clinical Research Associate or Coordinator plays a vital role in directing and supervising clinical trials conducted by physicians, nurses, and other science professionals. This career path is particularly attractive to many foreign doctors with completed medical degrees (MBBS) who can utilize their expertise in the US healthcare system by pursuing a CRA career instead of taking the USMLE or repeating residency training. For those interested in coordinating aspects, consider the Clinical Research Coordinator course .
Distinct Skillset : Unlike the traditional medical field you may be familiar with after years of schooling, Clinical Research Associate training provides a distinct and valuable skillset. For comprehensive understanding of Good Clinical Practice, see the ICH-GCP course .
Most Extensive Online Course : Our program goes beyond basic introductions, offering a comprehensive curriculum with over 110 modules – the most extensive Clinical Research Associate course available online. This in-depth training ensures you're well-prepared to secure a coveted CRA position.
Superior Coursework : Securing a CRA role is a strategic career move compared to the limitations of many traditional medical positions. While generic courses abound, we've observed that graduates often struggle due to a lack of substantive content. Our Clinical Research Associate course addresses this gap by providing Senior Clinical Research Associate-level training through 110 intensive modules grounded in the latest scientific principles. For those looking to assist in clinical trials, the Clinical Trials Assistant Training may also be of interest.
Diverse Career Opportunities : This high-demand science-based medical field offers diverse opportunities:
Work in the Private Sector : Pursue a CRA career with renowned pharmaceutical companies like Pfizer. Enhance your skills with the Advanced Clinical Research Project Manager Certification .
Academic Opportunities : Work in the academic sphere at medical schools. Those aiming for higher responsibilities may consider the Advanced Principal Investigator Physician Certification .
Unmatched Flexibility and Knowledge : In addition to our exceptional course content, we boast the largest number of clinical research courses available online, providing you with unmatched flexibility and knowledge. For those interested in safety monitoring of drugs, the Pharmacovigilance Certification and Medical Monitor Certification can enhance your capabilities in these critical areas.
Why Take A CRA Certification Course
The role of the clinical research associate is to ensure that medical devices, new treatments and new drugs are approved for patients' use.
This field is taken as a certificate program course in many schools. For example, you may find associate degree programs. These programs can be completed in two years and can be offered through both the online and the hybrid formats. Hybrid formats combine both online and on-campus courses together.
If you opt for a fully online program, you can still get an immersive education. Different platforms like emails and discussion boards are used to ensure and promote interaction between the students as well as the lecturers.
Online learning platforms are used to upload the syllabus, course materials, lectures and assignments. Some online programs include field work as part of their requirements, in order for students to gain first hand experience working with clinical trials and patients. Depending on the school, they may have a list of approved clinical research institutes and other facilities. Otherwise, you will have to find a facility for yourself and get the school's approval.
These certificate programs are generally designed for professionals that are already in the medical fields (like medical assistants or nurses) and are interested in moving to the field of clinical research.
They may therefore ask for a copy of your CV or resumé or they may ask for a letter from your employers to verify that you have the needed medical experience. Some programs may require just an undergraduate degree in a medical science or life science related field.
Clinical research associates are trained to assist clinical researchers and investigators in the coordination, administration and management of clinical trials.
During this training, different courses will be taught revolving around subjects like safety procedures, subject recruitment, regulatory requirements, drug development, accountability, trial management, medical terminology etc.
The importance of the role of the clinical research associate means that companies that conduct clinical trials are usually very selective. The need to comply with strict regulations often inform their decision when making a choice of their clinical research associate. It is therefore very difficult to get a job as a clinical research associate without previous experience in clinical trials.
Many companies require around at least two years experience in clinical monitoring as a clinical project assistant or clinical trial administrator before considering applicants for this important role.
In applying for the post of a clinical research associate , ensure that you read the job description and indicate or highlights the relevant experience on your curriculum vitae. Your cover letter should be specific to the company you're applying to.
Do not use a one-for-all cover letter. Personalize your cover letter to each company and highlight the skills that fit the specific requirements of the role. Not all companies advertise their vacancies, so you can try to find out about other unadvertised vacancies, you might increase your chances.
Further certification can enhance your resume such as the ACCRE accredited CRA program which contains 110 learning modules for Clinical Research Associate Training and Placement
The Best CRA Certification Course For Entry-Levels
There is a huge shortage of well-trained CRAs, but many companies are reluctant to hire untrained entry-level clinical monitors because of patient and trial safety. Because of this, even the beginner entry-level jobs require certification or training.
Our program is considered one of the top clinical research graduate programs online. Most courses provide very light training that may look good because of the company names, but alone is not sufficient to pass the interview rounds a company conducts.
Because our modules are prepared help even Senior Clinical Research Associates, we find more of our students with no background quickly passing their interview rounds.
CCRPS Course covers double to triple the amount of course content than other courses. While many courses are simply 5-20 simple interactive modules, our course covers 140 dense modules in thorough detail.
After each session, students can ask their questions privately with the course instructor, all of whom have 15+ years of CRA experience.
Currently, 82% of our students are hired within the first month of taking the course. Students with limited background or those looking to gain extra experience are offered a remote internship of up to 6 months during the time they are interviewing.
This advantage allows many students with limited experience to get hired with a higher paying job than previously offered.
While a majority of our students are physicians, a majority of the CRA workforce are Science Grads and Nurses. nonetheless, we train all students at a Senior CRA level regardless of background because clinical research monitoring is vastly different from any lab or science course you may have taken.
Clinical research associates are given the protocol of a study including all medical protocol that must be followed but because they do not diagnose or treat. Medical knowledge is supplemental but not sufficient in this career path.
This is the main reason why our Clinical Research Training includes all possible scenarios you may face at the protocol and guideline level in your future company.
How To Get Experience For Clinical Research Associate Jobs
CCRPS, like other educational institutes, is only associated with educating and certifying clinical research professionals so we do not provide job placement. We want to make sure you apply with your best foot forward. Below are links we readily refer to graduates who are looking for job support. Having a great CV and cover letter are essential to applying for jobs. Recruiters are paid by the company which hires you and thus are free for searching employees. Be realistic but also be driven. Make sure you get continue reaching out until you get a true rejection from any job you apply to as they may never have seen your application if you received no response.
Clinical Research Job Advising: Kunal at ClinicalTrialPodcast
Free Resume Review: TopCV TopCV provides a free review and feedback for your current resume.
Resume Distribution: ResumeRabbit Resume rabbit distributes your resume to 60 job posting sites.
Clinical Research Recruiters: I-Recruit I-Recruit distributes your resume to clinical research recruiters.
Clinical Research Job Bulletin: Indeed Indeed usually provides the most uptodate job bulletin for clinical research jobs
Always use a cover letter specific for the company and job when applying if you are not using a recruiter.
The ICH-GCP in Clinical Research
Regardless of the type of clinical research or function of an IP being tested, it is important that clinical research should meet two critical criteria:
The clinical research process should respect the rights, freedom and dignity of tested patients (human participants).
Data from the clinical research process should be accurately collected, safely stored, rigorously scrutinized and correctly interpreted.
One way to ensure that these requirements are met is to follow a set of internationally recognized and accepted standards for clinical research.
Most countries across the world today follow ICH-GCP, that is, International Committee for Harmonization of Good Clinical Practice guidelines in conducting clinical research on human participants7.
The ICH-GCP outlines procedures and precautions that are essential in order to protect the safety and wellbeing of human research participants during clinical research, and to ensure the integrity of data from clinical research studies.
In the USA, clinical studies are required to comply with the FDA Guidance for Good Clinical Practice, outlined in a document titled ‘E6(R2) Good Clinical Practice: Integrated Addendum to E6(R1)’8.
In the USA, clinical studies are required to comply with the FDA Guidance for Good Clinical Practice, outlined in a document titled ‘E6(R2) Good Clinical Practice: Integrated Addendum to E6(R1)’8.z
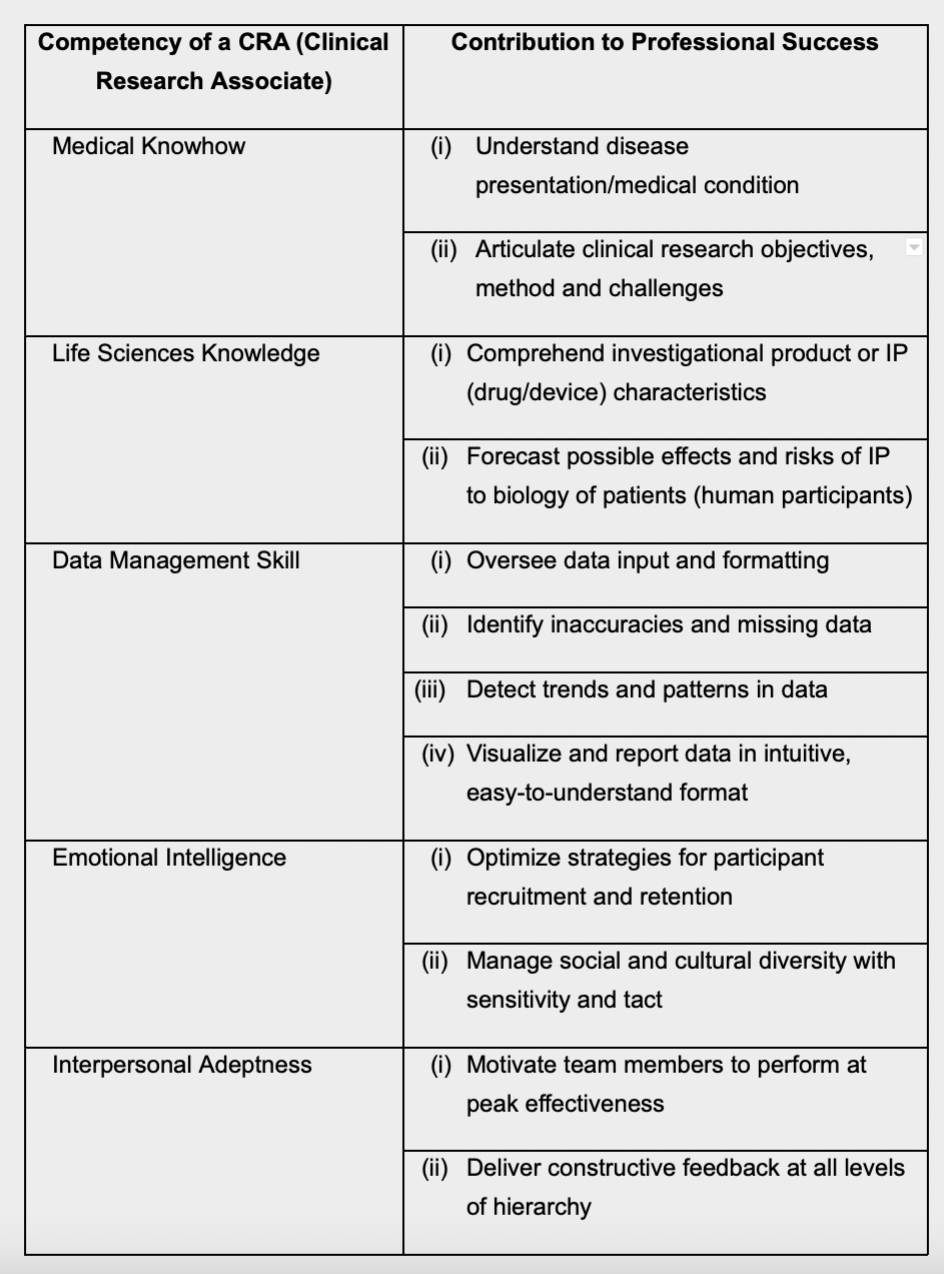
Qualifications and Qualities of a CRA
According to the International Accrediting Organization for Clinical Research (IAOCR), candidates for CRA positions usually hold either a biological science degree, or one in medicine or nursing10.
The New Scientist recommends that aspiring CRAs should possess a good working knowledge of one or more of the following subjects – anatomy, biology, biochemistry, chemistry, immunology, microbiology, pharmacology, physiology or toxicology11.
In addition to a background in medical or life sciences, a CRA is required to have a good grasp of data management, including Electronic Data Capture (EDC), data analytics and reporting12.
Sketching the CRA work profile, the authors Diane St. Germain and Marjorie Good state that CRAs are the ones who scrutinize clinical study data most closely from start to finish—as a result, they are often the first to notice critical patterns and interesting trends, and to report these to the research team as well as to the CRO13.
Equally if not more importantly, a CRA must possess a high level of emotional and interpersonal savvy. This is a crucial area, since a CRA’s success hin ges upon his/her ability to elicit the best from team members, in terms of both performance and probity.
Core Competency Framework for CRAs
To illustrate, the ACRP’s ‘Core Competency
Framework for Clinical Study Monitoring’
requires that a CRA should be able to identify
and correct compliance violations at a study
site. The CRA must not only bring such
violations to the attention of site staff, s/he
must induce them to take corrective action,
as well as reporting the matter and even
escalating it, where necessary14.
The table below summarizes the ideal
competencies of a CRA, and provides
insights on how each ability contributes to
the CRA’s performance.
CRA Career Path
In the past, CRA positions were often filled by individuals with medical or nursing backgrounds, with little thought given to their lack of research training15. As awareness grew about the importance of research experience for a CRA, employers began preferring those with years of experience in clinical research settings, such as Clinical Trials Assistants (CTAs) and Clinical Research Coordinators (CRCs)16.
However, in recent years, the focus has shifted once again from a tenure-based mindset to a skills-based evaluation17. In part, this change has been brought about by the growth in professional courses and training programs in the field.
For instance, many leading US Universities today offer master’s programs in clinical research18. In addition, there are some widely recognized certification programs for clinical research associates, such as those offered by the ACRP19 and the Society of Clinical Research Associates (SOCRA) 20.
Note: You must already be working as a CRA to qualify for the ACRP and SOCRA certification programs.
A Toe in the Door: CRA Certification for a Non-CRA
By this point, you might be wondering, “I have no research experience… I’ve never worked as a Clinical Trials Assistant (CTA) or a s a Clinical Research Coordinator (CRC). Nor do I have a degree in Clinical Research. Can I still become a CRA?”
The simple answer is, yes, you can.
You might be a life sciences graduate looking for a lucrative career in the pharmaceutical or biotechnology sectors. Or, you’re excited by a career in research, but unsure whether the drudgery of a Ph.D. is your thing.
Maybe you’re just looking for a job that represents a great option for someone with your combo of science background plus detail-orientedness.
Whichever of these descriptions best applies to you, a career as a Clinical Research Associate could be exactly right for you.
With the right training, you can be recruited directly to a Clinical Research Associate position, even without a background in clinical research.
So, what kind of training will help me break through the ‘experience’ barrier and land a job as a CRA?
As you’ve already gathered from the table, the skill-set required to be a successful CRA is pretty extensive.
Aside from an in-depth knowledge of scientific and medical concepts and principles, a CRA must have a sound grasp of medical research regulatory requirements, a penchant for being thorough and systematic, as well as a knack for coordinating and managing people with diverse skills, roles and backgrounds.
To our knowledge, CCRPC’s ‘Advanced Clinical Research Associate Certification’ (ACRAC) is one of a kind: The ACRAC is the only multi-accredited* certification program in the US that offers the kind of exhaustive as well as intensive training that equips candidates from a non-clinical background with the abilities and competencies that make a good CRA.
Best of all? The ACRAC is open to fresh graduates holding a B.S. degree in any of the life sciences, with no requirement for prior exposure or experience in clinical research.
*The ACRAC program offered by CCRPC is accredited to ACCRE (Accreditation Council for Clinical Research & Education), ACCME (Accreditation Council for Continuing Medical Education), ACPE (Accreditation Council for Pharmacy Education), ANCC (A merican Nurses Credentialing Center), as well as Transcelerate Biopharma.

Training to be a CRA through CCRPS ACRAC
The ACRAC program includes over 100 course modules that cover all the important knowledge domains and skill-sets required by a CRA.
Designed for a total study time of approximately 250 hours, this training program can be completed at your own pace, or, for those able to dedicate the whole day to study, in as little as two to three weeks.
Starting with a broad overview of clinical research jargon and terminology, the course walks students through the principles of Good Clinical Practice, familiarizing you with the relevant sections of the ICH-GCP and the FDA’s E6(R2).
The program places particular emphasis on ethical practices in research with vulnerable populations.
Students going through the ACRAC are trained in all major aspects of designing a Clinical Trial Protocol in keeping with the Code of Federal Regulations (CFR).
They additionally learn the steps involved in the IRB/IEC approvals process and how to prepare required documents.
Finally, students become aware of the importance of pharmacovigilance and the regulatory process for new drug testing.
A major chunk of the ACRAC certification centers around equipping the CRA for day-to-day responsibilities, such as different types of site visits – preliminary (Site Qualification), preparatory (Site Initiation) and progress monitoring visits (Routine Monitoring).
Crucially, the ACRAC covers essential documentation such as the Case Report Form and Trial Master File, as well as electronic data capture (EDC) and remote monitoring systems.
A vital component of the training program involves empowering students to tackle challenging situations.
For a CRA, these include identifying protocol deviations and violations, and recognizing as well as reporting research fraud and ethical misconduct.
In addition to its comprehensive coverage, the ACRAC certification offers the great advantage of including 17.5 CME credits – that is, course credits that count towards ‘Continuing Medical Education’.
These credits can be used by individuals desiring to further their education and/or careers in healthcare-related fields, including medicine, nursing, pharmacy and research.

Clinical Research Associate Training
Get ahead in clinical research with advanced accredited online CRA certification for $450. Demo our on-demand course below.
Clinical Research Associate Certification
Advanced clinical research associate certification (acrac).
Chapter 1: Introduction
This chapter orients you to the concept of Continuing Medical Education (CME) and outlines how the CCRPS CRA program contents meets AMA requirements for CME. Given that, across the US, physician practitioners are required to complete between 20 and 50 hours of CME credits yearly, the ACCME-accredited CCRPS CRA course can be used not only to build knowledge and skills in the field of clinical trial management, but also to further a successful medical career. Additionally, the introductory chapter introduces you to the clinical terminology and abbreviations commonly encountered in clinical research, for example, Investigational Product (IP), Good Clinical Practice (GCP), Institutional Review Board (IRB) and so on.
Chapter 2: Roles and Relationships in Clinical Trials
The unit presents the foundational background to beginning and building a career as a clinical research associate (CRA). As you know, a CRA plays a critical role in setting up as well as monitoring the clinical trials process for an investigational product or IP – a medical drug or device under development. In this unit, you will learn how a CRA interacts with other stakeholders, including the Clinical Research Organization (CRO) or Sponsor of the clinical trials, the Principal Investigator (PI) as well as other research site staff, the trials monitoring team including the Clinical Research Coordinator (CRC),other CRAs and the Data Safety Monitoring Board (DSMB), as well as the research ethics committee (Institutional Review Board or IRB).
Chapter 3: Sponsor and Investigator Roles
In this unit, you will gain insight into the ICH-GCP guidelines, particularly addendum E6, sections 2 through 5, which outline procedures and precautions essential for protecting the safety and wellbeing of human research participants during clinical research. These include guidelines for obtaining informed consent from human subjects, maintenance of trial records, reporting of compliance, safety and research progress, as well as procedures for suspension or termination of the trials process. The chapter familiarizes you with the critical importance of monitoring for Adverse Events (AEs), including types of AEs and regulations for documentation and reporting.
Chapter 4: Clinical Trial Design
In this chapter, you will acquire insight into the different phases of the clinical trials process, from the pre-clinical phase through Phases 0 to 4 of clinical testing. The unit will familiarize you with important concepts of clinical trials, such as the structure and goals of each phase of clinical trials, approaches to dosing, toxicology of pharmaceutical products, in vitro and in vivo testing, dose escalation and so on. Finally, the chapter reviews the FDA’s drug approval process.
Chapter 5: ICH-GCP – Overview
The chapter dives deep into GCP, including a review of the history of medical research leading up to the ICH-GCP. The unit covers all four QSEM categories of the guidelines for ensuring Quality, Safety and Efficacy of the IP, as well as Multidisciplinary guidelines (mainly pertaining to documentation and electronic data safety standards). In addition, the chapter includes an overview of MedDRA software that provides a standardized system of terminology and notation for documenting clinical research, as well as principles of budgeting for clinical trials.
Chapter 6: Ethical Research in Vulnerable Populations
The unit provides a detailed walk-through of the regulations and compliance requirements for conducting clinical trials with human subjects who meet the definition of a ‘vulnerable population’, including pregnant women and fetuses, children, mentally incapacitated individuals (those with cognitive functioning impaired by neurolopsychological conditions or chronic substance abuse), as well as prisoners. You will acquire familiarity with the challenges of research in such populations, including the requirement for parental consent, fair but not excessive incentive, justifiable deception or incomplete disclosure, coercive practices and so forth.
Chapter 7: Adverse Events
Through this module, you will gain a bird’s eye view of the protocol for documenting, reporting and responding to AEs or adverse events during the clinical trials process. The unit covers concepts such as expectedness, severity and seriousness of AEs, Adverse Drug Reactions (ADRs) as a sub-category of AEs, Investigational New Drug or IND reports, causality analysis for AEs and so on. In addition, the chapter reviews the responsibilities of both research sponsors as well as IRBs in sharing AE information with subjects.
Chapter 8: Clinical Trial Protocol
The chapter provides an in-depth tutorial on the structure and elements of a CTP or clinical trial protocol, as well as guidelines on writing a CTP. Important concepts reviewed include study Risk Benefit Analysis (RBA), study sample statistics (sample size, statistical power, plan for data analysis), risk management and study administration. Additionally, the module covers concepts central to study sample selection, addressing inclusion and exclusion criteria, especially safety and ethics considerations in sampling.
Chapter 9: Protocol Deviations and Violations
Through this unit, you will gain familiarity with the many potential causes of protocol deviations and violations, learning to distinguish between minor (deviations) and major departures or violations of protocol. Content provides understanding of the most commonly occurring violations, including both minor (off-schedule subject assessments, subjects’ use of prohibited drugs, and so on) as well as major violations (failure to obtain informed consent, failure to report AEs and so forth). Further, the chapter reviews principles for reporting protocol deviations, IRB approval for planned deviations and related concepts.
Chapter 10: IRB and DSMB
This chapter briefly reviews the history of IRBs and examines the principles guiding IRB decision-making. In addition, the unit discusses recent developments in compliance, including sIRB (single IRB) and SmartIRB for institutions that are part of the CTSA (Clinical and Translational Science Awards). The bulk of this module dives into the categories of IRB review, including full board and expedited review, examining criteria for review exemption such as educational or purely behavioral research, as well as studies collecting identifiable data, surveys and interviews.
Chapter 11: Review Questions
The module provides a self-assessment tool by including questions that review the content covered in previous chapters. The set of 71 questions examines all aspects of ICH-GCP previously discussed.
Chapter 12: Site Monitoring Visits
In this module, an overview is provided of the different types of site monitoring visits, including site selection or qualification visit, study initiation visit, routine or progress monitoring visit, as well as study termination or close-out visit. Important concepts discussed include pre-qualification preparations and site feasibility assessment as well as study monitoring criteria (data omission, incorrect entries, inaccurate calculations, documentation of corrections and so on). For each type of site monitoring visit, the chapter reviews relevant documentation.
Chapter 13: Site Qualification Visit (SQV)
The chapter gives an in-depth understanding of the stages and steps involved in selecting a study site. Elements reviewed within the module include the process of investigator selection and criteria for site evaluation (the four P’s: Patient, Protocol, Performance, Profit). Importantly, the module reviews the most common errors in feasibility assessment, including overestimation of sample availability at site, selection of site staff with low motivation, poor-performing sites owing to high competition for personnel and resources (for example, owing to multiple studies running on a single site), and so on.
Chapter 14: Site Initiation Visit (SIV)
The module dives into the details of an SIV or site initiation visit. You will review the procedure for pre-SIV preparation, including filing for IRB and other necessary approvals, permits and licenses. Additionally, the chapter examines elements of the SIV agenda, mainly orientation and training of site staff, creation of important study-related documents such as the Trial Master File (TMF) and post-SIV filing of compliance documents such as FDA form 1572 and Financial Disclosure Form (FDF) for relevant site personnel.
Chapter 15: Routine Monitoring Visit (RMV)
In this unit, the elements of a routine or periodic monitoring visit are discussed in detail. You will become familiar with the agenda of an RMV, which prioritizes receiving updates on AEs from site staff (incidence, documentation, seriousness and so on), as well as oversight of the overall progress of trials. The chapter covers different approaches to site monitoring, contrasting traditional (full-scale) monitoring with risk-based monitoring (RBM), as well as comparing on-site monitoring with remote monitoring. A crucial concept addressed by the unit is Source Data Verification (SDV), which is central to obtaining meaningful, high-quality data from clinical trials.
Chapter 16: Site Close-Out Visit (SCOV)
The module gives you a comprehensive overview of the protocol and procedures involved in terminating or closing out a trial site. Aspects covered in the chapter include pre-SCOV preparations such as IRB notification and schedule coordination among site staff (PI, other investigators, medical staff) and monitoring team (CRC, CRAs and so on), agenda for an SCOV – drug inventory management, database verification and lockdown, subject intimation and completion of all subject-related documents, staff-related documentation as well as other administrative tasks including close-out report compilation.
Chapter 17: Tools for Monitoring Visits
This unit outlines a host of tips and tools that can help a CRA in successfully tackling the complex process of monitoring clinical trials. The chapter lists numerous physical accessories you can use for effective monitoring, including scheduling and calculation aids, ready reckoners for drug information and medical terminology, as well as document templates to speed up the process of obtaining trial updates while also serving as checklists for the site visit agenda. Additionally, the unit highlights helpful strategies that a CRA can use to ensure that site visits go smoothly, from travel advice to team-building suggestions.
Chapter 18: Audit and Inspections
The module deals with one of the most crucial and often most feared aspects of a CRA’s career – audits and inspections by the CRO (sponsor), FDA or other regulatory authority. Starting from the basic distinction between an audit and an inspection, the chapter covers in detail the protocols for both audits and inspections. Crucially, the chapter will enable you to grasp the difference between a routine audit/ inspection and a ‘for-cause’ audit/ inspection. Further, it lays out the sequence of an FDA inspection in full (including a detailed walk-through of the FDA BIMO or Biomedical Research Monitoring Program inspection), and provides important guidelines on the do’s and dont’s for CRAs during an audit/ inspection, such as the critical ‘3 to 5 minute rule’. You will acquire familiarity with important audit and inspection-related documents such as FDA Form 482 (Notice of Inspection) and Form 483 (Notice of Observation) as well as the Establishment Inspection Report (EIR) prepared by the auditor/ inspector. Finally, you will gain insight into the classes of observations provided in an EIR, including NAI (no action indicated), VAI (voluntary action indicated) and OAI (official action indicated)—the last is commonly termed an ‘FDA warning letter’.
Chapter 19: Review Questions
The unit contains a self-assessment tool comprising 65 questions that review the content covered in previous chapters, as well as a 15-item quiz. Questions and quiz examine all aspects of clinical trial quality monitoring, including monitoring visits, tools as well as audits and inspections.
Chapter 20: SDV and Informed Consent
In this chapter, the ICH-GCP section 4.8 guidelines on obtaining informed consent from subjects are discussed in detail, highlighting the need for using non-technical language, transparent delineation of risks, consent without undue influence, obtaining consent (and assent) from minors and their Legally Acceptable Representatives (LARs), as well as consent from non-English speakers and sedated subjects. The chapter additionally covers important aspects of Source Data Verification (SDV) with respect to electronic as well as paper-based medical records, and highlights the central goal of SDV, which is to conform to ICH-GCP requirements that subject trial data (as recorded in Case Report Forms or CRFs) must correspond to source data (previous medical records).
Chapter 21: Case Report Form
The module provides an in-depth tutorial on the structure and elements of a Case Report Form or CRF, including the different forms for PI verification, subject enrollment, eligibility and randomization, medical history, physical examination and laboratory data, compliance, adverse events and so on. In addition, the chapter outlines important data notation rules, such as the use of accepted acronyms (‘ND’ for missing data and ‘UNK’ for unknown information, MM-DD-YY format, time-stamp data and so forth), as well as guidelines for the design of CRFs (such as consistency of notation, avoidance of data fields that can be computed and of duplicate data fields and so on).
Chapter 22: Quality Control and Safety
Within this unit, you will learn the central concepts of Quality Control (QC) in the context of clinical trials, including definitions of QC and its relationship with the complementary process of Quality Assurance (QA), the use of Key Performance Indicators (KPIs) in QC, need for a Corrective and Preventive Action (CAPA) plan and so on. Additionally, the module examines the QA process, focusing on the central role of RBM or risk-based monitoring in present-day QA as well as providing guidelines on Quality Metrics (QMs) for evaluating the trials process. The chapter also reviews ICH-GCP guidelines on subject safety, underlining risk-benefit assessment, stoppage rules (for instance, in case of SAEs) and reporting responsibilities. Finally, it introduces the FDA’s Human Research Protection Program (HRPP) as a platform that provides training and support for personnel involved in clinical trials.
Chapter 23: Technology in Trials
In this chapter, an in-depth tutorial is provided of the systems used in modern clinical trials for Electronic Data Capture (EDC) and database management. Systems such as Interactive Response Technologies (IRTs) including IVRS and IWRS (Interactive Voice and Web Response Systems, respectively) as well as RTSM systems for Randomization and Trial Supply Management are examined. The unit reviews the benefits of standardized data management and data sharing, approaches to database management and the concept of an Independent Data Monitoring Committee (IDMC). Critical elements of data integrity, such as proper anonymisation and coding, completeness of data, data safety precautions and logging of site visits and other progress reports are highlighted. The unit further examines the essential features of a good Clinical Data Management (CDM) system that complies with FDA CFR Title 21 and HIPAA regulations, such as setting access privileges, tracking changes and updates, data security and locking, flagging and reconciliation of AEs and so forth. Finally, the chapter looks at CTMSs (Clinical Trial Management Systems) in depth, covering the aspects that allow management of day-to-day trials in multi-site studies.
Chapter 24: Modernized Monitoring (Remote, Risk-based, Centralized)
This chapter offers a detailed walk-through of modern, remote monitoring of clinical trials, which evolved into a full-fledged system in response to the COVID-19 pandemic. Important concepts discussed include the critical site initiation process, Electronic Source Data Verification (ESDV) and FDA regulatory guidance for remote monitoring of clinical trials. In this module, you will learn how FDA’s ALCOA (Attributable, Legible, Contemporaneous, Original and Accurate) criteria for data quality have been adapted to remote monitoring. Further, the unit discusses how HIPAA compliance in remote monitoring is achieved by using limited data sets (wherein sensitive individual information is concealed through anonymous subject codes) regulated by data use agreements. Finally, the unit examines how risk-based monitoring approaches have allowed centralized monitoring to evolve into a cost-effective and safe method for clinical trial monitoring.
Chapter 25: Pharmacovigilance and Regulatory Affairs
Through this unit, you will gain insight into the process and rationale behind pharmacovigilance (PV) and its central role in the clinical trials process. The chapter reviews the statistics on AEs, distinguishes between Type A and Type B AEs, and profiles seriousness of ADRs or Adverse Drug Reactions as well as the iGuard Drug Risk Rating System. Importantly, the unit covers ADR causality assessment in detail, including both severity and probability assessment. An important element of PV addressed in this module is the Individual Case Safety Report (ICSR), its structure, content and role in trial monitoring. Other concepts discussed include types of PV inspections (routine vs. ‘for cause’), PSURs or Periodic Safety Update Reports and study criteria for instituting DSMBs (Data Safety Management Boards). Finally, the module also reviews the domain of Regulatory Affairs (RA) as a function of PV, outlining roles and responsibilities of RA personnel as well as the importance of RA in streamlining the process of drug development by ensuring compliance throughout manufacturing, clinical trials, marketing and advertising.
Chapter 26: Investigational Product
In this chapter, an in-depth review is provided of the protocol for receiving, storing and dispensing the IP or investigational product. At every stage, guidelines lay down strategies for ensuring verifiability, accountability and safety of both study subjects and staff. Thus, IP handling precautions include the need for logging date of manufacture, temperature throughout transit, as well as batch number and individual unit numbers (such as bottle or tube identifiers) carefully and accurately, as well as recording shipping details and filing shipping receipts. Additionally, the unit addresses the need for IP dispensing precautions, such as limiting dispensation to authorized personnel only, as well as maintaining individual subject IP logs.
Chapter 27: Local and Central Labs
The module profiles the evolution of lab testing in clinical trials, from error-prone localized laboratory testing to centralized testing that allows homogeneity of testing procedures and measurements, thus minimizing errors and improving outcomes. The chapter reviews standards for clinical trial laboratories as per the GLCP (Good Clinical Laboratory Practice) and CLIA norms (Clinical Laboratory Improvement Amendments), as well as providing guidelines for lab audits, including fire safety, protective gear, staff training and so forth.
Chapter 28: Review Questions
The unit contains a self-assessment tool comprising 65 questions that review the content covered in previous chapters, as well as a 15-item quiz. Questions and quiz examine all aspects of trial documentation (SDV, CRF, ICSR), quality control, pharmacovigilance, as well as IP and lab guidelines.
Chapter 29: Regulatory Documents in Clinical Trials
The chapter reviews essential documentation to be created and maintained throughout the course of the clinical trials, including the Trial Master File (TMF), FDA forms 1571, 1572, 3674, 3454/3455 and CFR Title 21 Form 312, besides ethics approval documents such as the IRB-approved protocol, informed consent form, subject education and study advertising materials. You will acquire in-depth familiarity with each of these forms, and learn the importance of maintaining and updating records, for example by incorporating IRB revisions and amendments, periodic renewals of permissions and licenses and copies of submitted reports. In addition, the unit summarizes the need for filing documents outlining study- and site-specific procedures, including SOP (Standard Operating Procedure), MOP (Manual of Procedures), Investigator Brochure (IB), Delegation of Authority Log (DOAL), site staff CVs, SAE notifications, logs of subject screening and enrollment, IP storage (temperature, humidity, etc.) and all relevant study parameters.
Chapter 30: CFR Title 21 Part 11 – Electronic Signatures
This unit gives you an overview of Title 21 of the FDA Code of Federal Regulations (CFR), including Chapter 1 sections on informed consent (Section 50), IRB approval (Section 56) and so on, Series on food (100), pharmaceuticals (200 and 300) and so on, as well as FDA Drug Schedules. The major part of the module focuses on Part 11 which deals with Electronic Records and Electronic Signatures (ERES), laying down the criteria for determining safety and reliability (trustworthiness) of electronic data and signatures.
Chapter 31: New Drug Application
Through this module, you will gain knowledge of the FDA process for evaluating a drug under development, and the role of a CRA in streamlining this process. An important distinction covered here is the difference between an IND (Investigational New Drug) and an NDA (New Drug Application). The chapter discusses in-depth the criteria used in evaluating an IND, including toxicology and pharmacokinetics data, as well as requirements for different drug classes (oncology vs. non-oncology). Additionally, the unit covers FDA requirements for AE reporting, including assessment of seriousness, expectedness and format for expedited reporting of life-threatening SARs, as well as safety reporting requirements for investigators.
Chapter 32: Trial Master File
The unit provides a detailed breakdown of the organization of a TMF or Trial Master File, listing the various binders that should be included within the TMF, as well as their contents. Thus, the TMF should contain binders pertaining to the study protocol and IRB, investigator qualifications, FDA forms and correspondence, FDFs or Financial Disclosure Forms, communications with the CRO, and other relevant trial aspects. A helpful templatic guide to creating a TMF is also provided in this chapter, as well as a self-assessment quiz of 10 items on important sections of a TMF.
Chapter 33: Disclosures and Payments for PI, Site, Patients
In this chapter, FDA guidelines regulating financial disclosure are discussed in-depth, covering the definition of ‘conflict of interest’ and the stipulations of Title 21 Section 54 on disclosure requirements. The unit helpfully contrasts FDA requirements with Canadian and UK/EU policies. You will study real life case examples of conflict of interest, as well as lawsuits pertaining to financial disclosure disputes to help gain a better understanding of the potential problems arising from failure to disclose financial interests in clinical trials. Another important dimension covered in the module is the regulation of payments to PIs and other investigators as well as patient payments, which must comply with CMS (Center for Medicare and Medicaid Services) policy on ‘fair market value’ as well as the Federal ‘Anti-Kickback Statute’. The unit contains guidelines on clinical trial budgeting and subject payments. Finally, the chapter reviews IRB guidelines on advertising to recruit human participants for clinical trials, including stipulations against misleading and coercive language, as well as excessive incentives.
Chapter 34: Patient Recruitment, Retention and Compliance
The unit provides an overview of the process of patient (subject) recruitment in clinical trials, from population research to identify motives for participation, to media support for building up public awareness and interest, to community and physician outreach for referrals and enrollment. Additionally, the chapter identifies common barriers to meeting recruitment goals and outlines strategies for maximizing recruitment, such as relaxing overly stringent criteria, offering reasonable incentives such as travel reimbursement and highlighting benefits of participation. Similarly, the unit covers common causes of patient drop-out as well as strategies for minimizing drop-outs, such as improving patient experience (increased attention and listening to patients, flexible scheduling of visits to suit patients’ convenience and so on). Finally, the unit discusses novel strategies to increase patient retention and improve compliance in clinical trials; these techniques harness technology to yield better outcomes, for example, simplifying form completion through digitized forms with auto-fill features, gamifying elements of compliance reporting, and so forth.
Chapter 35: Misconduct and Fraud
This module discusses the various motives for committing scientific fraud and the fallout of fraudulent practices in clinical trials. A scale for classifying errors in clinical trial data is presented, with ‘honest, isolated mistake’ at one end of the spectrum and ‘deliberate data falsification with malicious intent’ at the other. Types of clinical data that may be falsified, methods used in falsification (fabrication, substitution, omission), as well as scenarios in clinical trials where falsification may be occurring are presented. Through this chapter, you will gain familiarity with the signs to watch out for during the actual clinical trials process.
Chapter 36: Review Questions
The unit contains a self-assessment tool comprising 65 questions that review the content covered in previous chapters, including questions on all aspects of regulatory documents, site documents (TMF and contents), trial budgeting and payments, patient recruitment and scientific fraud.
Chapter 37: Site Visit Templates
This module contains a set of templates that you can use for documenting the details of site monitoring as a CRA, either in their current form, or in a form adapted to the needs of your own study. The templates included in this unit include:
Site Qualification Visit (SQV) – checklist for preparations, questionnaire for assessing the site prior to the actual visit, assessment form and follow-up letter
Site Initiation Visit (SIV) – agenda for visit, confirmation letter to request PI attendance during SIV, report following SIV
Routine Monitoring Visit (RMV) – confirmation letter to request PI attendance, report following RMV, follow-up letter
Site Close-Out Visit (SCOV) – confirmation letter to request PI attendance, agenda for SCOV, report following SCOV, follow-up letter
CRA transition letter – document notifying site PI of appointment of new monitor (yourself as CRA)
Chapter 38: Interviewing and Career
In this unit, you will find suggestions and recommendations for making a positive impact in interviews for CRA positions, as well as tips and strategies for making rapid progress in a clinical research career.
Chapter 39: Final Examination
This module comprises a comprehensive 51-item, self-paced quiz to assess your competency in the skills and knowledge required for a Clinical Research Associate position.
https://www.beroeinc.com/category-intelligence/clinical-research-organizations-market/
https://www.linkedin.com/jobs/search?keywords=Clinical%20Research%20Associate&location=United%20States&geoId=103644278&trk=public_jobs_jobs-search-bar_search-submit&position=1&pageNum=0
https://www.centerwatch.com/articles/24791-demand-for-experienced-clinical-trial-professionals-outpacing-supply-acrp-says
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3317309/
https://www.niaid.nih.gov/research/dmid-investigational-product
https://www.fda.gov/patients/clinical-trials-what-patients-need-know/what-are-different-types-clinical-research
Dixon JR. 1999. The international conference on harmonization good clinical practice guideline. Quality Assurance. 6(2): 65-74. DOI: 10.1080/105294199277860
https://www.fda.gov/files/drugs/published/E6%28R2%29-Good-Clinical-Practice--Integrated-Addendum-to-ICH-E6%28R1%29.pdf
https://www.who.int/groups/research-ethics-review-committee/recommended-format-for-a-research-protocol/
https://iaocr.com/finding-first-clinical-research-job/
https://jobs.newscientist.com/en-au/article/a-career-in-clinical-research/
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3326906/
St. Germain DC, Good MJ. 2017. Data management in clinical trials. In: Gallin JI, Ognibene FP, Lee Johnson L, editors. Principles and practice of clinical research. San Diego: Academic Press. p. 531-545. ISBN 978-0-12-849905-4
https://acrpnet.org/wp-content/uploads/dlm_uploads/2017/04/clinical-study-monitoring-competencies.pdf
https://www.clinicalleader.com/doc/starting-a-career-in-clinical-research-things-we-wish-we-knew-0001
https://www.proclinical.com/blogs/2021-9/how-to-get-a-job-as-a-clinical-research-associate-cra
https://acrpnet.org/2018/06/11/5-clinical-research-trends-emerge-at-acrp-2018/
https://www.collegechoice.net/sciences/clinical-research/best-masters-degrees/
https://acrpnet.org/certifications/cra-certification/
https://www.socra.org/certification/program-overview/
Pharmacovigilance: A Complete Guide to Pharmacovigilance and Drug Safety Training
The ultimate guide to clinical research monitoring.

A Day in the Life of a Clinical Research Associate
For the 2023-2024 academic year, we have 112 schools in our MHAOnline.com database and those that advertise with us are labeled “sponsor”. When you click on a sponsoring school or program, or fill out a form to request information from a sponsoring school, we may earn a commission. View our advertising disclosure for more details.
Every pill, cream, tonic, and capsule a pharmacist passes over the counter to an individual has taken a long journey from concept to the point of sale. In fact, many medications that do not require a prescription, such as those found in the allergy, cold, and flu aisle, also go through the same arduous process.
Before a drug can be prescribed and sold, it must be proven safe and effective. Pharmaceutical companies, universities, and health organizations put drugs through clinical trials or clinical study processes to obtain this proof. In the clinical trials world, the company that provides financial support to a research group selected to put the drug through clinical trials is known as a contract research organization or CRO. Primarily, CRO organizations offer financial aid for pharmaceutical development and biotechnology for agricultural and medical device industries.
In the case of pharmaceuticals, drugs that have not yet been approved for sale are administered to vetted participants in a controlled manner during a clinical trial. Clinical trials involve a great deal of documentation, analysis, observation, and organization. A team of professionals is engaged in administering a clinical trial, including a clinical research associate (CRA).
The CRA acts as a liaison between the study’s sponsor CRO (e.g., pharmaceutical company) and the clinics where the study occurs. Because the clinical trial results must be kept entirely transparent and not influenced by the sponsor’s interests, this is a critical role. Therefore, a successful CRA will be detail-oriented, highly educated, and communicate clearly with sponsors and clinical representatives.
THANK YOU FOR YOUR INTEREST IN Southern New Hampshire University Online MS - Construction Management
Work environment of clinical research associates.
A clinical research associate works both at clinical sites and sponsor locations. During a trial, the CRA conducts regular site visits—virtually and physically—to ensure good progress and record-keeping on the clinical site.
CRAs are often responsible for multiple trials at one time, meaning significant amounts of travel between these sites. Sometimes, a CRA may be assigned to a specific geographical region, limiting travel.
Clinical Team of a Clinical Research Associate
Typically, a clinical research associate needs to have direct contact with the participants involved in the study. However, CRAs must work in a collaborative environment, coming into frequent contact with the clinical team at the study site and the supervisors from the study sponsor. Thus, clinical research associates work in the middle of the chain of command, which begins at the top with:
- Contract research organization (CRO) or sponsor
- Principal investigator (PI)
- Clinical research associate (CRA)
- Clinical research coordinators (CRC)
In short, a CRA is focused more on data, accuracy, and quality control while a CRC collects data and interacts with patients. Dan Sfera , in his YouTube channel titled The Clinical Trials Guru, summarizes the CRA profession with this description: “CROs select various PIs across the country, and PIs make sure that sites follow good clinical practices and protocols. They ensure this by hiring CRAs who look at all the recorded data and do not interact with patients when they are completing a site visit.”
Typical Daily Responsibilities of Clinical Research Associates
The daily responsibilities of a CRA are mainly dependent on the stage of the trial they are supervising. As such, below is a breakdown of the typical duties of a CRA during the beginning, middle, and end of a clinical study.
Before a Study Begins
Every clinical study must take place in an appropriately equipped clinical location. The CRA plays a critical role in selecting a site for a clinical study and may even be asked to suggest sites based on their previous experiences. CRAs may also evaluate the applications of sites that self-select as eligible for a particular study. A pool of potential study sites is narrowed down by having sites complete and submit a feasibility survey.
When the pool of sites has been narrowed down, the clinical research associate conducts site selection visits with the chosen locations. During these visits, the CRA spends up to half a day confirming the validity of the feasibility study, meeting with the team (mainly the assigned coordinator), and observing the capabilities and equipment at the facility.
Upon completion of the site visit, the CRA compiles a report for the study sponsor and presents their findings and recommendations for proceeding with the study.
During a Study
Once a site has been selected, the CRA is now responsible for ensuring that the site knows the sponsor’s required protocol and is appropriately set up to conduct the study. In addition, the clinical research associate conducts site visits at regular intervals throughout the study to ensure that protocols are followed and data is effectively collected.
Depending on the study, CRAs may conduct in-person site visits as well as virtual visits. In recent years, the use of remote visit technology has allowed CRAs to review paperwork online, for instance, and reserve in-person visits for necessary personal interactions. During site visits, the CRA ensures that the study is always proceeding with good clinical practices. More details on the specific methods are available below.
Ultimately, since the CRA is a liaison, developing and maintaining positive relationships is an essential part of the job. During the trial, the CRA must communicate effectively and help the clinical staff appropriately to ensure the study progresses smoothly.
When a Study Ends
The CRA typically conducts a closeout visit after a study or when it becomes necessary to end a study—e.g. when enrollment is too low.
During the closeout visit, the CRA verifies that all paperwork is in order and that all obligations have been met on both sides. For example, the verification process may mean ensuring the trial drugs are returned or destroyed, completing and adequately filing all documentation, and compiling all information necessary to complete a final report for the study sponsor.
Required Skills & Knowledge of Clinical Research Associates
Based on the responsibilities outlined above, it should be no surprise that CRAs must have a knack for detail. Proper documentation, filing, and storage are all critical parts of the job description. It is up to the CRA to ensure that the sponsor and clinic understand their obligations at every point of the study. All policies and procedures are being followed to ensure successful data collection.
In addition to being detail-oriented, a good CRA also pays heed to the ethics of the position. For example, clinical trials and studies can have extensive consequences for the organizations that sponsor them, as well as for the trial participants, and eventually, for consumers at large. Therefore, CRAs act as vital monitors for ethical issues and should be able to stand up to any perceived transgressions.
Above all, it is essential that a CRA understands and can implement good clinical practices (GCP) successfully. The GCP guidelines are an internationally developed set of standards from the International Conference on Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) that details ethically and effectively conducting drug trials with human subjects.
The ICH topics are divided into four categories: quality, efficacy, safety, and multidisciplinary. CRAs can find the guidelines in more detail on the ICH website. The YouTube Channel, GCP Mindset , outlines the different roles and responsibilities of CRAs. In short, upholding the GCP standards is the most critical part of the CRA job, meaning that any CRA must be familiar with GCP and recognize and enforce these practices in a clinical setting.
Certification for Clinical Research Associates
While certification is not a legal requirement to work as a clinical research associate, it can provide an important differentiator when looking for employment or career advancement in many professions.
Two main bodies offer certification for CRAs: the Association of Clinical Research Professionals (ACRP) and the Society of Clinical Research Associates (SOCRA).
Association of Clinical Research Professionals (ACRP) – Certified Clinical Research Associate (CCRA) Certification
To earn the Certified Clinical Research Associate (CCRA) credential from ACRP, applicants have two available pathways:
- Pathway 1 – Clinical research professionals with 3,000 hours of verifiable work experience are eligible to sit for the CCRA Exam.
- Pathway 2 – Clinical research professionals with 1,500 hours of verifiable work experience and a clinical research degree are eligible to sit for the CCRA Exam.
Applicants also have to submit proof of their current job description and resume and pass the CCRA exam. In addition, CCRAs must complete at least 24 hours of continuing education credits and be recertified every two years to maintain certification. Beyond the CCRA certification, ACRP also offers certifications for project managers, research coordinators, and principal investigators, and an umbrella certification for clinical professionals.
Society of Clinical Research Associates (SOCRA) – Certified Clinical Research Professional (CCRP) Certification
To earn the Certified Clinical Research Professional (CCRP) certification from SOCRA, applicants must either:
- Have at least two years of full-time experience as clinical research professionals or 3,500 hours of part-time experience in the past five years
- Have a degree in clinical research plus at least one year of full-time experience (or 1,750 hours part-time during the previous two years)
- Have a certificate in clinical research (undergraduate or graduate), a degree in science, health science, pharmacy, or a related field (associate’s or bachelor’s), plus at least one year of full-time experience (or 1,750 hours part-time during the previous two years)
Applicants must also pass the CCRP exam and be recertified every three years. Recertification requires 45 total hours of continuing education credits.
The critical difference between the SOCRA and ACRP certifications is that ACRP expressly certifies CRAs, while the SOCRA certification can apply to other clinical research professionals.
Matt Zbrog is a writer and researcher from Southern California. Since 2018, he’s written extensively about emerging issues in healthcare administration and public health, with a particular focus on progressive policies that empower communities and reduce health disparities. His work centers around detailed interviews with researchers, professors, and practitioners, as well as with subject matter experts from professional associations such as the American Health Care Association / National Center for Assisted Living (AHCA/NCAL) and the American College of Health Care Executives (ACHCA).
Related Programs
- 1 Online Master’s in Clinical Research Administration Programs
- 2 Online Doctor of Health Sciences (DHS)
- 3 Online Master’s Degrees in Food Safety – Regulatory Affairs & Quality Assurance
- 4 Online Master’s in Bioinformatics Programs
- 5 Online Master’s in Biotechnology Programs
Related FAQs
- 1 Clinical Significance vs. Statistical Significance
- 2 How Do I Become a Clinical Trials Research Nurse?
- 3 How Do You Become a Clinical Research Coordinator (CRC)?
- 4 What Can You Do with a Degree in Clinical Research Administration?
- 5 What is Clinical Data Management (CDM)?
Related Posts
A day in the life of a clinical data analyst.
Every day, health organizations like hospitals, clinics, and physician offices collect data about their patients. This information is used to make data-driven decisions in order to provide the highest level of care to their patients as well as reduce expenses and errors. From the outcome of a particular treatment to a large-scale clinical trial, health data is a critical part of the modern healthcare process. Clinical data analysts help to make sense of the extensive data that is at their fingertips, creating stories that turn numbers into actionable intelligence to improve healthcare outcomes.
Healthcare Debates: Does Costly US Healthcare Fund R&D and Medical Research?
This article explores research and development in the context of greater healthcare expenditures, including looking into the main drivers of healthcare spending, influences on pricing, and where R&D fits into the whole ecosystem.
Pharmaceutical Quality Director – A Day in the Life
Consumers and corporations need to know that the drugs dispensed by pharmaceutical companies are manufactured safely and efficiently. Pharmaceutical quality directors ensure that quality control protocols are followed within the industry’s manufacturing, testing, and inspection procedures.
Closing the Gap: Women Leading Healthcare
Women drive healthcare. They are the industry's biggest consumers and workers. They serve as caregivers in their homes and make most decisions for their family's health. Why is it, then, that of the 40 Fortune 500 healthcare companies, not a single one is helmed by a woman?
Clinical Application Analyst – A Day in the Life
Electronic health records (EHR) and other clinical software systems are the new industry standard in healthcare. However, plenty of clinics, hospitals, and private practices have yet to make the full migration. That’s where clinical application analysts come in.
CCRA Certification
Ccra® (certified clinical research associate) is a credential formally recognizing clinical research professionals with experience monitoring and supervising the conduct and progress of clinical trials on behalf of a sponsor., this trusted mark of excellence in clinical research is awarded to clinical researchers who have demonstrated proficiency of specific knowledge and skills by passing the standardized ccra® certification exam..
Apply for Your Exam

Clinical research professional with 3,000 hours of verifiable work experience are eligible to sit for the CCRA ® Exam. Complete eligibility criteria is defined in the Academy’s policy manual .
What qualifies as work experience, work related to human subject research, paid contractual agreement – employer/employee, can be verified by acrp through employer, what is excluded from work experience, any work that is part of a degree track or education program, any experience older than ten years, internships paid or unpaid.


Clinical research professionals with 1,500 hours of verifiable work experience and a clinical research degree are eligible to site for the CCRA ® Exam.
What qualifies as a clinical research degree, any degree awarded in clinical research from a chea accredited institution, major in clinical research, what does not qualify, any degree not in clinical research (biology, psychology, public health, epidemiology, nursing, doctorate), graduate certificate programs.

The CCRA ® exam consists of 125 multiple choice questions that must be answered within 180 minutes.
The exam is referenced only to the international conference on harmonization (ich) guidelines. no other regulatory framework is tested, including country-specific regulations (i.e, fda or ema)., the following are the only references for which the ccra ® certification exam content can be supported:, ccra ® exam detailed content outline >, guideline for good clinical practice e6 (r2) >, definitions and standards for expedited reporting (e2a) >, general considerations for clinical trials (e8) >, statistical principles for clinical trials (e9) >, clinical trials in pediatric population (e11) >, the declaration of helsinki (doh) >, the global ccra ® exam committee uses psychometrically sound practices to develop certified clinical research associate (ccra) examinations that meet the current test specifications as determined by the most recent job task analysis (jta)..

Review the Detailed Content Outline and make sure your experience and work hours are appropriate, as outlined in the Eligibility tab.
We also strongly encourage you to review the entire acrp certification handbook , which provides full details about every facet of acrp certification..

Create a free ACRP account so you can begin the application process. Follow the on-screen prompts to enter any requested information and documentation.
If you already have an acrp account, please proceed to step three., create account >.

You’re almost there! Please note, applications selected for audit will undergo a formal review by ACRP’s subject matter experts. In most instances, you will receive a status update about your application within 7 business days.
In accordance with the americans with disabilities act, acrp will provide reasonable accommodations for candidates with disabilities. please complete this special accommodations form for submission with your application before proceeding., acrp’s testing partner psi offers in-person testing, as well as on-demand remote testing available 24 hours a day, every day, during the testing windows., watch these videos to learn what to expect from each option before scheduling your exam..

Find Test Centers Near You >
Schedule your in-person exam >.

Schedule Your Remote Exam >
Check system requirements >.

The best way to prepare for the CCRA ® exam is to fully understand the scope of the exam content and its references.
Please be sure to thoroughly review the following:, acrp certification handbook >, remember: the exam is referenced only to the international conference on harmonization guidelines. no other regulatory framework is tested, including country-specific regulations (i.e, fda or ema)..

We also recommend leaning on your community! Thousands of ACRP Certified members have been in your shoes. They are active community members and always willing to share tips and advice for ACRP exam prep.
Visit the acrp community >.

ACRP offers a variety of training and continuing education programs focused on the key ICH guidelines covered in the CCRA ® exam.
Learn more >.

Exam results are shared immediately at the conclusion of your exam, but PSI will send you an email with your full score report within 24 hours.
Your acrp account will reflect your results within 3 weeks of your exam date..

Congratulations! You just passed a major milestone on your professional journey and are now a member of the elite club of ACRP Certified clinical research professionals.
Keep an eye on your email because you will soon receive information from our digital badging partner credly about claiming your digital badge and how you can use it to tout your accomplishment. also learn how to use your new credential by reviewing the certification mark policy ., you have 2 years to keep your certification in good standing by continuing your professional development, and we’ll be right there with you every step of the way. in the meantime, we highly recommend you review all the details about maintenance of certification . don’t leave it to the last minute.

Don’t worry. It happens to the best of us. Give it another try!
Refer to your acrp certification examination results email or the acrp certification handbook for guidance on the next steps in your certification journey., upcoming testing dates, spring 2024 testing february 15 – may 15, 2024, fall 2024 testing july 15 – october 15, 2024, 2024 registration dates and fees, early bird registration acrp members – $435 nonmembers – $485 spring 2024: october 15 – december 31, 2023 fall 2024: may 15 – july 15, regular registration acrp members – $460 nonmembers – $600 spring 2024: january 1 – april 30 fall 2024: july 16 – september 30, join acrp & save, joining acrp helps you save money. more importantly, acrp is where you will find the very best of what you need to design a career path that’s uniquely your own. connections through an engaged community. growth through gold-standard training. and elevation through rigorous certification., explore membership >, exam preparation, congratulations on your decision to earn the most recognized and respected endorsement of clinical research competency — acrp certification. as you start this important journey in your career, we’re here to support you every step of the way..
- Program Overview
The SOCRA Certified Clinical Research Professional (CCRP) program is your gateway to excellence in clinical research. Elevate your career with our internationally recognized certification, tailored for professionals dedicated to upholding the highest standards in the field. Join a community committed to ethical practices, continuous learning, and advancing global health.
The Society of Clinical Research Associates (SOCRA) established the Certification Program for Clinical Research Professionals in order to create an internationally accepted standard of knowledge, education, and experience by which clinical research professionals will be recognized by the clinical research community. Those individuals so recognized may use the "Certified Clinical Research Professional" or "CCRP ® " designation.
Path to Certification
CCRP certification is awarded upon meeting two criteria: a successful written application and a passing CCRP examination score. The benefits of obtaining certification are numerous. It not only validates knowledge, skills, and abilities but also enhances credibility and peer recognition. Career advancement and increased earning potential become tangible outcomes, reflecting a commitment to standards, compliance, and integrity.

Scope and Standards of Practice
The standards upon which this certification program is based have been set forth by SOCRA to promote recognition and continuing excellence in the ethical conduct of clinical trials. It is the goal of SOCRA to encourage members, and assure the competency of certified members, in their knowledge, understanding, and application of the conduct of clinical investigations involving humans in accordance with the ICH Guidelines, the U.S. Code of Federal Regulations, and the ethical principles that guide clinical research. Members are expected to adhere to national, state, local and provincial regulations and to international guidelines published by the International Conference for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH) and all applicable federal, state and local laws and policies.
Standards of Practice include an understanding of and application of basic concepts of Good Clinical (Research) Practice, including:
- The Nuremberg Code
- The Belmont Report
- The Declaration of Helsinki
- 21 U.S. Code of Federal Regulations – Parts 11, 50, 56, 312, 812
- 45 U.S. Code of Federal Regulations - Part 46
- ICH Harmonised Guideline for Good Clinical Practice E6(R2), and
- ICH Clinical Safety Data Management: Definitions and Standards for Expedited Reporting (E2A)
- 42 CFR Part 11 (ClinicalTrials.gov)
Certification Exam
The SOCRA Certification Examination is offered in two formats: paper and pencil (at SOCRA sponsored sites), and computer based (at Prometric testing centers or through Home Proctoring).
SOCRA Sponsored Sites: Paper and Pencil
- Hosted exams offered in various location throughout the US and Canada.
- Visit the paper and pencil exam schedule for dates and locations.
- A complete application must be received by the deadline date as stated on the examination schedule.
- Score reports mailed to you in 4-6 weeks after exam.
Computer Based Testing: Testing Centers and Remote Proctoring
- Offered at Prometric testing centers throughout the world or through Home Proctoring
- Click here for a list of test centers.
- Allow 2-4 weeks for application processing.
- Once application is approved, schedule exam at a testing center. Exam sessions are available at least 6 weeks in advance.
- Score reports received immediately upon completion of exam.
Candidate Handbook
For more information, please view the Candidate Handbook.
Certification
- CCRP Certification Quick Facts
- Definition of a Clinical Research Professional
- Certification Program Policies
- Removal of CCRP® Credential
- Verify Certification
- Exam Overview
- Candidate Eligibility
- Application and Fee
- Computer Based Testing Exams
- Paper and Pencil Exams
- Refunds, Rescheduling and Retesting
- SOCRA Sponsored Exam Schedule
- Preparing for the Exam
- Preparation Resources
- Examination Results
- Host an Exam at Your Site
- Apply Online
- Exam Schedule SOCRA Sponsored Sites
- Requirements for Maintaining Certification
- Continuing Education Requirements
- Descriptions of Acceptable CE
- CE Recordkeeping Requirements
- Request for SOCRA CE for Courses / Workshops
- Installment Plan Payment
- Renewal of Certification
- Recertification Audit
- Recertification Learning Module
- Accreditation
Summary of Certification Activities
11,145 CCRPs (as of 12/31/2022)
- 1,391 candidates took CCRP exam
- 73% passed CCRP exam
- 2,649 CCRPs recertified
- 946 candidates took CCRP exam
- 65% passed CCRP exam
- 2,783 CCRPs recertified
- 2,060 candidates took CCRP exam
- 70% passed CCRP exam
- 3,801 CCRPs recertified
- 1,980 candidates took CCRP exam
- 71% passed CCRP exam
- 3,188 CCRPs recertified
- 104 exam sites hosted
- 2,175 candidates took CCRP exam
- 2,491 CCRPs recertified
- 91 exam sites hosted
- 2,141 candidates took CCRP exam
- 2,421CCRPs recertified
A day in the life of CRA (Clinical Research Associate)

Working as a Clinical Research Associate (CRA) can be a very exciting job that offers great opportunities to travel, work from home, meet new people and play a pivotal role in the drug development process.
As a CRA is an important role within drug discovery, we recently conducted an interview with a Clinical Research Associate who works for a global pharmaceutical organisation. We wanted to find out what a typical day is like, how did they become a CRA, what are the best and hardest bits about the role and where can this career take them.
We had a great interview and this is their viewpoint on what life is like working in a Clinical Research Associate job:
What does a Clinical Research Associate do?
The role of a CRA can vary between different companies. Typically, a CRA will set up, monitor and close-down clinical studies, which is what I do. The amount of studies and number of sites allocated to a CRA will depend on the complexity of the trial. I currently work on 3 studies across 2 therapy areas and have 13 sites to monitor across the UK. My nearest site is 15 minutes away and the furthest 3.5 hours.
What is a typical day like for a Clinical Research Associate?
I’m a field-based CRA so my office is at home. I typically spend 3 days a week working on-site with the Investigators and Nurses at the hospitals where the company has its trials running checking the data and 2 days at home, writing up reports, following up on emails and actions and the general admin side of things. Then there are the days where I go into head office for meetings. It’s quite a varied role!
How did you get into being a CRA?
I studied Human Biology at Leeds University and soon after graduating started out in the pharmaceutical industry as a Data Manager working for a small CRO. This was my foot in the door! I then moved into Drug Safety in a global pharmaceutical company where I stayed for a couple of years before I transferred internally into Clinical Research working as a Pharmacology Study Manager in Phase I clinical trials (Healthy Volunteer Studies). From there I moved into patient studies and have been working as a CRA for 4 years now.
What do you enjoy most about being a Clinical Research Associate?
I enjoy the variety of the role: the studies/therapy areas, traveling to the different hospitals and working with the Research Nurses. Although I work to monitoring plans and many deadlines, I organise my own time which is great!
What is the hardest part of being a CRA?
Trying to get an investigator to do something he doesn’t want to do – that can be a challenge! Also, sometimes, priorities on studies clash which can lead ‘interesting times’.
Why did you decide to pursue this career?
I really enjoyed studying my degree and wanted to make use of it and the pharmaceutical industry was just what I was looking for.
Once I’d got into the industry and tried my hand in a few areas, I knew I wanted to be a CRA but it can take time to get into it – experience is always required!
What can this type of job lead to?
Many CRAs go into Project Management. However, the pharmaceutical industry and the roles within it are vast so there are always different opportunities in areas to suit all from Clinical Operations to Regulatory to Medical Affairs, the list is endless.
What advice would you give to somebody considering Clinical Research Associate as a career?
Trainee CRA opportunities are few and far between these days so getting experience is key. Many CRAs start out as Clinical Trial Assistants or like me, get experience in other roles in the industry before transferring across. Also, Nurses who work on clinical trials often become CRAs. If you are still at university and have the opportunity do a year in industry, try and get a placement at a pharmaceutical company or CRO.
Please tell us anything else that you would like to add about your job…
A CRA needs to be well organised, meticulous and be able to prioritise as well as have a reliable alarm clock and a car with good lumbar support!
You may also like to read:
Clinical Research Associate job profile
A day in the life of a Clinical Research Nurse
A day in the life of a Clinical Data Manager
Advice centre

Free resources

Job profiles

Research and knowledge

Thanks for starting your application to {{companyName}}.
To complete your application you must do one of the following:
Forward an email from your mobile device with your resume attached to {{fromEmail}}
Reply to this email from your laptop or desktop computer with your resume attached.
Thank you for your interest, The Recruiting Team
Reply to this email from your laptop or desktop computer with your cover letter attached.
In order to create an account with us and submit applications for positions with our company you must read the following Terms and Agreements and select to agree before registering.
In the event that you do not accept our Terms and Agreements you will not be able to submit applications for positions with our company.
You agree to the storage of all personal information, applications, attachments and draft applications within our system. Your personal and application data and any attached text or documentation are retained by Jibe Apply in accordance with our record retention policy and applicable laws.
You agree that all personal information, applications, attachments and draft applications created by you may be used by us for our recruitment purposes, including for automated job matching. It is specifically agreed that we will make use of all personal information, applications, attachments and draft applications for recruitment purposes only and will not make this information available to any third party unconnected with the our recruitment processes.
Your registration and access to our Careers Web Site indicates your acceptance of these Terms and Agreements.
Dear ${user.firstName},
Thanks for choosing to apply for a job with ${client.display.name}! Please verify ownership of your email address by clicking this link .
Alternatively, you can verify your account by pasting this URL into your browser: ${page.url}?id=${user.id}&ptoken=${user.token}
Please note that your job application will not be submitted to ${client.display.name} until you have successfully verified ownership of your email address.
The ${client.display.name} Recruiting Team
Nice to meet you. 👋
Lets quickly set up your profile
to start having tailored recommendations.
You agree to the storage of all personal information, applications, attachments and draft applications within our system. Your personal and application data and any attached text or documentation are retained by Sequoia Apply in accordance with our record retention policy and applicable laws.
This career site protects your privacy by adhering to the European Union General Data Protection Regulation (GDPR). We will not use your data for any purpose to which you do not consent.
We store anonymized interaction data in an aggregated form about visitors and their experiences on our site using cookies and tracking mechanisms. We use this data to fix site defects and improve the general user experience.
We request use of your data for the following purposes:
Job Application Data
This site may collect sensitive personal information as a necessary part of a job application. The data is collected to support one or more job applications, or to match you to future job opportunities. This data is stored and retained for a default period of 12 months to support job matching or improve the user experience for additional job applications. The data for each application is transferred to the Applicant Tracking System in order to move the application through the hiring process. \nYou have the right to view, update, delete, export, or restrict further processing of your job application data. To exercise these rights, you can e-mail us at [email protected] . \nConversion Tracking \nWe store anonymized data on redirects to the career site that is used to measure the effectiveness of other vendors in sourcing job candidates.
Consent and Data Privacy
This application protects your privacy by adhering to the European Union General Data Protection Regulation (GDPR). Jibe will not use your data for any purpose to which you do not consent.\n
We request use of your data for the following purposes:\n \n User Authentication \n
\n This site retains personally identifiable information, specifically e-mail addresses, as a necessary part of user login. This data is retained for the duration of the user profile lifecycle and enables user authentication.\n
\n \n Usage Analytics \n
We store anonymized usage data to measure and improve the effectiveness of this CRM application in filling job requisitions and managing talent communities.\n
\n \n E-mails to Candidates \n
We collect your personal information such as name and email address. This information is used when you send marketing or contact emails to candidates.\n\n
Enter your email address to continue. You'll be asked to either log in or create a new account.
There was an error verifying your account. Please click here to return home and try again.
You are about to enter an assessment system which is proprietary software developed and produced by Kenexa Technology, Inc. The content in this questionnaire has been developed by Kenexa Technology, Inc., Kenexa’s Suppliers and/or Yum Restaurant Services Group, Inc.’s (“Company”) third party content providers and is protected by International Copyright Law. Under no condition may the content be copied, transmitted, reproduced or reconstructed, in whole or in part, in any form whatsoever, without express written consent by Kenexa Technology, Inc. or the applicable third party content provider. Under no circumstances will Kenexa Technology, Inc. be responsible for content created or provided by Company’s third party content providers.
IN NO EVENT SHALL KENEXA, AN IBM COMPANY, KENEXA’S SUPPLIERS OR THE COMPANY’S THIRD PARTY CONTENT PROVIDERS, BE LIABLE FOR ANY DAMANGES WHATSOEVER INCLUDING, WITHOUT LIMITATION, DAMAGES FOR LOSS OF VOCATIONAL OPPORTUNITY ARISING OUT OF THE USE OF, THE PERFORMANCE OF, OR THE INABILITY TO USE THIS KENEXA ASSESSMENT SYSTEM OR THE CONTENT, REGARDLESS OF WHETHER OR NOT THEY HAVE BEEN ADVISED ABOUT THE POSSIBILITY OF SUCH DAMAGES.
By clicking below, you are also confirming your identity for purposes of the questionnaire. You may not receive assistance, refer to any written material, or use a calculator (or similar device) while completing the questionnaire.
Unless otherwise directed by the Questionnaire Administrator, you are only authorized to take each requested questionnaire once. Failure to comply may result in disqualification. All Kenexa SelectorTM questionnaires are monitored.
A web browser is a piece of software on your computer. It lets you visit webpages and use web applications.
It's important to have the latest version of a browser. Newer browsers save you time, keep you safer, and let you do more online.
Try a different browser - all are free and easy to install. Visit whatbrowser.org for more information.
If you are using a later version of Internet Explorer, please make sure you are not in compatibility mode of an older version of the browser.
- Medpace.com Home
- Search Jobs
- About Medpace
- Explore Careers
- Life at Medpace
- My Career Portal
Cookies are used on this site to assist in continually improving the candidate experience and all the interaction data we store of our visitors is anonymous. Learn more about your rights on our Privacy Policy page.
Clinical Research Associate - 12 month FTC
- Location: UK
- Categories Clinical Monitoring
- __vacancyopjusttionswidget.opt-Business Area__ ICON Strategic Solutions
- __vacancyopjusttionswidget.opt-Remote Working __ Remote

TA Business Partner
- Icon Strategic Solutions
Send me a message
About the role.
Role: Clinical Research Associate - monitoring experience required (12 month FTC)
Location: UK (nationwide travel, must be happy to travel to Northwick Park regularly)
Fully sponsor dedicated
ICON plc is a world-leading healthcare intelligence and clinical research organisation. From molecule to medicine, we advance clinical research providing outsourced services to pharmaceutical, biotechnology, medical device and government and public health organisations. With our patients at the centre of all that we do, we help to accelerate the development of drugs and devices that save lives and improve quality of life. Our people are our greatest strength, are at the core of our culture, and the driving force behind our success. ICON people have a mission to succeed and a passion that ensures what we do, we do well.
As a Clinical Research Associate, you will be dedicated to one of our global Healthcare clients who have a strong legacy of innovation and leading the way within a specialist therapeutic area. They display an ongoing commitment to changing the lives of patients and the accessibility of products in order to change the lives of those that need them
The Clinical Research Associate is ultimately responsible for the successful management of investigator sites and monitoring activities for assigned clinical studies throughout the trial lifecycle.
Key responsibilities include:
- Full ownership of investigator sites for assigned studies, with responsibility for the successful management of the site right through to close-out, in accordance with the clinical monitoring plan.
- Developing effective relationships with investigator site staff to ensure that key clinical metrics are met.
- Ensuring integrity of clinical data through an ability to sufficiently maintain site tracking records and working diligently to relevant guidelines.
An experienced, ambitious self-starter looking for the opportunity to prove yourself in an embedded role at one of the largest pharmaceutical companies.
Here at ICON we want our employees to succeed and ensure that they are set up for this success through constant training, development and support.
To enable success in this position you will have:
- Prior experience of working in investigator site management, including conducting monitoring visits, from either a pharmaceutical company or a CRO environment.
- A working knowledge of ICH-GCP guidelines and local and international regulatory requirements is essential
- You will be educated to degree level in a life science discipline or be a licensed healthcare professional.
Why ICON? Our focus is to provide you with a comprehensive and competitive total reward package that comprises, not only an excellent level of base pay, but also a wide range of variable pay and recognition programs. In addition, our best in class employee benefits, supportive policies and wellbeing initiatives are tailored to support you and your family at all stages of your career - both now, and into the future. Our success depends on the knowledge, capabilities and quality of our people. That’s why we are committed to developing our employees in a continuous learning culture – one where we challenge you with engaging work and where every experience adds to your professional development. ICON, including subsidiaries, is an equal opportunity and inclusive employer and is committed to providing a workplace free of discrimination and harassment. All qualified applicants will receive equal consideration for employment without regard to race, colour, religion, sex, sexual orientation, gender identity, national origin, disability or protected veteran status. If, because of a medical condition or disability, you need a reasonable accommodation for any part of the application process, or in order to perform the essential functions of a position, please let us know. Interested in the role, but unsure if you meet all of the requirements? We would encourage you to apply regardless – there’s every chance you’re exactly what we’re looking for here at ICON whether it is for this or other roles.

ICON and you

ICON history

Career Pathways

Benefits & Rewards

Environmental, Social & Governance

Women in IT

A better career. A better world. A better you.
Day in the life

Teaser label
Content type
Publish date
To excel as a Clinical Research Associate (CRA) in a Clinical Research Organization (CRO), you need a combination of education, skills, and the right mindset. Brazil-based CRA II Debora shares her
Brazil-based CRA II Debora Oh shares her tips on how to become a great CRA and provides insight into life at ICON.

How to progress as a Clinical Research AssociateTo thrive as a Clinical Research Associate (CRA), it is imperative to cultivate a multifaceted skill set and demonstrate unwavering commitment to exce
Senior CRA Yemi Moses recounts her development and shares her career ambitions with ICON plc.

Senior Clinical Research Associate Suzaita Hipolito talks about the satisfaction gained from working in Clinical Research. “What would it look like to wake up every day feeling happy and fulfilled?
Senior Clinical Research Associate, Suzaita Hipolito, talks about how working in Clinical Research gives her happiness and fulfilment.

Suzaita Hipolito
Press play to find out more

Similar jobs at ICON
Clinical Monitoring
Remote Working
Business Area
ICON Strategic Solutions
Job Categories
Description
A Clinical Research Associate is a professional who contributes to accelerated drug/device/outcomes research through independent monitoring of studies to ensure patient safety and data integrity. You
2024-110200
Expiry date

Poland, Czech Republic, Slovakia, Spain
Czech Republic
To be an integral part of the study team by providing administrative and project tracking support to the Project Manager(s) and Clinical Trial Manager(s) with emphasis on supporting inspection readine
2024-110344

As Clinical Research Associate (CRA), you will be joining the world's largest & most comprehensive clinical research organization, powered by healthcare intelligence. A CRA is a professional who cont
2023-103904

As a Clinical Research Associate you will be joining the world’s largest & most comprehensive clinical research organisation, powered by healthcare intelligence.Clinical Research Associate Hangzhou of
2024-110421
2024-110420
2024-110419
Browse popular job categories below or search all jobs above

IMAGES
VIDEO
COMMENTS
Clinical Research Associate IV. 1 year initial contract. 100% remote - team is in Miami, FL. Hourly Pay Rate: $52.10/hr (client mandated for pay equity purposes) The Clinical Research Associate (CRA) for our global medical device client's team in Miami, FL is responsible for supporting clinical activities to generate clinical evidence that will ...
Clinical Research Associate. The first job on our list is a clinical research associate (CRA). CRAs are responsible for conducting and overseeing clinical trials. This job is perfect for individuals who love to travel as CRAs typically travel to different sites around the world to manage clinical trials. To become a CRA, you need a bachelor's ...
If you like to travel, the life of a Clinical Research Associate (CRA) is a blur of airports, frequent flyer miles, taxis and hotel rooms. This career requires so much travel that living near a major airport can help you land the plum jobs. The work of a CRA requires concentration and attention to detail.
Clinical research associate job requirements Enter the field as a Clinical Research Associate (CRA) with CCRPS's accredited training. Remote roles, $6,500-$12,000 monthly, and 33% annual promotion. 7-day CRA certification for a swift career start. ... (and some allow you to travel 45-75% with full expenses including travel, accommodation, meals ...
the travel clinical research associate (cra) role The Travel CRA is an important member of the Alira Health Clinical team. CRAs are highly motivated and function independently to conduct site ...
Date Added. 3. clinical research associate travel jobs. Travel Clinical Research Assistant, US Based (Chicago, IL) Care Access —Chicago, IL2.3. This role will perform a variety of clinical and administrative tasks in support of clinical trials and assist in the performance of clinical procedures to…. Estimated: $41K - $51.9K a year.
A clinical research associate works both at clinical sites and sponsor locations. During a trial, the CRA conducts regular site visits—virtually and physically—to ensure good progress and record-keeping on the clinical site. CRAs are often responsible for multiple trials at one time, meaning significant amounts of travel between these sites.
The ACRP offers the Certified Clinical Research Associate credential. To earn this certification, you must have one of the following: A bachelor's degree and at least 3,000 hours of experience as a CRA. A current CCRC, CPI or ACRP-CP certification and be able to substitute 1,500 hours of work experience.
Here's how to get started as a clinical research associate. 1. Qualify for certification. You can take several paths to becoming a certified CRA in Canada. One path is to earn a high school diploma and clock 3,000 to 3,500 part-time hours of work experience in the field.
Check out this video where I briefly discuss the typical CRA travel requirements.Like, subscribe, comment, and turn on your notification to get more informat...
Posted 2:59:58 PM. Travel Clinical Research Associate Date: September 8, 2023Reports to: Lead…See this and similar jobs on LinkedIn.
Clinical research is a branch of healthcare science that determines the safety and effectiveness (efficacy) of medications, devices, diagnostic products, and treatment regimens intended for human use. ... Make the most of the travel but also make sure you are getting involved in other things as well because many people burnout quickly when they ...
ACRP Certification is an ideal solution for managers looking to enhance team knowledge, grow leaders, and signify to others that their study teams are among the best of the best. Clinical research professional with 3,000 hours of verifiable work experience are eligible to sit for the CCRA ® Exam. Complete eligibility criteria is defined in the ...
The SOCRA Certified Clinical Research Professional (CCRP) program is your gateway to excellence in clinical research. Elevate your career with our internationally recognized certification, tailored for professionals dedicated to upholding the highest standards in the field. Join a community committed to ethical practices, continuous learning ...
A day in the life of CRA (Clinical Research Associate) Working as a Clinical Research Associate (CRA) can be a very exciting job that offers great opportunities to travel, work from home, meet new people and play a pivotal role in the drug development process. As a CRA is an important role within drug discovery, we recently conducted an ...
Clinical research is a branch of healthcare science that determines the safety and effectiveness (efficacy) of medications, devices, diagnostic products, and treatment regimens intended for human use. ... When I travel for pleasure (or, at least pre-COVID), I always booked FC though, since I had ~gotten used to it~. Reply reply More replies.
Job Summary . Join our team in Cincinnati, Ohio!. The Clinical Research Associate position at Medpace offers the unique opportunity to have an exciting career in the research of drug and medical device development while making a difference in the lives of those around them. For those with a medical and/or health/life science background who want to explore the research field, travel the US, and ...
Role: Clinical Research Associate - monitoring experience required (12 month FTC) Location: UK (nationwide travel, must be happy to travel to Northwick Park regularly) Fully sponsor dedicated. ICON plc is a world-leading healthcare intelligence and clinical research organisation. From molecule to medicine, we advance clinical research providing ...
Gold Medal Award. Mary Phillips, MD, MD (Cantab) (Distinguished Professor of Psychiatry, Clinical and Translational Science, and Bioengineering, and Pittsburgh Foundation-Emmerling Endowed Chair in Psychotic Disorders), received the 2024 SOBP Gold Award. The Gold Award honors pioneering contributions to the field, as well as significant and sustained work that advances and extends knowledge in ...